Identify all of the conditions that would facilitate the following conversion. > CI AICI3 1. , AICI 3 2. NaBH4, MeOH 1. CI AICI 3 2. NaBH, MeOH 1. Cl , AICI 3 2. Zn(Hg). HCI 1. , AICI 3 2. Zn(Hg), HCI only product Give detailed Solution with explanation needed with reaction. don't give Handwritten answer
Identify all of the conditions that would facilitate the following conversion. > CI AICI3 1. , AICI 3 2. NaBH4, MeOH 1. CI AICI 3 2. NaBH, MeOH 1. Cl , AICI 3 2. Zn(Hg). HCI 1. , AICI 3 2. Zn(Hg), HCI only product Give detailed Solution with explanation needed with reaction. don't give Handwritten answer
Chapter31: Introduction To Analytical Separations
Section: Chapter Questions
Problem 31.21QAP
Related questions
Question
None
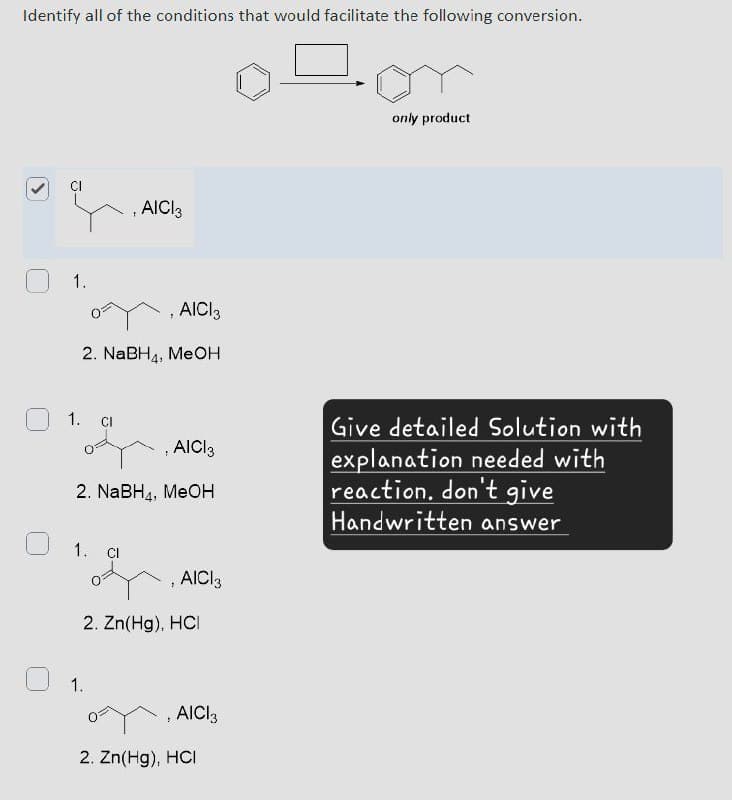
Transcribed Image Text:Identify all of the conditions that would facilitate the following conversion.
>
CI
AICI3
1.
, AICI 3
2. NaBH4, MeOH
1.
CI
AICI 3
2. NaBH, MeOH
1.
Cl
, AICI 3
2. Zn(Hg). HCI
1.
, AICI 3
2. Zn(Hg), HCI
only product
Give detailed Solution with
explanation needed with
reaction. don't give
Handwritten answer
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you



