If the substrate for an enzyme catalyzed reaction contained a negative charge, which of the following amino acids would most likely be present in the active site to provide electrostatic destabilization of the ES complex? Leu O val O ASP O LyS
Q: running the reaction at 83 °C cooling the reaction to 11 °C changing the pH to 5.4 Increase reaction…
A: The rate of an enzyme catalyzed reaction depends on various factors like temperature and pH of the…
Q: Which of the following equations accurately shows enzyme-catalyzed conversion of substrate to…
A: Enzymes are protein molecules that increase the rate of reaction by decreasing the activation…
Q: In First order, the rate of reaction would be . ., if the concertation of an enzyme is increased by…
A: The reaction may be classified according to the order of reaction, which is the number of reacting…
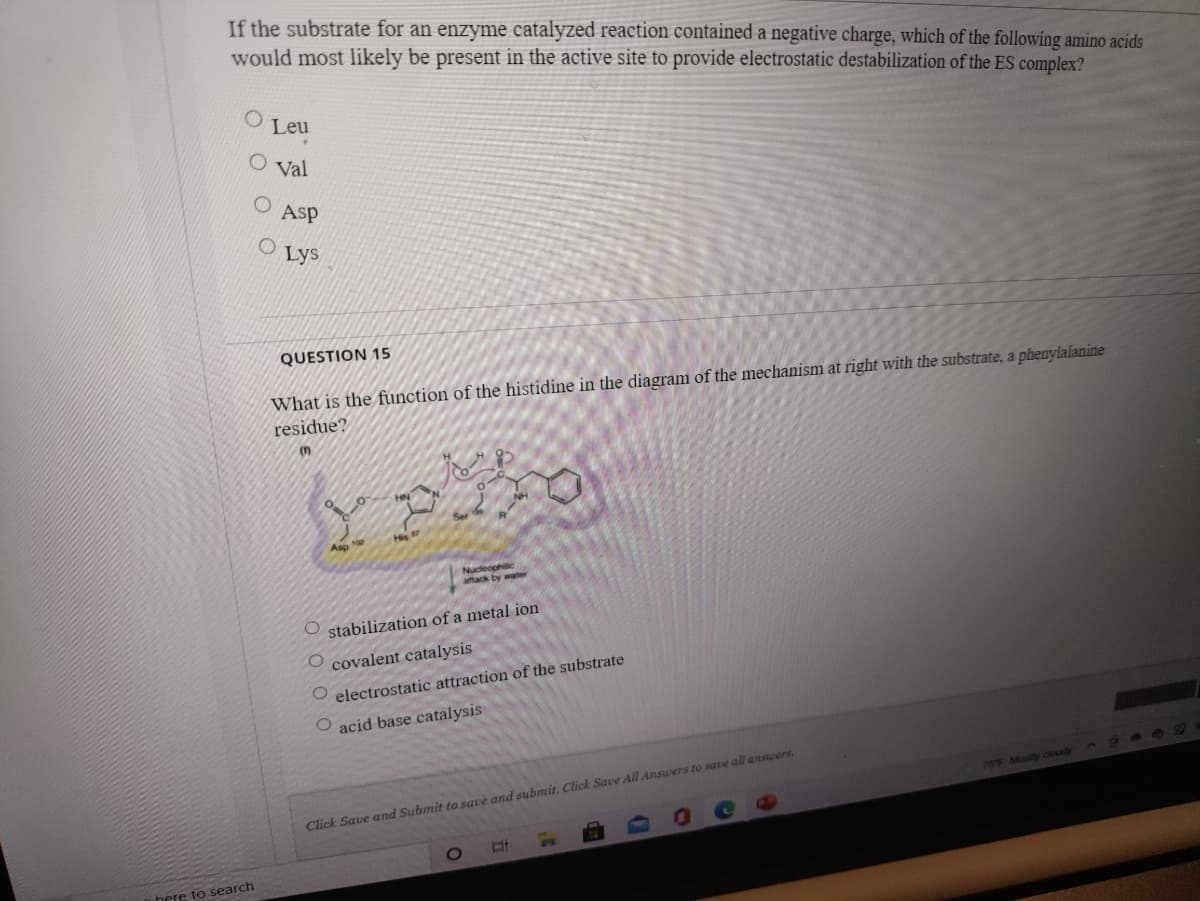
Q: Which of the following is incorrect about the enzyme chymotrypsin? Oa. It catalyzes the addition of…
A: Chymotrypsin is a digestive enzyme component of pancreatic juice. The enzyme catalyzes the cleavage…
Q: According to the graph, which of the following is TRUE? O AMP acts as a substrate for catalysis. AMP…
A: Glucose is catabolized to form two molecules of pyruvate via glycolysis. Glycolysis is a ten-step…
Q: Which of the following is incorrect about the enzyme chymotrypsin? a. It catalyzes the addition of…
A: Chymotrypsin is a serine protease that catalyses the cleavage of peptides on the Carboxy side of the…
Q: The enzyme which catalyzes the reaction below belongs to which enzyme classification? C-0- HC=0 +…
A: Enzyme is basically biocatalyst that increase the rate of chemical reaction without itself being…
Q: OH || CH3CH=CHCSR CH3CHCH,CSR
A:
Q: Allosteric regulation of an enzyme is accomplished when the enzyme pathway's product accumulates and…
A: Enzymes are proteins. These act as catalysts and regulate the rate of reaction. During the…
Q: Which of the following is incorrect about chymotrypsin? a. It uses acid-base catalysis Ob. It has…
A: Chymotrypsin is a protease that cleaves the carbonyl side of certain peptide bonds. The protease…
