Q: During hot days, fish stay at the deeper cooler part of a body of water. Can you explain this…
A: Answer: In a solution substance that is in lower quantity is called as solute and that is in excess…
Q: Jeff followed to procedure for Experiment 4, and collected the following data, which he recorded on…
A:
Q: What is the reactant that is classified as Bronsted-Lowry acid? CO3²¯(aq) + H₂O(l) ↔ HCO3¯(aq) +…
A: Bronsted Lowry acid is a substance which donates a proton to other compound and form conjugate base.…
Q: 23. Assuming that the heat capacity of a 10 g substance to be 42 J/K, the heat required to raise i…
A: Thermodynamics is branch of chemistry in which we deal with amount of heat evolved or absorbed by…
Q: Classify each of the following as a Lewis acid or a Lewis base. Drag the appropriate items to their…
A: We have to classify the given species as Lewis acid and Lewis base. Introduction: Lewis acid: A…
Q: What volume of ClF3 will be produced when 75.0ml of F2 reacts with 25.0 mL of Cl2 according to the…
A:
Q: CARBON -14 DATING The skeletal remains of the so-called Pittsburgh Man, unearthed in Pennsylvania,…
A: Radioactive decay follows first order kinetics. Half-life time, t1/2 of C-14 = 5730 years For first…
Q: A certain weak acid, HA, has a Ka value of 4.5x10-7. Part A Calculate the percent ionization of HA…
A:
Q: 9. 2-Nonacontyne is a 90-carbon alkyne. Its formula is: a. C90H182 b. C90H180 C. C90H178 d. C90H135
A: General formula for alkyne ( are the hydrocarbons which contain carbon carbon triple bond )is…
Q: A chemical engineer is studying the following reaction: H₂(g) +1₂(g) → 2 HI(g) At the temperature…
A: The direction in which the reaction proceeds to reach equilibrium can be predicted by determining…
Q: For which one of the following reactions is the value of AH°rxn equal to AHf for the product? O2Ca…
A:
Q: Mg(s) + 2HCl(aq) → MgCl2 + H2(g) You will measure the rate of the reaction using a continuous…
A: Rate of reaction can be obtained by monitoring how fast/slow a reactant is being used up or how fast…
Q: Question 5 of 32 Determine the value of Ksp for Mg(CN), by constructing an ICE table, writing the…
A: Given,The molar solubility of MgCN2 is 1.4 × 10-5 MRequired, The value of Ksp for…
Q: What happens to the reaction below when more A is added to the reaction mixture? A (g) + 4B (g) ⇌…
A: The question asked about the effect of adding more A to a chemical reaction mixture, which is an…
Q: Write reasons for the regioselectivity of hydroboration-oxidation of alkene by drawing the reaction…
A: Hydroboration is the addition of borane to alkene, alkyne to form the alkyl borane, alkenyl borane.…
Q: - ] 3 Consider the reaction 1205(g) + 5 CO(g) → 5CO2(g) + 2(g) 80.0 grams of iodine pentoxide, 1205…
A: Number of moles is calculated by dividing the given mass with molecular mass. In the given question…
Q: 7. Which of the following solutes would create the most change in the boiling point and freezing…
A: Since you have asked for question number 7 only, we are providing you a solution for it only. If you…
Q: 1. A) Starting with the following line angle structure, convert the structure to a Fischer…
A: To assign R/S nomenclature first decide priority order according to CIP rule. Then move from…
Q: Draw the major product of this reaction. Ignore inorganic byproducts. + AICI 3 CI a Q
A: In these two questions, we will draw the structure of the major organic product. You can see the…
Q: Curved arrows are used to illustrate the flow of electrons. Using the provided arting and product…
A:
Q: 11. The ester pictured below could be synthesized from: a. Ethanol and butanoic acid b. Butanol and…
A: Ester can be synthesized with the reaction of an acid with an alcohol in acidic or basic condition.…
Q: Identify suitable reactants and reagents that can be used to produce the following compound using a…
A: The Claisen condensation is a powerful organic reaction used to form carbon-carbon bonds in the…
Q: Calculate the molarity of hydrochloric acid solution if 88.75 mL of this solution are needed to…
A: Given that, The volume of the HCl, acid is Va = 88.75, The volume of the NaOH, base is Vb = 54.34…
Q: What is the pH of a 0.420 M solution of CsHeNHBr (Kb of CsHaN is 1.7 x 10-9?
