Q: 2)Write a balanced chemical equation for each of the following processes, Structural or displayed fo...
A: Since you have posted a question with multiple sub-parts,we will solve first three sub-parts for you...
Q: standard deviation
A: If there is two results having standard deviation Sa and Sb. Therefore standard deviation indicate t...
Q: 1. Br2 Mg SOCI2 КОС(СН)з 1. ВНз Et20 pyr 2. H2O2,OH FeBr3 2. H3O* Br2 КОН 1. Na HNO3 Fe 2. EtBr H2SO...
A: All are multi step synthesis reaction . To find out the major final product detail synthetic route i...
Q: YOur onswer wilh four decimal paces and enclose them a box Solve s neally an possible Messy work wil...
A: Step 1 Here a chloride ion is added in a solution containing 4.144g/L of Pb2+ and 1.088g/L of Ag+ W...
Q: Which of the following statements correctly describe the peptide bond hydrolysis reaction? [Select a...
A: We have to predict the correct statement about peptide hydrolysis.
Q: 8
A:
Q: If you have a solution with 1 mole of acetic acid HC2H3O2, which of the following can be added to ma...
A: We have to predict the chemical added to make a buffer solution.
Q: What happens to your muscles when they get cold? Does that have implications for adding previously f...
A: Given:- What happens to your muscles when they get cold?Does that have implications for adding previ...
Q: Identify the reagents nacessary to accomplish cach of the following transformations. Choose the corr...
A:
Q: Give each detailed examples of the five conditions of chemical changes 1) color change 2) formatio...
A: 1) color change : When the lead nitrate solution and potassium iodide solution react together then t...
Q: e pressure
A:
Q: The Zn electrode is the
A: In cell notation left side written are anode half cell and right side of species after salt bridge a...
Q: Determine the solubility of AgBr in A. Water B. 6.0x10^-2M AgNO3 C. 0.20M NaBr
A:
Q: 2. For the first part of the lab, the following graph was produced. 0.6 Absorbance Curves for BCG ba...
A:
Q: 4. What mass of HCI(g) must be dissolved in 1.50 Lof a NaOH solution having a pH of 11.176 to produc...
A:
Q: 1. a USE AN ICE TABLE METHOD To DETERMINE THE RELATONSHIP BETWEEN THE Kse VALUE AND THE MOLAR SOLUBI...
A:
Q: The preparations of two aqueous solutions are described in the table below. For each solution, write...
A: Concept: An acid is defined as a substance whose water solution tastes sour, turns blue litmus red a...
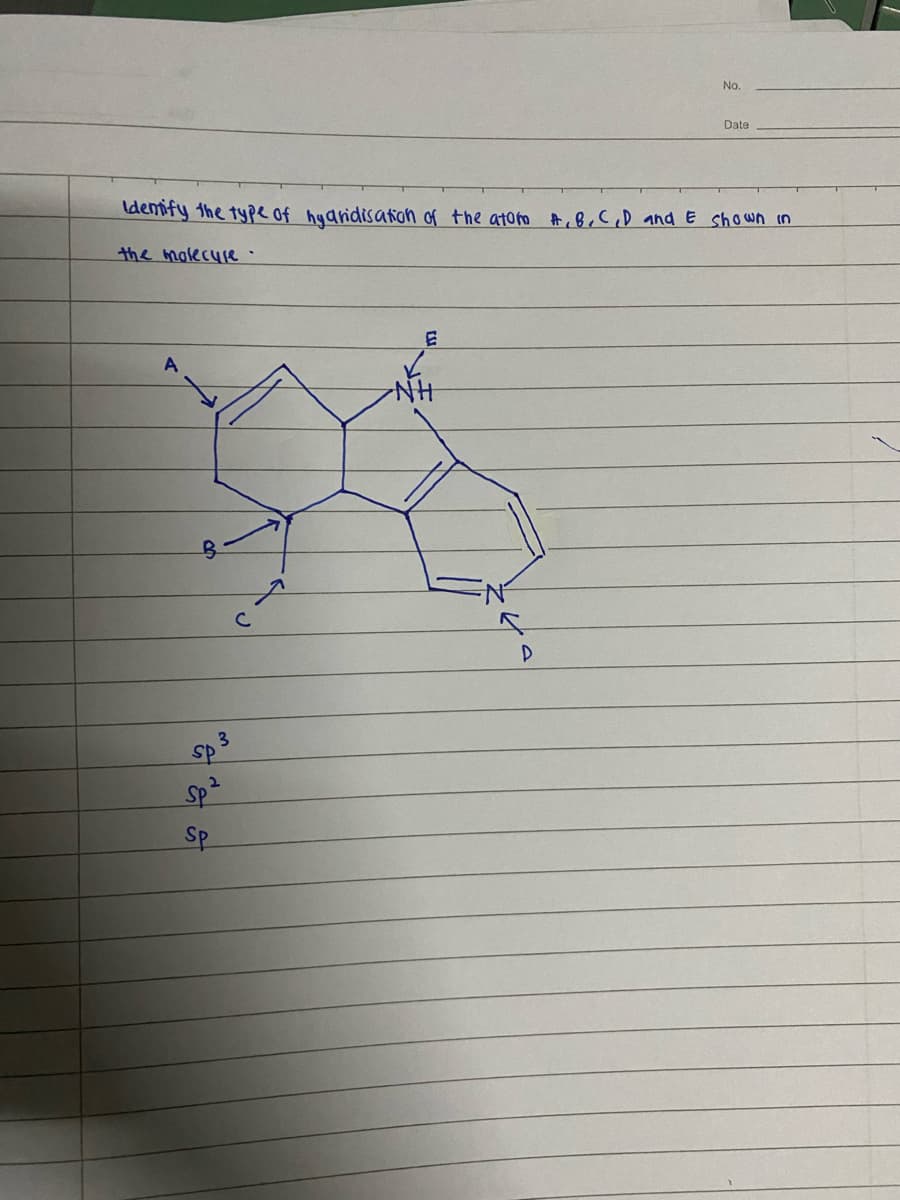
Q: Name the following compound:
A: IUPAC name= prefix+Root word+Suffix I+ suffix ii