Q: Given the following enzyme catalyzed reaction, identify the class and subclass of the enzyme…
A: According to reaction they catalyse the enzymes have been categorised in 6 classes: Oxidoreductases,…
Q: Enzyme cofactors are usually inorganic ions that enhanceenzymatic activity by improving the “fit”…
A: True.
Q: An enzyme active site contains a lysine that acts as through electrostatic catalysis (proximity and…
A: Hi! Thank you for the question. As you have posted a question with multiple subparts, I will be…
Q: The active (catalytic) site of an enzyme contains the side chains of amino acid residues that…
A: The active site of the enzyme is responsible for making a small but significant change on another…
Q: is when one substrate molecule primes an enzyme to act on additional substrate molecules more…
A: Enzymes are proteins that act as biological catalysts. Catalysts accelerate chemical reactions. The…
Q: WHich arrow represents the ΔG for the catalyzed reaction and which arrow represnts the activation…
A: We have to understand what catalyzed and uncatalyzed reaction means. When we go to the office we…
Q: A type Il beta turn has what residue as one of the four residues that make up the turn? OAP O B. G O…
A: Ramachandran structure describes a protein all possible conformations.
Q: An example of an enzyme-catalyzed reaction proceeding via a transition-state stabilization mechanism…
A: Chymotrypsin is synthesized by the pancreas as inactive chymotrypsinogen. Chymotrypsin is a…
Q: The mechanism by which trypsin, chymotrypsin, and elastase catalyzes proteolysis is: O Acid-base…
A: Trypsin, chymotrypsin, and elastase are examples of serine poteases. Proteases specifically break…
Q: What would be the result of an enzyme having a greater binding energy for the substrate than for the…
A: Enzymes are proteins that work as biological catalysts. Enzymes act on the molecules called…
Q: Which of the following statements describes an enzyme ALLOSTERIC site? O It is where an inhibitor…
A: Allosteric site is the site that allows enzyme activity to be activated or inhibited by molecules.
