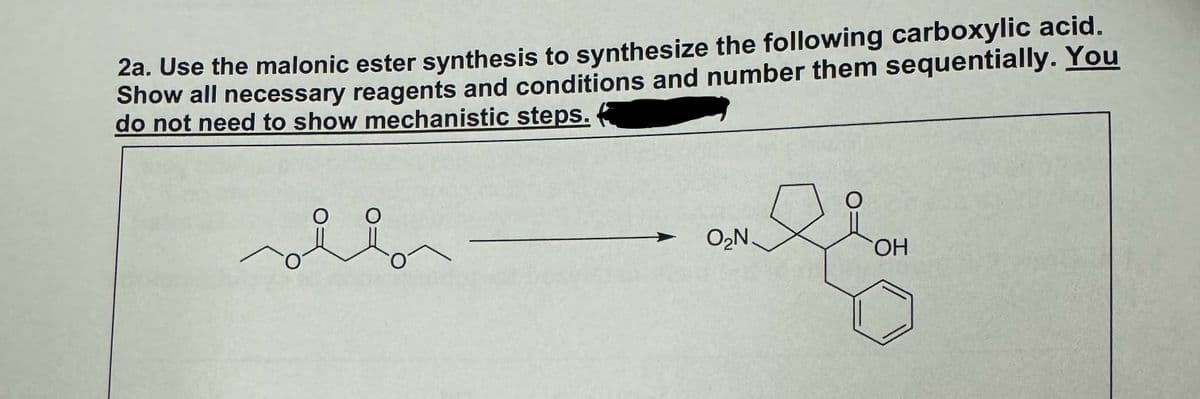
2a. Use the malonic ester synthesis to synthesize the following carboxylic acid. Show all necessary reagents and conditions and number them sequentially. You do not need to show mechanistic steps. O O O₂N OH
Q: A 3.55 gram sample of an unknown gas is found to occupy a volume of 1.72 L at a pressure of 805 mm…
A: The objective of the question is to find the molecular weight of an unknown gas given its mass,…
Q: Identify unknown diatomic gas that filled a gas-tight cylinder to 33.5 mL, at pressure of 2.41 atm…
A: The objective of the question is to identify the unknown diatomic gas by calculating its molar mass…
Q: CH Bra ? Modify the given carbon skeleton to draw the major product(s) of the given reaction. If a…
A: The reaction between ethenylcyclohexane and Br2 proceeds via an electrophilic addition mechanism.…
Q: 1. Which reaction most certainly results in a negative ASrxn? a. H₂O(g) b. CaCO3(s) H₂O(s) Cao(s) +…
A:
Q: Propose a series of synthetic steps by which each of the following transformations can…
A: Step 1: Step 2: Step 3: Step 4:
Q: A 0.760-g sample of an unknown gas has a volume of 759 mL and a pressure of 399 mm Hg at 34.2 °C.…
A: The objective of this question is to calculate the molar mass of an unknown gas given its mass,…
Q: || wwwwww wwwwwwww Benzophenone dissolved in ethanol (EtOH) reacts with 2, 4-dinitrophenylhydrazine…
A:
Q: 20
A: b , c, d and e in all cases there is alpha hydrogen(hydrogen attached to the nest to the carbonyl…
Q: synthetic polymers: Draw AIBN + heat or light.
A: Step 1: Step 2: Step 3: Step 4:
Q: Write down the oxidation state of each of the labeled atoms in this molecule: HO C-C HICH OH O H H…
A:
Q: Imagine gaseous Kr at 298K confined to move in a two-dimensional plane of area 4.00 cm2. What is the…
A: To calculate the translational partition function q_trans for krypton gas in a two-dimensional…
Q: Problems: 1. Provide a mechanism to explain the following transformation. OEt منذ EtO + als NaOEt…
A:
Q: Consider this step in a radical reaction: о hv ? What type of step is this? Check all that apply.…
A: Thank you.
Q: Predict the structure of the product of each of these reactions. If no reaction occurs write NR…
A: The final product is a carboxylic acid. Aldehydes can be converted to carboxylic acids when it is…
Q: draw out the mechanism of the following reaction. be sure to draw all intermediates and use arrows…
A: This reaction is the conversion of GTP (Guanosine triphosphate) +IMP + L-Aspartate into GDP…
Q: How many 1H NMR signals do you expect to see in the following molecule? 6 1 8 5
A:
Q: 1. What compounds are in the following? Sodium bicarbonate Baking powder Baker's ammonia 2. What is…
A: What compounds are in the following?Sodium bicarbonate: It is composed of sodium ions (Na+) and…
Q: Calculate and balance of equations that show how the substance dissolves in water.
A: The objective of the question is to balance the equations that show how the given substances…
Q: A patient ingests 1.42 μg�g of 131I (iodine-131), a beta emitter with a half-life of 8.0 days.…
A: Convert the initial activity from micrograms (μg) to disintegrations per second (dps):We know that 1…
Q: Please help me with the question below. A detailed explanation to aid in understanding is welcome.