Q: Write the balanced combustion equation. Record fractions as ratios (e.g. 1/2) if needed. Include agg...
A: Since you have posted multiple questions, the answer for first question is given below. Kindly repos...
Q: Sometimes in lab we collect the gas formed by a chemical reaction over water (see sketch at right). ...
A:
Q: Q.8. Propose mechanisms for the following reactions. OH MgBr 1. 2. H* b) LO HO
A:
Q: A 9.00 L tank at 27.3 °C is filled with 16.7 g of dinitrogen monoxide gas and 19.1 g of dinitrogen d...
A: Given :- mass of dinitrogen monoxide = 16.7 g mass of dinitrogen difluoride = 19.1 g volume = 9.0...
Q: In the combustion of heptane, CO2 is produced Assume that you want to produce 600kg of dry ice per h...
A:
Q: ce the reagents below in the correct order necessary to accomplish the transformation shown in the b...
A:
Q: An indicator has a pk, of 7.63. In a solution with a pH of 9.35, the color in the solution will be d...
A:
Q: 9. Show the dipole moment of each bond and then predict the overall molecule as either being polar o...
A:
Q: major product result when hexanone is reacted with each of the following. a) excess CH,OH and trace ...
A:
Q: 1. Draw the Lewis dot structures for the following imaginary elements a. X, an element from the Grou...
A: While drawing Lewis dot structure for an element only valence electrons are taken into consideratio...
Q: temperature
A:
Q: Consider the chrom 6 M KOH are added after the addition of hydrochloric acid in the last step of the...
A:
Q: In the lab, a student generates 0.450 g of a product in an experiment. The student then calculates t...
A: Given :- product obtained from an experiment = 0.450 g Theoretical yield of product = 0.398 g To d...
Q: What are the accompanying changes of: A. Crushing a can B. Shredding paper C. Chopping wood D. Melt...
A: There are two type of changes occur in the universe: Physical change and chemical change.
Q: State the orbital angular momentum of an electron in the following orbitals: a. 4d b. 3p Give the nu...
A:
Q: Question 8: Identify each of the following disubstituted cyclohexane rings as either cis or trans. D...
A:
Q: A 6.00 L tank at -2.1 °C is filled with 17.3 g of boron trifluoride gas and 13.4 g of dinitrogen mon...
A:
Q: A sodium hydroxide solution has a pOH of 3.11. Determine the [H"] concentration in this solution.
A: Given, pOH of sodium hydroxide (NaOH) solution = 3.11 Concentration of H+ ion = [H+] = ? The formula...
Q: Ammonia and oxygen react to form nitrogen and water, like this: 4NH, (g)+30,(g) - 2N,(g)+6H,O(g) Sup...
A: Given:- Here we have to think about the concept of Le-Chatlier's principles which are used to expla...