Q: A compound was suspected of being a negative allosteric regulator of enzyme A. Which of the…
A: Allosteric regulation is the regulation of an enzyme in the body by binding an effector molecule at…
Q: Which of the following statements regarding enzyme catalysis is false? All options are false. Once…
A: Normally an energy barrier exists between substrates (S) and products (P) i.e., S -> P…
Q: Consider the following reaction. CH₂-CH-COO-CH₂-C-Coo- он b Which group of enzymes catalyzes this…
A: The six functional classes of enzymes are hydrolases, oxidoreductases, lyases, transferases, ligases…
Q: Which of the following statements is TRUE in describing the activity of the lactase enzyme? A.…
A: Lactase enzyme is produced by our body to help in breaking down the lactose. It is produced by cells…
Q: What general kinds of reactions do the following types of enzymes catalyze?(a) Kinases (b)…
A: An enzyme accelerates the rate of a chemical reaction. The formation of enzyme-substrate complex is…
Q: Protein biosynthesis is best described as an example of: a) Catabolism b) Anabolism c)…
A: The term "metabolism" is used to define the collective of all chemical reactions and pathways that…
Q: The following is the chemical structure of nicotinamide adenine dinucleotides, a coenzyme commonly…
A: Coenzymes are the complex organic molecules which pump up activity of enzyme and function as…
Q: From the enzymatic data below, estimate he Vmax and Km for the enzyme presenting these dat Vo nM/s…
A: Introduction Maximal Velocity (Vmax): Increasing the substrate concentration indefinitely doesn't…
Q: Which type of reaction is catalyzed by the coenzyme nicotinamide adenine dinucleotide? O Hydride ion…
A: Introduction: The non-protein molecule, that is organic and has a low molecular weight associated…
Q: Given the following reaction, identify the class of the enzyme involved. CO (CH;2 ATP + NE H- NH,…
A: The international commission on enzymes has classified the enzymes based on the type of reactions…
Q: || CH3CSCOA "02CCH2CSCOA
A:
Q: An enzyme catalyzes the reaction MN. The enzyme is present at a concentration of 0.0000000022 M, and…
A: Enzyme catalyzed reaction involves the binding of enzyme to substrate leading to the formation of…
Q: Which of the following is NOT TRUE about allostery? O The enzyme changes conformation upon binding…
A: When a chemical reaction occurs, a catalyst is a substance that accelerates the process without…
Q: An enzyme that catalyzes a reaction such as the one below is alan: || C-0 || C-0 C=0 C-0-H || H- C-H…
A: Enzymes are catalysts that perform chemical reactions at an exceptionally high rate under the mild…
Q: Given the following enzyme catalyzed reaction, identify the class and subclass of the enzyme…
A: This is the process of conversion of pyruvate to lactate. The reduction of the pyruvate to lactate…
Q: The following reaction would most likely be catalyzed by an enzyme of which class? sucrose + H2O →…
A: Enzymes are substances which act as catalyst within a biological system to catalyze various…
Q: The inhibition that make permanent damage to an enzyme is called: O Noncompetitive O Competitive O…
A: The enzymes are biocatalysts. These work as biochemical catalysts and catalyze different reactions…
Q: Which of the listed effects would be brought about by any enzyme catalyzing the following simple…
A: Introduction: Those compound that increases the rate of the reaction without undergoing any change…
Q: OH || CH3CHCH2CSR || CH3CH=CHCSR
A: Enzymes are the biomolecules majorly comprise of protein chains. The purpose of the enzymes is to…
Q: Which of the following commonly regulates enzyme activity in vivo? O a. An amino acid replacement O…
A: Introduction Enzymes are proteins that function as catalysts in biological reactions to speed up the…
Q: Which of the following is incorrect about the enzyme chymotrypsin? a. It catalyzes the addition of…
A: It does not catalyze the addition of water across a double bond. It is basically a serine protease…
Q: Lysozyme catalyzes a "bi-bi" reaction, which means there are (how many) reactants and (how many)…
A: The reaction that is catalyzed by single enzyme and involves two substrates and two products is…
Q: What is the minimum and maximum pH in which Enzyme X will work? What is the optimal (best) pH? Y X 1…
A: Enzymes are chemical substance formed of proteins which accelerate the rate of reaction . They are…
Q: Which of the following statements DOES NOT describe the enzyme active site? O Itis where the…
A: Enzymes are a class of proteins except for ribozymes (class of RNA) that elevates the pace of the…
Q: Which of the following cofactors will eventually dissociate from their enzyme polypeptide given…
A: Enzymes are usually composed of proteins that contain active sites for the substrate binding. It…
Q: Although we have introduced catalytic strategies separately, an enzyme typically employs more than…
A: Serine proteases are enzymes that have a serine residue in their active site which plays a vital…
Q: Allosteric activators block the active site, so the enzyme cannot bind True Or False
A: Enzymes are proteins in nature, that are capable of altering rate of chemical reactions taking…
Q: For a lot of enzymes that work on fatty acids, the rate determining step is the release of the…
A: Firstly, fatty acid synthesis is synthesis of fatty acids from acetyl-CoA and also utilizing NADPH.…
Trending now
This is a popular solution!
