Identifying titration curves Vhat to notice when making the identification a Use the trends identified in A1a-f ) Does the pH decrease (increase) with added titratant? Then, the solution is tritrated with HCI (NAOH). (u) Is there a large pH change at the start of the titration? Then, the solution is distilled water. (u1) Does the solution resist pH change with acid or base until about 9-10 ml of titrant is added? Then, the solution is buffer no 1 (iv) Does the solution resist pH change with acid until about 12-13 ml of titrant is added? Then, the solution is buffer no 2 (v) Does the solution resist pH change with base until abut 4-5 ml of titrant is added? Then, the solution is buffer no 2 Titration Curve Identification O distilled water titrated with HCI 13.0 11.0 O buffer no. 1 bitrated with HCI 9.0 O buffer no. 2 titrated with HCI 좀 70 5.0 O distilled water titrated with NaOH 3.0 O buffer no. 1 btrated with NaOH 10 10 15 O buffer no 2 bitrated with NaOH Titrant Volume/ml distilled water titrated with HCI 13.0 11.0 O buffer no 1 titrated with HCI 9.0 O buffer no. 2 titrated with HCI 종 7,0 O distilled water titrated with NaOH 5.0 3.0 O buffer no. 1 btrated with NaOH 1.0 10 15 5n Titant Volume/ml O buffer no 2 titrated with NaOH O distilled water titrated with HCI 13.00 11.00 O buffer no. 1 titrated with HCI 9.00 O buffer no 2 titrated with HCI 종 7,00
Identifying titration curves Vhat to notice when making the identification a Use the trends identified in A1a-f ) Does the pH decrease (increase) with added titratant? Then, the solution is tritrated with HCI (NAOH). (u) Is there a large pH change at the start of the titration? Then, the solution is distilled water. (u1) Does the solution resist pH change with acid or base until about 9-10 ml of titrant is added? Then, the solution is buffer no 1 (iv) Does the solution resist pH change with acid until about 12-13 ml of titrant is added? Then, the solution is buffer no 2 (v) Does the solution resist pH change with base until abut 4-5 ml of titrant is added? Then, the solution is buffer no 2 Titration Curve Identification O distilled water titrated with HCI 13.0 11.0 O buffer no. 1 bitrated with HCI 9.0 O buffer no. 2 titrated with HCI 좀 70 5.0 O distilled water titrated with NaOH 3.0 O buffer no. 1 btrated with NaOH 10 10 15 O buffer no 2 bitrated with NaOH Titrant Volume/ml distilled water titrated with HCI 13.0 11.0 O buffer no 1 titrated with HCI 9.0 O buffer no. 2 titrated with HCI 종 7,0 O distilled water titrated with NaOH 5.0 3.0 O buffer no. 1 btrated with NaOH 1.0 10 15 5n Titant Volume/ml O buffer no 2 titrated with NaOH O distilled water titrated with HCI 13.00 11.00 O buffer no. 1 titrated with HCI 9.00 O buffer no 2 titrated with HCI 종 7,00
Chapter17: Complexation And Precipitation Reactions And Titrations
Section: Chapter Questions
Problem 17.35QAP
Related questions
Question
3
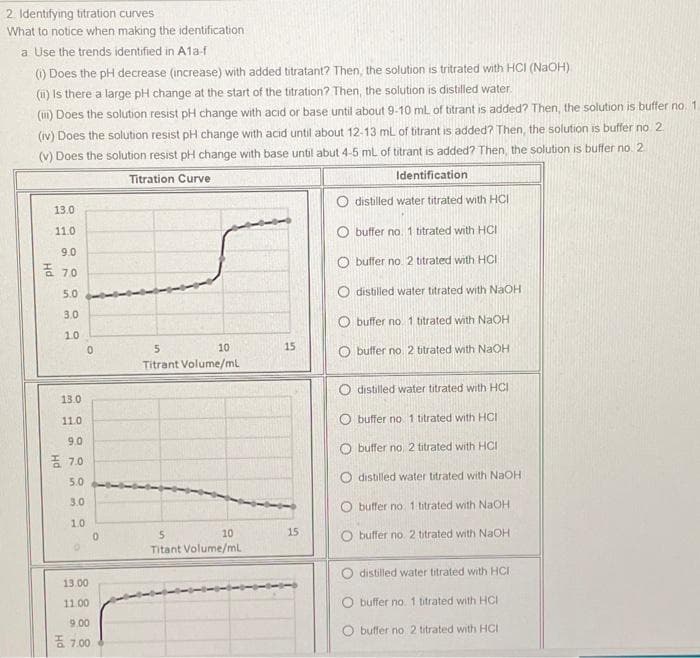
Transcribed Image Text:2. Identifying titration curves
What to notice when making the identification
a. Use the trends identified in A1a-f
() Does the pH decrease (increase) with added titratant? Then, the solution is tritrated with HCI (NaOH)
(1) Is there a large pH change at the start of the titration? Then, the solution is distilled water.