Q: Propose an efficient synthesis for the following transformation: он ?. OH The transformation above c...
A:
Q: Given the dissolution of silver phosphate: A93PO4 3 Ag* + PO43- Which of the following conditions wi...
A: The equilibrium reaction given is Ag3PO4 ⇌ 3 Ag+ + PO43– ----------- (1)
Q: What is the activation energy of the reaction? Number Units What is the frequency factor for the rea...
A: Arrhenius equation is: k = A e-Ea/RT Where, k = rate constant, Ea = Activation energy, R = Gas cons...
Q: moles
A:
Q: What is the IUPAC name for the following compound?
A: Given ; structure of molecule
Q: how many grams of CO2 are present in 8x10^12 molecules of CO2?
A:
Q: Give the description of accompanying changes of: A. Crushing a can B. Shredding paper C. Chopping w...
A: Change in physical properties include change in state of matter, shape, size, volume, length and col...
Q: C2H5Cl(g)---->C2H4(g)+HCl(g) is the first order in C2H5Cl.The rate constant is 1.60 x 10-6/s.In an i...
A: Let initial conc = A conc. at time t = a
Q: For the acid HA (where Ka >> 1), what is the pOH at 25o C if the initial HA concentration is 0.047 M...
A: Given: concentration of HA = 0.047 M To find: pOH = ?
Q: ).(a) Draw Newman projections for the anti, gauche and eclipsed conformers 3(1 of 3,3-dichlorohexane...
A: Anti: Dihedral angle between two ethyl groups is 180 degrees Gauche: Dihedral angle between two ethy...
Q: Me Et Ме Me' Cl2 CH:OH
A:
Q: 25 nm, which çan be used for the spectrophotometric determination of ammonia. OCF 2 OH + NH3 indophe...
A: Solution -
Q: If I have an unknown quantity of gas at a pressure of 0.99 atm, a volume of 13 liters, and a tempera...
A: Given data from the question, P=0.99 atm V=13 L T= 47℃ = (47+273) K = 320 K no. of moles n = ?
Step by step
Solved in 2 steps with 1 images

- A natural products chemist isolated the sphingolipids shown at the right(alpha-GalCerBf) as the mixture of compounds that differed in their chain lengths. The structures shown, including the branching pattern, were determined NMR. The table below includes the found high-resolution masses of the three major isomers (negative mode,{M-H}-). Fill in the calcualted masses and the error ppm.Define F–centre. Mention its one consequence.please help me solve tghe following question and explain pls, its important, thanks!!
- Giventhefollowingsetofreactionsandtheirenthalpies,drawtheBorn-Habercyclefortheformationof LiCl(s)anddeterminethelatticeenergyforLiCl.Reactions:H (in kJ)(1)Li(s)Li(g);Hs= 161;(2) Li(g)Li+(g)+e–;IE= 520;(3) ½Cl2(g)Cl(g);½BE =121.5;(4)Cl(g)+ e–Cl–(g);EA= –349;(5)Li+(g)+ Cl–(g)LiCl(s);UL= ?(UL= latticeenergy)(6) Li(s)+ ½ Cl2(g)LiCl(s);Hof=–408.6._______________________________________________________________________________154- please don’t copy from chegg because they have a wring answer and unclear processes thank you :)a) Draw tge HY^3- species of EDTA to illustrate that it is a Zwitterion. b) Explain the function of a SEM by drawing a sketch and giving caption for it.
- 1 - bir elektron bir ‘tuzak’a düştüğünde meydana gelen birleşmeye ne ad verilir? What is the combination that occurs when an electron falls into a "trap"? A) dislokasyon dislocation B) foton photon C) Tuzak destekli rekombinasyon Trap assisted recombination D) hiçbirisi none of them E) fonon phononIn which solution will 0.000015 BaCl2 form a participate?a. 7.0x10^-7 M H2SO4 (ksp of BaSO4 = 1.1x10^-10)b. 0.10 M Pb(NO3)2 (ksp of PbCl2 = 1.7x10^-5)c. 0.010 M NaF (ksp of BaF2 = 1.7x10^-6)d. 5.0x10^-7 M AgNO3 (ksp of AgCl = 1.8x10^-10)Solve for the Keq of these 2 successive reactions at 25C. (Glcose-1-phspte to glcose-6-phspte G'= -1.75Kcal) (Glcose-6-phsphte to frctose-6-phspate G'= -0.40Kcal) at pH7

