Q: What products would you would expect to obtain when the following compounds react with ozone and…
A: Ozonolysis is an organic reaction where the unsaturated bonds in alkenes and alkynes compounds are…
Q: For each N-substituted benzene, predict whether the compound reacts faster than, slower than, or at…
A:
Q: 11. Provide the reagents required to achieve the follow transformations. Multiple steps may be…
A: The reactions given are,
Q: 11. Identify the reagent necessary to accomplish the following transformations:
A: Here, the first and second step involves the stepping up of carbon. This can be done by treating…
Q: ) State reagent
A: Benzene upon treated with nitration mixture to give nitrobenzene by electrophilic substitution…
Q: 3. The following reaction is used in the industrial synthesis of the antiviral drug, oseltamivir…
A: This problem is based on deprotactation and protection of diols by using different type of…
Q: 9. Predict the products obtained when benzoquinone is treated with excess butadiene:of the following…
A: The final product of the above reaction is
Q: Which of the ethers shown could be prepared in good yield by an acid-catalyzed dehydration of a…
A: Alcohols are dehydrated in presence of an acid catalyst. If we use the mixture of two different…
Q: Benzene undergoes electrophilic aromatic substitution reactions with aziridines in the presence of a…
A: (a) Benzene undergoes electrophilic aromatic substitution reactions with aziridines in the presence…
Q: 9. Predict the products obtained when benzoquinone is treated with excess butadiene:of the following…
A: The given reactant is Benzoquinone which reacts with excess butadiene.
Q: 3. The following compound has been synthesised in the lab. (a) Give two synthesis methods for the…
A: We are required to show the synthesis for the ether by 2 method and also state which pathway is more…
Q: Which set of starting materials could be used to prepare the following compound in one step? * Meo-…
A: Given product is a diels-alder product . In diels -alder reaction conjugated diene reacts with a…
Q: 7. Provide the major organic product of the following reaction sequence. 1. Mg 2. CO2 3. H,0*, A -Br
A: Grignard Reaction is an organometallic reaction in which different alkyl, aryl-magnesium halides add…
Q: 2.11. What is the best description of reactions to carry out that will arrive at the following…
A: Synthesis of aldehyde using the right steps.
Q: 8. The preparation of the sex pheromone of the boll worm moth, (E)-9,11-dodecadien-1-yp acetate from…
A: MnO2 oxidizes allylic primary alcohol to aldehyde Reaction of aldehude with Wittig reagent gives…
Q: 1. What set of reagents is necessary for the synthesis of the following compound: A A В C D D CN CN…
A: Diels-Alder reaction: The Diels–Alder reaction is a pericyclic reaction (cycloaddition) between a…
Q: 7. Compound X is an alcohol. Upon oxidation by acidify potassium permanganate, a ketone, Y is…
A: Since, you have asked multiple question, we will solve the first question for you. If you want any…
Q: 3. For each of the following, state whether it would undergo nitration faster, slower or at the same…
A:
Q: 7. Give the stereochemistry for the product of each step of the conversion of trans-4-t-…
A: The step by step transformations are given below -
Q: 7.62 Below are two potential methods for preparing the same ether, but only one of them is…
A:
Q: After ozonolysis and treatment of the unstable ozonide with CH3SCH3, compound A was converted to the…
A:
Q: Identify the major product obtained when this compound is treated with fuming sulfuric acid. SO3…
A: Ans : Options : (2) IV Right. Explain : Benzaldehyde React with fuming conc. H₂SO₄ to…
Q: For each compound below, propose an efficient synthesis using diethyl malonate as a starting…
A: (Note- Since it is a multiple questions , hence I am solving first question) Here we have to…
Q: 11) Give an arrow-pushing mechanism for the following reactions: a. H3O*
A: The answer is given as follows
Q: What alkyl halide reacts with lithium divinylcuprate 3(CH2“CH)2CuLi4 for the synthesis of each of…
A: The molecular formula of lithium divinylcuprate = C4H6CuLi (a) When bromocyclohexane is treated with…
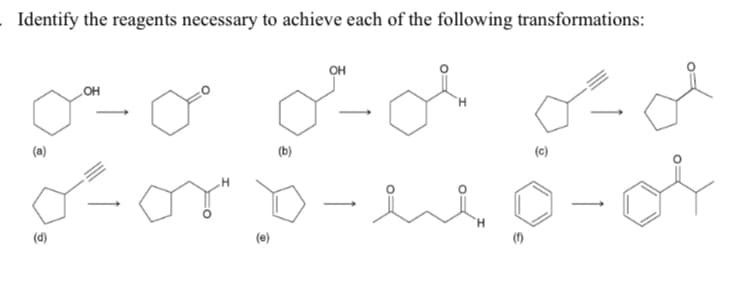
Q: . Identify the reagents you would use to achieve each of the following transformations: (a) (b)
A: In question a terminal alkyne converts to ketone and in question b terminal alkyne converts to…
Q: 3. Consider the following retrosynthetic analysis of Disparlure, the sex pheromone of the female…
A:
Q: Compound A yields the following two products upon ozonalysis. Which of the following is the correct…
A: Solution: We know alkene on ozonolysis gives carbonyl compounds by cleaving alkene C=C bond &…
Q: 3. What is of the following compounds will produce a mixture of constitutional isomers when treated…
A: Unsymmetrically disubstituted alkene/alkyne upon reaction with HBr give a mixture of constitutional…
Q: ite the structures of A-C and mechanisms of the following reactions. (i) Me2CuLi (i) PhMgBr N' HCI H…
A: In the above reaction, first step is formation of enamine Second step is reaction of Gilman Third…
Q: Identify the reagents necessary to achieve each of the following transformations: он он (a) (b) (c)
A:
Q: 3. (a) Strategies the synthesis of the given transformations to ensure no side-product (provide the…
A:
Q: Identify the major product that is expected when nitrobenzene is treated with Brzin the presence of…
A:
Q: 3. Consider the following retrosynthetic analysis of Disparlure, the sex pheromone of the female…
A: From given For the retrosynthesis of Disparlure given Forward synthesis is drawn below accordingly
Q: b) What starting materials are needed to prepare the compound below by intramolecular Mic reaction?…
A: The answer is given as follows
Q: Select the expected product of the following reaction. Br NaCN DMSO
A: SN2 attack In RDS step two molecules participated Walden inversion product CN- ion approach to…
Q: b) OH Hemiacetal H3C D Compound D could be converted into a cyclic hemiacetal in the presence of an…
A: Aldehydes on treatment with an alcohol in the presence of an acid produces hemiacetal.
Q: (c). Blattellaquinone is the sex pheromone of the female German cockcroach. It is released by…
A:
Q: Benzene undergoes electrophilic aromatic substitution reactions with aziridines in the presence of a…
A: (a) Benzene undergoes electrophilic aromatic substitution reactions with aziridines in the presence…
Q: A OH B D
A: Here molecular formula is given so first of all we have to find the DBE i.e double bond equivalency…
Q: 3. (a) Strategies the synthesis of the given transformations to ensure no side-product (provide the…
A:
Q: Identify the reagent needed to accomplish the transformation below (enter the appropriate code from…
A: The answer is given as follows
Q: 12. Starting from any alkyne of your choice, suggest a method to synthesis the following products:…
A: Hydration of alkyne: Alkyne gives hydration reactions in presence of an H2SO4 and HgSO4 catalyst. In…
Q: 1) NANH2 2) benzyl chloride/acetone SH
A: When thiol react with a base, it abstracts the proton and then thiol becomes a very good…
Q: The last step in the synthesis of β-vetivone, a major constituent of vetiver, a perennial grass…
A: Vetivone is an organic compound which is derived from three isoprene units and belongs to the class…
Q: Which of the following provides the best description of the two products of the following ether…
A: Given ether is isopropyl propyl ether.
Q: 18.28 Starting with benzene and using any other necessary reagents of your choice, design a…
A: Requirement from question Four organic compounds and their synthetic route.
Q: 7. Compound X is an alcohol. Upon oxidation by acidify potassium permanganate, a ketone, Y is…
A:
Q: Describe how the following compounds can be prepared from compoundscontaining no more than six…
A: EXPLANATION: These compounds are prepared by the compound containing not more than six carbons.To…
Q: Which of these molecules will cyclize to give the insect pheromone frontalin? OH Но HO Frontalin (A)…
A:
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- 7. Roadmap. Complete the following roadmap synthesis by providing all the missing reagents. If more than one step is required, be sure to indicate so with sequential steps. No sequence should be more than 3 steps.16. When cis-1,2-cyclopentanediol reacts with acetone in dry HCl, compound M is formed. What do you expect the hydrolysis of compound M to be when subjected (i) to base; and, (ii) acid A. resistant to base; resistant to acid B. resistant to base; readily hydrolyzed by acid C. readily hydrolyzed by base; resistant to acid D. readily hydrolyzed by base; readily hydorlyzed by acidIdentify the reagents you would use to achieve of the following transformation. Identify these reactions as SN1 or Sn2. (a) (b) (c) (d)
- 1a). Predict the major product obtained in the following reaction: B). Write out the 3-step arrow pushing mechanism leading up this product.16. When cis-1,2-cyclopentanediol reacts with acetone in dry HCl, compound M is formed. What do you expect the hydrolysis of compound M to be when subjected (i) to base; and, (ii) acid a. resistant to base; resistant to acid b. resistant to base; readily hydrolyzed by acid c. readily hydrolyzed by base; resistant to acid d. readily hydrolyzed by base; readily hydorlyzed by acid 17. Following (16), would you expect that a similar compound be formed if the reactant was trans-1,2-cyclopentanediol? Why is this so? a. Yes; hydroxyl groups are on opposite side b. No; hydroxyl groups are on opposite site c. Yes; hydroxyl groups are on the same side d. No; hydroxyl groups are on the same sidePredict the expected product for each reaction and provide IUPAC name for the correct starting material to yield the desired epoxide: 1. Name of compound 2. Answer A, B, C, or D
- Treatment of W with CH3Li, followed by CH3I, affords compound Y (C7H14O) as the major product. Y shows a strong absorption in its IR spectrum at 1713 cm−1, and its 1H NMR spectrum is given below, (a) Propose a structure for Y. (b) Draw a stepwise mechanism for the conversion of W to Y.Explain briefly by illustrations the chemistry behind each answer 1. Preamble :A reaction flask contains a 2-bromopentane in an ethanolic solution of sodium ethoxide at room temperature and results in the formation of two olefinic products(1-pentene and 2-pentene) I) What reaction pathway is followed by the reaction above?A. E2 dehydrohalogenation B. E1 dehydrohalogenationC. SN1 dehydrohalogenationD. SN2 dehydrohalogenationE. A mixture of E1 and E2 pathways II) What is responsible for the formation of different products (major and minor).A. The different activated complex involved in the mechanism.B.Bimolecular Nucleophilic substitution reaction C.Bimolecular Elimination reaction D.The presence of sodium ethoxideE.The hybridisation nature of the secondary carbocation III) All the following is true about the reaction in question 1 except?A. The reaction follows Zaistev’s ruleB. Sodium ethoxide is the nucleophile in this reactionC. The more highly substituted alkene is the most stable…8.b)Provide mechanisms for the following reactions:
- Predict the most stable product for the following reaction sequence: 1. A 2. B 3. C 4. D 5. EPredict the product C of the following reaction below and briefly explain youranswer.[PtCl3(C2H4)]¯ + NH3 → C1. What effect would you expect on the rate of reaction of ethanol with 2-iodo-2-methyl-butane if the concentration of the halide is tripled? 2. Explain the difference between addition, elimination and substitutionreactions. 3. Optically active 2-butanol slowly racemizes on standing in dilute sulfuricacid. Propose a mechanism to account for this observation.