A: Explanation is along with answer. *****GIVE HELPFUL RATING*****
Q: The balanced reaction of oxalic acid with potassium dichromate in an acidic solution is: 3H2C2O4(aq)…
A:
Q: HNO3, H2SO4
A:
Q: NaNH + Glue the starting material NO₂ NH2
A:
Q: o Four amino acids are shown. COO Macmillan Learnin COO COO COO CH2 -NH3 H3N- H H -NH3 H3N- -H CH…
A: When the amino group (NH2) in amino acid is present on the left side of the Fisher projection, then…
Q: Please don't provide handwritten solution ...
A: Approach to solving the question: Avogadro's Law: Volume of a gas is directly proportional to the…
Q: Consider each of the following molecules and/or ions. Give the hybridization for the central atom in…
A: Steric number helps predict the hybridization, molecular geometry, and electron geometry of a…
Q: Draw one of the two stereoisomers of the major product from this reaction. Use a dash or wedge bond…
A:
Q: A 0.175 mol sample of N2 gas is contained in a 4.00 L flask at room temperature and pressure. What…
A: The objective of this question is to calculate the density of nitrogen gas under given conditions.…
Q: Draw a detailed mechanism, using curved arrow notation,for the synthesis of 3-carboxy-umbeliferone…
A: Step 1: Step 2: Step 3: Step 4:
Q: 2) Predict the product of the following reaction and provide a step-by-step mechanism for this…
A: The given reaction involves the substitution of a bromine atom with a carboxylate anion. This…
Q: how to get 1-(1-methyl-3,4-dihydro-1H-isoquinolin-2-yl)ethanone from…
A: 1. Starting Material Analysis- **N-ethenyl-N-(2-phenylethyl)acetamide** has the structure where an…
Q: 10 1/1 point Find K for the oxidation of ethanol to acetic acid at 25°C: CH3CH2OH (1) + O2(g) →…
A:
Q: A sample of argon gas occupies a volume of 6.57 L at 53.0°C and 0.880 atm. If it is desired to…
A: The objective of this question is to find the temperature of a sample of argon gas when its volume…
Q: A student conducts a chemistry experiment that is taking about two hours to complete. He decides to…
A: The question is asking about the effect of temperature on the rate of a chemical reaction. According…
Q: Chemistry
A: 4a. Given:…
Q: Precipitation Stoichiometry PL 1. When solutions of lead(II) nitrate and aluminum chloride are…
A: The objective of the question is to solve various problems related to precipitation stoichiometry.…
Q: Consider the following carbocation (i.e., a molecule with a positive formal charge on a carbon).…
A: Step 1: Step 2: Step 3: Step 4:
Q: This is another synthesis problem. Show reagents and intermediates synthesized along the way that…
A:
Q: Using this reaction energy diagram, determine the activation energy for the reverse reaction. 140…
A:
Q: 13
A:
Q: Draw the starting structure that would lead to the major product shown under the provided…
A:
Q: 15
A: So, option (b) is correct.
Q: What is the major organic product obtained from the following reaction? EtOH acid catalyst OH OEt OH…
A: If you have any queries, please comment.
Q: J9 cmsingi eltha noitulos eucoupe M 00 ebixonbyr Oxidation-Reduction Stoichiometry PL sin (1)ego to…
A: The objective of the question is to calculate the mass of copper produced, the excess reactant…
Q: 3. Propose syntheses of the following two targets beginning with the indicated starting material. Но…
A: Step 1: Step 2: Step 3: Step 4:
Q: O CH¸CH₂CH₂CH₂CH₂CH₂ČCI the struct + 2 NH3
A: Step 1:Nucleophilic attack by NH3 to the carbonyl carbon of the acid chloride. Step 2:-Cl is good…
Q: Please help me with the question below. A detailed explanation to aid in understanding is welcome.
A:
Q: Q2a: Design a titration experiment where 125 mL of a sodium hydroxide solution is titrated by 0.100…
A: The objective of this question is to design a titration experiment where a sodium hydroxide solution…
Q: Practice Problems 1. The concentration of H₂O* in a solution is found to be 1.75 x 108 M at 25°C.…
A: Given: [H3O+]=1.75x10−8M;[OH−]=???MStep 1: Write the formula we will use.pH=−log[H3O+]14.00=pH+pOH…
Q: 1 a). Find the concentration of hydroxide ion in 0.10 M NaClO 4 saturated with Mn(OH)2(Ksp = 1.6 x…
A: To find the concentration of hydroxide ions in a 0.10 M NaClO4 solution saturated with Mn(OH)2,…
Step by step
Solved in 2 steps with 1 images

- propose a synthesis for the following molecule starting from benzene. There is no need to show the mechanism, just show all reagents and conditions that need to be used on each stepprovide a 5-step synthesis to arrive at the final structure. Provide intermediate steps and show arrow pushing mechanism. Please use the following reagents: 1. Mg, Et2O 2. CO2 3. H3O+ 4. SOCl2 5. 2 eq of HN(Et)2Provide a synthesis for the following 3 reactions.
- Propose a synthesis for the following reaction. Dont show reactive intermediates or mechanisms, but show each isolable compound along the synthetic routeComplete the following multistep syntheses by showing the major products and the reagents nessesary for each stepShow the major organic product and mechanism for the transformations below.