A: pH of a salt of weak base and strong acid having concentration C given as pH = 7– (pKb +log C) /2
Q: How many grams of hydrogen are in 36.56 g of propane, C3H8
A: According to the mole concept, in terms of mass, the amount of substance in moles is equal to the…
Q: Which of following compounds can undergo a rearrangment after forming the carbocation intermediate?…
A: Carbocation shows rearrangement for greater stability. +I effect, resonance , aromatisation,…
Q: What will be the major organic product of this reaction? A A COOH Тсоон CHO CHO E CHO 1) KMnO4, OH,…
A: We have find out the answer
Q: Name the following compounds: ---- H H T H-C-C-C-H | H ي H C-H | H-C-H H ن
A: we have to name the given structure
Q: 3. Complete con ecuaciones balanceadas: a. CH₂ (CH₂) 16COO™ b. CH, (CH₂) 12COO- c. Na₂B₂0₁ CH₂O d.…
A: Balanced the given chemical reaction
Q: 31) "When a nonvolatile solute is dissolved in a liquid to form a solution, vapor pressure…
A: The boiling point is a physical property and it depends on van't Hoff factor. With the increase of…
Q: You have been contracted to determine how different salts affect the pH of water. Which of the…
A: Answer: When a salt of weak base and strong acid is placed in water it generates H3O+ through…
Q: Option 1 These two solutions have the same boiling point. 25) Which aqueous solution has a higher…
A:
Q: 21. Draw the predicted product(s) of each reaction. Naming the compounds is not necessary. a. ОН b.…
A: Given that, the reactions are shown below We have to give the product of the above reactions.…
Q: Curved arrows are used to illustrate the flow of electrons. Using the provided starting and product…
A:
Q: Draw and discuss in details the Pourbaix plot representing the following reactions with a…
A: A Pourbaix diagram is a plot of the equilibrium potential of electrochemical reactions against pH.It…
Q: provide the IUPAC name of the product when 2-butanol undergoes oxidation
A:
Q: 12. The reaction between a carboxylic acid and an amine is an example of: a. addition C.…
A:
Q: Write a balanced chemical equation based on the following description: aqueous iron(III) chloride…
A: element has an equal number of atoms in the reactant and product side. Therefore, the balanced…
Q: 5) each carbon atom. Subane C8H8 (s) is a cubic-shaped hydrocarbon with a carbon atom at each corner…
A: Cubane is a highly unstable hydrocarbon that has a cubic structure ( carbon atom at each of its…
Q: A certain reaction is second order in N₂ and first order in H₂. Use this information to complete the…
A:
Q: What is the concentration of salicylate in mg/dL in an unknown salicylate solution whose absorbance…
A: Graph is
Q: Use the drop-down selection boxes to rank the IR absorptions of the coloured bonds in terms of…
A: IR spectroscopy is mainly used for identification of functional group present in the molecules. Each…
Q: How would you differentiate a sodium chloride solution from a sugar solution? Explain your answer.
A: Note: Since you have asked multiple questions, we will solve the first question for you. If you want…
Q: Study this chemical reaction: Zn + Cl₂ → ZnCl₂ Then, write balanced half-reactions describing the…
A:
Q: Calculate the specific heat capacity of copper, given that 205.65 J of energy raises the temperature…
A:
Q: UESTION 3 alculate the molar mass of potassium chloride, KCI. 6.745 g/mole 54.55 g/mole 19.00 g/mole…
A:
Q: The preparations of two aqueous solutions are described in the table below. For each solution, write…
A: A chemical reaction occurs between an acid and a base to give salt and water. This reaction is known…
Q: (21-25). Given that the vapor pressure of pure water at 30°C is 31.82 mm Hg, calculate the vapor…
A: The question is based on the concept of colligative property. relative lowering in vapour pressure…
Q: Two 25.0-mL samples, one of 0.100 of HCI and the other 0.100 M of HF are each titrated (separately)…
A: Answer: During the titration of acid HA and base BOH below shown reaction takes place:…
Q: 3. In the calorimeter which component do you think is gaining heat (metal or water)? Explain how you…
A: Please find solutions of question 3 & 4. 3. In the calorimeter which component do you think is…
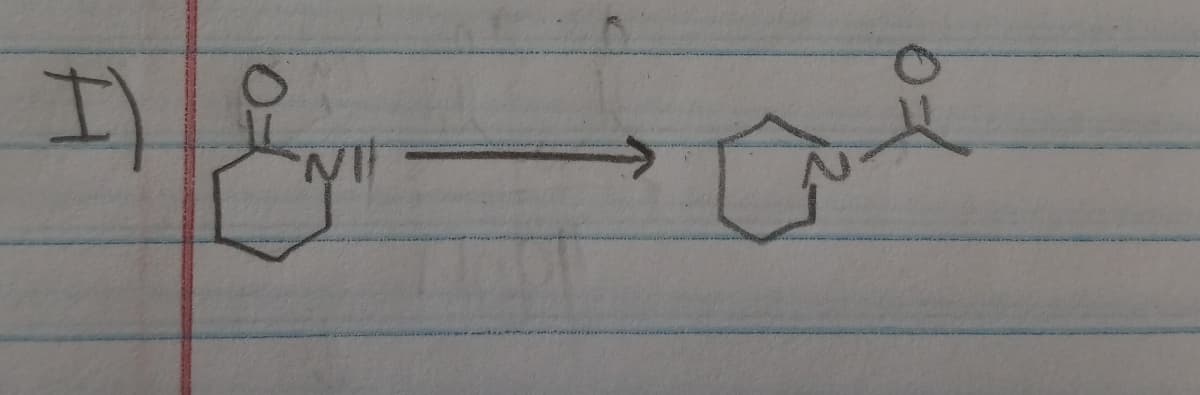
Step by step
Solved in 2 steps with 1 images


