Identify the functional groups in each molecule. Classify each alcohol, alkyl halide, amide, and amine as 1°, 2°, or 3º. H. a. с. Но- е. ibuprofen (analgesic) Но penicillin G (an antibiotic) Darvon (analgesic) H2N. b. d. OH pregabalin trade name Lyrica (used in treating chronic pain) pyrethrin I (potent insecticide from chrysanthemums) histrionicotoxin (poison secreted by a
Identify the functional groups in each molecule. Classify each alcohol, alkyl halide, amide, and amine as 1°, 2°, or 3º. H. a. с. Но- е. ibuprofen (analgesic) Но penicillin G (an antibiotic) Darvon (analgesic) H2N. b. d. OH pregabalin trade name Lyrica (used in treating chronic pain) pyrethrin I (potent insecticide from chrysanthemums) histrionicotoxin (poison secreted by a
Chapter6: Organic Chemistry
Section: Chapter Questions
Problem 60E: Identify each compound according to its functional group:
Related questions
Question
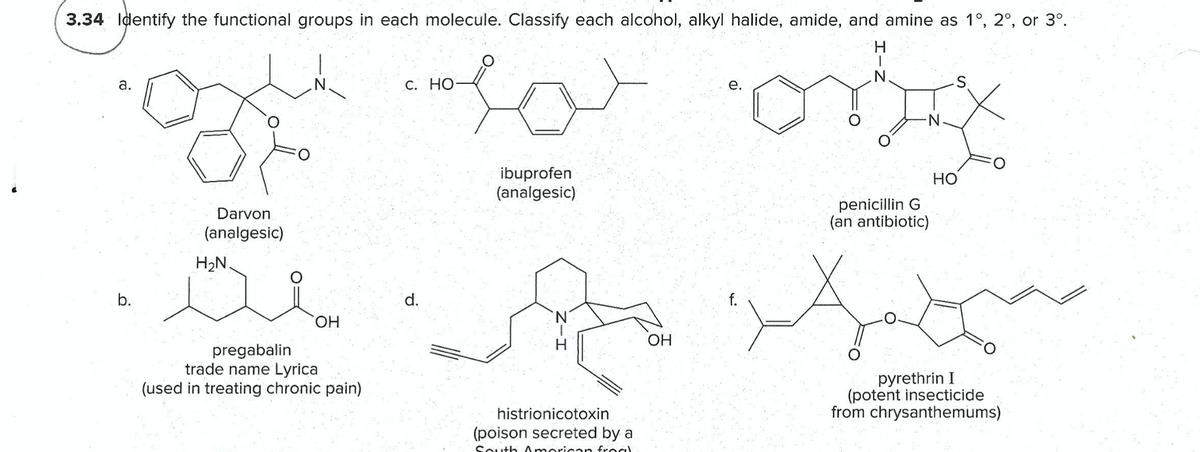
Transcribed Image Text:3.34 Identify the functional groups in each molecule. Classify each alcohol, alkyl halide, amide, and amine as 1°, 2°, or 3°.
H.
а.
С. НО
е.
ibuprofen
(analgesic)
Но
penicillin G
(an antibiotic)
Darvon
(analgesic)
H2N.
b.
d.
HO,
OH
pregabalin
trade name Lyrica
(used in treating chronic pain)
pyrethrin I
(potent insecticide
from chrysanthemums)
histrionicotoxin
(poison secreted by a
South American froc
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 6 steps with 4 images

Recommended textbooks for you