(m) Does the solution resist pH change with acid or base until about 9-10 mL of titrant is added? Then, the solution is buffer no. 1.
(iv) Does the solution resist pH change with acid until about 12-13 ml of titrant is added? Then, the solution is buffer no 2
(v) Does the solution resist pH change with base until abut 4-5 mL of titrant is added? Then, the solution is buffer no. 2
Titration Curve
Identification
O distilled water titrated with HCI
13.0
11.0
O buffer na. 1 titrated with HCI
9.0
O buffer no. 2 titrated with HCI
E 70
5.0
O distilled water titrated with NaOH
3.0
O buffer no. 1 titrated with NaOH
1.0
10
15
O buffer no 2 titrated with NaOH
Titrant Volume/ml
O distilled water titrated with HCI
13.0
11.0
O buffer no. 1 titrated with HCI
9.0
O buffer no 2 titrated with HCI
E 7.0
5.0
O distilled water titrated with NaOH
3.0
O buffer no. 1 btrated with NaOH
1.0
10
15
O buffer no. 2 titrated with NAOH
Titant Volume/mL
distilled water titrated with HCI
13.00
11.00
O buffer no. 1 titrated with HCI
9.00
O buffer no 2 titrated with HCI
5 7.00
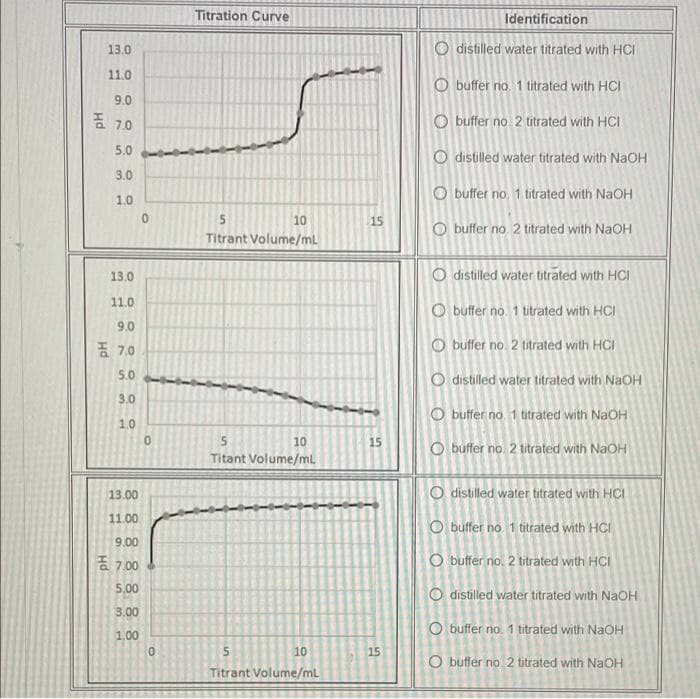
Transcribed Image Text:Titration Curve
Identification
13.0
O distilled water titrated with HCI
11.0
O buffer no. 1 titrated with HCI
9.0
증 7,0
O buffer no. 2 titrated with HCI
5.0
O distilled water titrated with NaOH
3.0
O buffer no 1 titrated with NaOH
1.0
10
15
buffer no. 2 titrated with NaOH
Titrant Volume/mL
13.0
O distilled water titrated with HCI
11.0
O buffer no 1 titrated with HCI
9.0
품 7,0
O buffer no. 2 titrated with HCI
5.0
distilled water titrated with NaOH
3.0
O buffer no 1 titrated with NaOH
1.0
10
15
buffer no. 2 titrated with NaOH
Titant Volume/ml
13.00
O distilled water titrated with HCI
11.00
O buffer no 1 titrated with HCI
9.00
है 7.00
O buffer no. 2 titrated with HCI
5,00
O distilled water titrated with NaOH
3.00
O buffer no. 1 titrated with NaOH
1.00
10
15
O buffer no. 2 titrated with NAOH
Titrant Volume/mL
in
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