Step by step
Solved in 4 steps

- Why Do Anabolic and Catabolic Pathways Differ? Why is the pathway for the biosynthesis of a biomolecule at least partially different from the pathway for its catabolism? Why is the pathway for the biosynthesis of a biomolecule inherently more complex than the pathway for its degradation?The active site of an enzyme that uses a general acid-base catalytic mechanism contains a Glu and an Asp residue (both of which are essential for catalysis) with pKa values of 5.9 and 4.5, respectively. If the enzyme is found in the lysosome (pH = 5.2), which residue will act as the general acid and which will act as the general base during the initial steps of the reaction?#1 Specify the role each of the following amino acids play within the crystal structure and/or active site for Be as specific as possible, with pictures (and mechanistic arrows) as necessary. His11 Arg140 Glu89 Trp68 #2 Provide a step-wise mechanism for the reaction Bisphosphoglycerate mutase catalyzes, using the amino acids responsible for aiding in catalysis. You do not need to add surrounding amino acids that aid in substrate specificity. (drawn out)
- In the first step of the aldolase reaction, an active site Lys229 residue, with its side chain amino group in the deprotonated state, acts as a nucleophile and attacks the carbonyl C2 carbon of fructose 1,6-bisphosphate to form a Schiff base (boxed in the scheme). Since the pKa of the Lys side chain amino group in free solution is ~10.5, the pKa of Lys229 side chain must have been perturbed to a (higher lower) value for the enzyme to be active at neutral pH. the answer should include sufficient details, including the definition of pKa.An enzyme contains an active site aspartic acid with a pKa = 5.0, whichacts as a general acid catalyst. On the accompanying template, draw thecurve of enzyme activity (reaction rate) versus pH for the enzyme (assumethat the protein is stably folded between pH 2–12 and that the active siteAsp is the only ionizable residue involved in catalysis). Briefly explain theshape of your curve.A prokaryotic species is facing a new environmental stressthat can be ameliorated by a catalytic activity that requiresthe side chain of a unique amino acid derivative called pyrovaline. How would such an organism develop a mechanism for the incorporation of this nonstandard amino acidinto an enzyme molecule? What would be the properties ofthe molecules required to solve this problem?
- How would chymotrypsin's catalytic triad be affected by extremely low and extremely high pH values (assuming the rest of the protein remains intact)?Trypsin, a peptidase that hydrolyzes polypeptides, functions in the small intestine at an optimum pH of 7.7–8.0. How is the rate of a trypsin-catalyzed reaction affected by each of the following conditions?During chymotrypsin-mediated catalysis, which of the following statements is true? Select any/all answers that apply. A. A basic (positively-charged) side chain of the substrate polypeptide sits within the oxyanion hole of the enzyme. B. A basic (positively-charged) side chain of the substrate polypeptide sits within the specificity pocket of the enzyme. C. Ser195 hydrogen-bonds with the backbone carbonyl group of the substrate polypeptide within the oxyanion hole of the enzyme. D. All three residues that comprise the catalytic triad of the enzyme interact directly with the substrate polypeptide chain. E. Both covalent catalysis and acid-base catalysis occur.
- Studies at diff erent pH’s show that an enzyme has two catalytically important residues whose pKs are ∼4 and ∼10. Chemical modifi cation experiments indicate that a Glu and a Lys residue are essential for activity. Match the residues to their pKs and explain whether they are likely to act as acid or base catalysts.At what substrate concentration would an enzyme with a kcat of 25.0 s-1 and a KM of 3.5 mM operate at 25% of its maximal rate? How many reactions would the enzyme catalyze in 45 seconds when it is fully saturated with substate, assuming the enzyme has one active site?for the following reactions: arrive at the Michaelis menten enzyme kinetics: E+2A <-----k1,k2----->A.E+A reversible A.E+A<------k3,k4------>A 2E reversible A2.E------k5--------> E+CD non-reverisble show all steps

