Hydrocodone bitartrate is used as a cough suppressant. After the drug is fully absorbed, the quantity of drug in the body decreases at a rate proportional to the amount left in the body, with constant of proportionality k. Suppose that the half-life of hydrocodone bitartrate in the body is 3.7 hours, and that the oral dose taken is 8 mg. Write a differential equation for the quantity, Q, of hydrocodone bitartrate in the body at time t, in hours, since the drug was fully absorbed. Write your answer in terms of Q, t, and k, e.g., Q' = 3t(1 – kQ). Solve your differential equation, assuming that at t = 0 the patient has just absorbed the full 8 mg dose of the drug. Your solution should include the constant of proportionality k, but should not include any other unspecified constants. Use the half-life to find the constant of proportionality, k. k =
Hydrocodone bitartrate is used as a cough suppressant. After the drug is fully absorbed, the quantity of drug in the body decreases at a rate proportional to the amount left in the body, with constant of proportionality k. Suppose that the half-life of hydrocodone bitartrate in the body is 3.7 hours, and that the oral dose taken is 8 mg. Write a differential equation for the quantity, Q, of hydrocodone bitartrate in the body at time t, in hours, since the drug was fully absorbed. Write your answer in terms of Q, t, and k, e.g., Q' = 3t(1 – kQ). Solve your differential equation, assuming that at t = 0 the patient has just absorbed the full 8 mg dose of the drug. Your solution should include the constant of proportionality k, but should not include any other unspecified constants. Use the half-life to find the constant of proportionality, k. k =
Principles of Modern Chemistry
8th Edition
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Chapter18: Chemical Kinetics
Section: Chapter Questions
Problem 6P
Related questions
Question
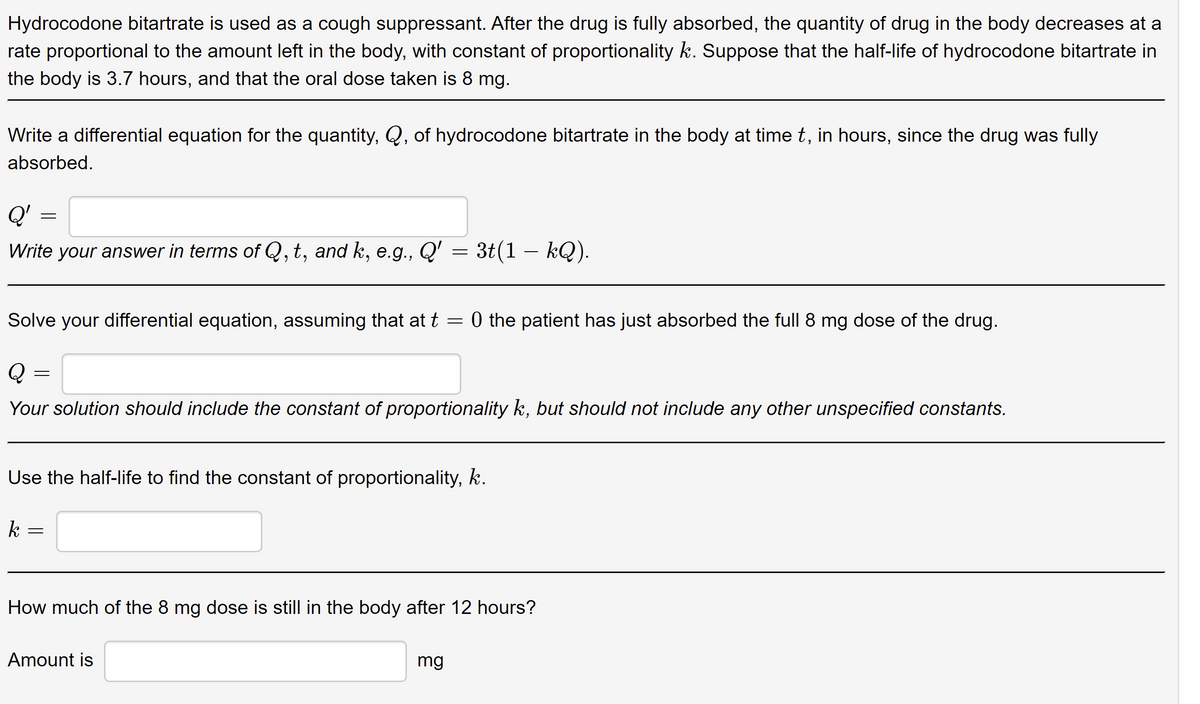
Transcribed Image Text:Hydrocodone bitartrate is used as a cough suppressant. After the drug is fully absorbed, the quantity of drug in the body decreases at a
rate proportional to the amount left in the body, with constant of proportionality k. Suppose that the half-life of hydrocodone bitartrate in
the body is 3.7 hours, and that the oral dose taken is 8 mg.
Write a differential equation for the quantity, Q, of hydrocodone bitartrate in the body at time t, in hours, since the drug was fully
absorbed.
Q'
Write your answer in terms of Q, t, and k, e.g., Q' = 3t(1 – kQ).
Solve your differential equation, assuming that at t = 0 the patient has just absorbed the full 8 mg dose of the drug.
Q
Your solution should include the constant of proportionality k, but should not include any other unspecified constants.
Use the half-life to find the constant of proportionality, k.
k =
How much of the 8 mg dose is still in the body after 12 hours?
Amount is
mg
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 6 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning