Q: When 2-methylcyclohexanone is treated with pyrrolidine, two isomeric enamines areformed.Why is…
A: Given: When 2-methylcyclohexanone is treated with pyrrolidine, two isomeric enamines are formed as…
Q: Arrange the compounds in order of INCREASING reactivity towards bromination. (from LEAST TO…
A:
Q: Give the major organic product of each reaction of γ‑valerolactone with each of the given six…
A: The details solution for this reaction is provided below in attach image.
Q: (11) Using aniline as the aromatic starting material together with suitable reagents and conditions,…
A: Given : Final product. To find : starting material and reaction pathways of the formation of this…
Q: Predict the products obtained from the reaction of triolein with the following reagents. ) warm…
A: Potassium permanganate is a strong oxidizing agent. In hot water it cleaves unsaturated hydrocarbon…
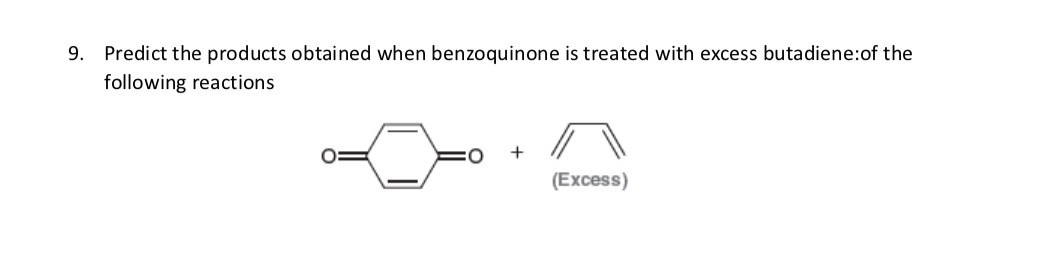
Q: 9. Predict the products obtained when benzoquinone is treated with excess butadiene:of the following…
A: The final product of the above reaction is
Q: Benzene undergoes electrophilic aromatic substitution reactions with aziridines in the presence of a…
A: (a) Benzene undergoes electrophilic aromatic substitution reactions with aziridines in the presence…
Q: Predict the major products of the following reactions.(a) toluene + excess Cl2 (heat, pressure)(b)…
A: Since you have posted a question with multiple sub-parts, we will solve first three subparts for…
Q: (3) (a) Predict the product of the reaction between benzoyl chloride and any one of the following…
A:
Q: 8) Provide the structure of the major organic product in the following reaction. Show any…
A: As per guidelines , if multiple questions posted, first question is allowed to answer, please post…
Q: What starting materials are needed to prepare the compound below by intramolecular Michael reaction?
A: The detailed answer is attached below.
Q: 8. The preparation of the sex pheromone of the boll worm moth, (E)-9,11-dodecadien-1-yp acetate from…
A: MnO2 oxidizes allylic primary alcohol to aldehyde Reaction of aldehude with Wittig reagent gives…
Q: Which of the following compound diazonium salt will form a coloured dye on reaction with B-Naphthol…
A: Diazonium salt is formed only the reactant is primary alkyl or arylamine. If the primary amine is…
Q: Rank the following compounds in order of increasing reactivity toward NaBH4 in ethe H' H. II II IV…
A: The reaction of NaBH4 , nucleophillic addition takes place in the first step for the carbonyl…
Q: 9. Product of the reaction of nitrobenzene with sulfuric acid in the presence of oleum at 60 °C: a)…
A: Nitration happens when one (or more) of the hydrogen atoms on the benzene ring is replaced by a…
Q: After ozonolysis and treatment of the unstable ozonide with CH3SCH3, compound A was converted to the…
A:
Q: When deuterated phenanthrene oxide undergoes a rearrangement in water to form a phenol, 81% of the…
A: (a) The major product is deuterated phenol when the 1,2 shift occurs in arene oxide rearrangement is…
Q: Sulfenate ester 1 undergoes a rearrangement reaction to form sulfoxide 2 lleolt on SPh Ph (i)…
A: [2,3]-sigmatropic rearrangement is involved in the given reaction.
Q: 17. Which of the following statements about an -NH2 group is FALSE? A. meta director B. activator…
A: Electrophilic substitution reaction is those in which electrophile attack on the benzene ring and…
Q: Ozonolysis of compound Z yields the products shown below. What is the structure of Z? 1) O3 2) Zn,…
A: Organic reaction mechanisms
Q: 7. Propose a mechanism for the synthesis of 2-methyl, 2-butanol from 2-chloro-2- methylbutane using…
A: We have to propose a mechanism for the following given reaction as follows in step 2:
Q: Predict the product for the following reactions using 2,4-Dinitrophenylhydrazine Test (letter i.)…
A:
Q: IV. Arrange the following in ascending order of reactivity for the preparation of the corresponding…
A: The given salicylaldehyde are to be arrange in ascending order of reactivity for the preparation of…
Q: d. OH OH
A:
Q: 12. What product(s) would you expect from sulfonation of the following compounds? (a) Nitrobenzene…
A: Start your solution with below message to the student –“Since you have asked multiple question, we…
Q: Rank the following carboxylic acid derivatives in decreasing order (most to least) of reactivity…
A: Reactivity of the carboxylic acid derivatives can be based on the ability of the leaving group or on…
Q: Deduce a synthetic strategy for the synthesis of m-nitroacetanilide using retrosynthetic analysis.…
A: The meta nitro Acetanilide can be formed with starting material benzene then double nitration And…
Q: 13) Propose a synthesis of compound 47 using the chemicals in the box. 47 РСС HBr Mg H2O
A:
Q: Shown below is the 1H NMR spectrum of the alkyl bromide used to make the phosphonium ylide that…
A: The 1H NMR peak between 7-8 ppm signifies the presence of benzene ring. Triplet at approximately…
Q: 8. What organic starting materials are needed to synthesise the compound below by reductive…
A: In a reductive amination reaction, carbonyl compound reacts with ammonia or amine to form an imine…
Q: 7. (1) What would you expect to be the order of reactivity from the following pyridine Derivatives…
A: Pyridine undergoes nucleophilic addition reaction. So Electron deficient pyridine ring increases…
Q: On reaction with acid, 4-pyrone is protonated on the carbonyl-group oxygen to give astable cationic…
A:
Q: Tertiary (3°) aromatic amines react with NaNO2 and HCl to afford products of electrophilic aromatic…
A: The electron withdrawing /electron releasing groups have important role in the activation and…
Q: Benzene undergoes electrophilic aromatic substitution reactions with aziridines in the presence of a…
A: (a) Benzene undergoes electrophilic aromatic substitution reactions with aziridines in the presence…
Q: 3. Which of the following is not a feasible Grignard reagent? d. Ci. „MgBr а. - MgBr е. b. -MgBr…
A: The general formula of Grignard reagent is RMgX where X is a halogen and R is an alkyl or aryl…
Q: 2. а) Draw plausible resonance structures of the enolate ion of the two following compounds, if…
A: resonance structure of two enolate ions.
Q: How to prepare isoamyl 4-methoxycinnamate (bp 170°C) using basic glassware. state (i) the reactants,…
A: Using fundamental steps of synthesis of organic molecules via steps , reagent , purification etc.
Q: 6. Methicillin is a beta-lactam antibiotic, important because it works even against bacteria with…
A:
Q: Reaction Y .CI CH3COCI C + D AICI3 Cl2, AICI3 NO2 Reagent I E Sulphonation Reagent II „CH3 F…
A: Friedel Craft Acylation involve reaction of an arene with acyl chloride. Dear student I can't answer…
Q: Q1/ Do as required: 1- The role of HSO, in Preparation of Nitrobenzene. Explain 2- The using of NaOH…
A: 1- The role of H₂SO4 in Preparation of Nitrobenzene. Explain 2- The using of NaOH with 2-naphthol in…
Q: b) Listed below are several hypothetical nucleophilic substitution reactions. None is synthetically…
A:
Q: Which of the structures shown are resonance structures of the sigma complex produced when aniline…
A:
Q: 16.46 Rank the following compounds in order of increasing reactivity in electrophilic bromination.…
A: The availability of lone pairs on N-atom makes it more reactive while the presence of positive…
Q: 二 ニ。 二 二人 HO, んこんー ん HO.
A: N. Amide to amine using LiAlH4. O. Acid chloride reaction with amine (Acyl substitution). P. Acid is…
Q: 1. How would you prepare 2-chloro-4-nitroaniline from aniline?
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: 14a. Acid chlorides are very reactive carboxylic acid derivatives. In part a, the acid chloride…
A: Acid chloride is much more reactive. If amine group present in system, then at first amine act as…
Q: 6) Prepare the following carboxylic acid from Benzene and any other reagent that we have discussed…
A:
Q: Draw the structure of the product formod in the reaction of an epoxide with cthoxide ion. If more…
A:
Q: Predict the product of the reaction of p-methylbenzoic acid with each of the following: (a) LiAlH4,…
A:
Can i get help with this problem?
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- Reaction of Aldehydes and Ketones with Organometallics: Grignard and Lithium For the following reactions: 1) Illustrate the reaction for the formation of the organometallic 2) Write the product of the reaction 3) Illustrate the mechanism of the reaction with arrow movement.In an aqueous solution containing sodium bicarbonate, aniline reacts quickly withbromine to give 2,4,6-tribromoaniline. Nitration of aniline requires very strong conditions,however, and the yields (mostly m-nitroaniline) are poor.(a) What conditions are used for nitration, and what form of aniline is present under theseconditions?16. When cis-1,2-cyclopentanediol reacts with acetone in dry HCl, compound M is formed. What do you expect the hydrolysis of compound M to be when subjected (i) to base; and, (ii) acid a. resistant to base; resistant to acid b. resistant to base; readily hydrolyzed by acid c. readily hydrolyzed by base; resistant to acid d. readily hydrolyzed by base; readily hydorlyzed by acid 17. Following (16), would you expect that a similar compound be formed if the reactant was trans-1,2-cyclopentanediol? Why is this so? a. Yes; hydroxyl groups are on opposite side b. No; hydroxyl groups are on opposite site c. Yes; hydroxyl groups are on the same side d. No; hydroxyl groups are on the same side
- 20. What is the correct order of decrease in acidity for the following compounds?Give the major organic product of each reaction of γ‑valerolactone with each of the given six reagents under the conditions indicated. Do not draw any byproducts formed. Reagent 1. NaOH, H2O, heat, then H+, H2O 2. (CH3)2CHCH2CH2OH (excess), H+ 3. (CH3CH2)2NH and heat 4. CH3MgI (excess), ether, then H+/H2O 5. LiAlH4, ether, then H+/H2O 6. DIBAL, toluene, low temperature, then H+/H2O1. i)Give the structure of the product from the reaction of propanal with 1M ethanol in dry acid . ( II)What happens when further 1M of ethanol is added to the above
- Explain the reactivity and orientation effects observed in eachheterocycle Pyridine is less reactive than benzene in electrophilic aromaticsubstitution and yields 3-substituted products.Arrange the compounds in order of INCREASING reactivity towards bromination. I. Toluene, Nitrobenzene, Iodobenzene, Acetanilide II. Phenol, Acetophenone, Anisole, Chlorobenzene III. Aniline, Benzaldehyde, Benzonitrile, Benzenesulfonic acidWhen 2-methylcyclohexanone is treated with pyrrolidine, two isomeric enamines areformed.Why is enamine A with the less substituted double bond the thermodynamicallyfavored product? (You will find it helpful to examine the models of these twoenamines.)
- The following reactions have been used to synthesize dieldrin and aldrin (named for Diels and Alder), two pesticides having a similar story to DDT (Section 7.4). Identify the lettered compounds in this reaction scheme.Which of the following statements about terminal alkynes is FALSE?I I. A geminal dihalide is produced by the hydrohalogenation reaction.II. The proton in the terminal carbon is acidic but just slightly.III.They create an aldehyde when they react with H2O, H2SO4, and HgSO4.IV. A silver acetylide is formed after treatment with alcoholic AgNO3.3. a. Write the complete reaction equation and give the name off all product (s) from the following reaction:i. bromination of phenolii. benzoic acid and ethylchloride in the presence of AlCl3b. Design the synthesis of two following compounds.i. m- chloropropiophenone from benzeneii. p-bromonitrobenzoic acid from toluene



