Q: How many millimoles of solute are contained in a) 250.0 mL of 0.0423 M KSCN? b) 500.0 mL of a…
A: Given Multiple question here.
Q: An arctic weather balloon is filled with 27.5 L of helium gas inside a prep shed. The temperature…
A:
Q: A vehicle is moving along a straight line having an initial speed of 10 m/s. It is experiencing a…
A:
Q: [Review Topics] [References] Use the References to access important values if needed for this…
A: Detail mathematical calculation is shown below
Q: A sensitive method for 12 in the presence of Cl₂ and Br₂ entails oxidation of the 12 to 103 with…
A: Given, mass of sample = 1.307 g Volume of thiosulfate = 19.72 mL Concentration of thiosulfate =…
Q: Question 13 For each item, indicate if it is true for galvanic cells only, electrolytic cells only,…
A: Electrochemistry can be defined as the branch of science that deals with production of electricity…
Q: What is the equilibrium constant K at 25°C for an electrochemical cell when E° = +0.0360 V and n =…
A:
Q: Instruction: Show your solutions. 2. A 0.5843 g sample of a plant food preparation was analyzed for…
A:
Q: How much water can you extract from 1.00 L of 0.35 M solution of sugar at 40 C by exerting a…
A: We will use PV= nRT
Q: 07 Question Consider the following standard reduction potentials. O₂(g) +2H₂O(1)-4e-40H (aq) E-0.401…
A:
Q: What is the reaction between sodium hydroxide and water? And why do you feel heat once you mix them…
A: Reaction between water and NaOH. NaOH(s) + H2O ---> NaOH(aq.)
Q: 3. Which polymer has higher Tg? O O 0 || -C(CH₂)4CNH(CH₂)6NH- C(CH₂)4CO(CH₂)40- (A VS. B) +CH₂-CH+ n…
A: Factors affecting the glass transition temperature (tg) : 1. Molecular weight : increases Molecular…
Q: Short Answer Section 1. 10.0 mL of 0.40 M weak acid is being neutralized with 0.10 M NaOH. The…
A: Given the molarity of the weak acid, HA = 0.40 M Volume of HA taken = 10.0 mL Molarity of NaOH used…
Q: 3. Draw the tripeptide described by Phe-Trp-Cys. Your drawing can be condensed structural formula,…
A: Amino acids contain carboxylic acid and an amine functional group on the same carbon atom. Amino…
Q: Calculate the standard free-energy change at 25° C for the Haber synthesis of ammonia using the…
A: Given : N2(g) + 3H2(g) → 2NH3(g) ∆H° = -92.2 kJ ∆S° = -198.7 J/K T = 25°C To calculate : ∆G°…
Q: + e → Cu+ (aq) +2e → Sn²+ (aq) + 2e → Pb(s) + 2e → Ni (s) + 2e- → Co(s) ) + 2e → Cd(s) -) + 2e → →…
A: From the given electrochemical series, Cathode is Pb electrode and Ag is anode E°cell = E°c - E°a
Q: (d) What is a hychiele? Outline the Step wise mechanisy Involved in the reaction of lithium…
A: Detail solution is given below
Q: IR.HNMR.C.NMR what the?
A: The IR and NMR spectroscopy helps us to find the structure of the unknown organic compounds.
Q: Calculate AG in kJ-mol¹ at 298 K for the following reaction: 2 A(g) + B2(s) 2 AB(g) AGO = 196…
A: Given data : 2A(g) + B2(s) -----> 2AB(g) standard gibbs free energy = 196KJmol-1 gibbs free…
Q: Draw the structure of the starting material in this hydration experiment.
A: This experiment is synthesis of hexan-1-ol by hydration of hex-1-ene.
Q: Gijven donda: ware speed Frequency Find wavelength. 140m/s =- 250 Hz
A: Given, Speed of a wave (c) = 140 m/s = 140 m.s-1 Note: 1 Hz = 1 s-1 Frequency of a wave (f) = 250 Hz…
Q: Solid zinc nitrate is slowly added to 175 mL of a 0.0572 M potassium sulfide solution. The…
A: Here we are required to find the concentration of zinc which is needed to just initiate the…
Q: What is the activity
A:
Q: my moles of
A:
Q: The diagram below illustrates graphically how the free energy changes during the chemical reaction:…
A: For the given chemical reaction, reactants → products
Q: A liquid mixture containing 30 mole% isopropanol and 70 mole% water is heated in a closed container…
A: Given that mole% of isopropanol=30% mole% of water=70% the mixture is heated to 83oC and separates…
Q: choose the best acid with which to make a buffered solution with pH =7.00. Describe how you would…
A: To identify which of the given acid will be best for preparing a buffer solution with pH 7.00.
Q: For the following show the calculation for both trails b) Volume of NaOH c) moles NaOH d) moles of…
A: Dear student, As you have asked multiple subpart questions but according to guidelines we will…
Q: 3 points Save Answer Consider the following reaction with vanadium and iron: 2 Fe²+ (aq) + √²+ (aq)…
A: Fe3+(aq) + e- -----> Fe2+. E° = +0.77 V V2+. + 2e- -----> V E° = -0.49 V
Q: You add 50.00 uL of 25.00 M NaOH (m = 39.9971 g/ mol) to 100.0 mL of a 0.252 mol/L NaCI (7=57.959 g/…
A: NaOH is a strong base while NaCl is an ionic salt hence it can't form buffer or can't react with…
Q: Question 2 V provides information about the position of equilibrium ✔ provides information about…
A:
Q: convert 5 mg of gold per mg of activated carbon to mg of gold per gram of activated carbon.
A: SOLUTION 1 mg = 0.001 g 1 mg activated carbon → 5 mg Au Or, 0.001 g activated carbon→5 mg Au Hence,1…
Q: Jaya needs to measure the unknown concentration of Sn2+. A Galvanic cell is set up in a room with a…
A:
Q: Provide suitable IUPAC name for each of the following compounds.
A:
Q: Calcium oxide and carbon dioxide react to form calcium carbonate, like this: CaO(s)+CO,(g)–CaCO3(s)…
A:
Q: Calculate AG in kJ.mol-1 at 298 K for the following reaction: 2 A(g) + B2(g) → 2 AB(s) AGO = -264…
A: GIVEN: Calculate ∆G in kJ.mol-1 at 298 K for the following reaction: 2 A(g) + B2(g) = 2 AB(s) ∆G°…
Q: D X Ⓡ Y Salt bridge A+ B- X2+ -Y2+ In this cell, the half-cell containing X is the In this cell, the…
A:
Q: When aqueous solutions of K₂SO4 and CrCl3 NiBr2 and AgNO3 Li₂CO3 and Csl O Nal and KBr O KOH and…
A: In a double displacement reaction if one of the product is soluble and another is insoluble then…
Q: Propose an efficient synthesis CI MeO O OMe OEt COOH Br NO₂ O EtO
A: We have to propose an efficient synthesis.
Q: provides information about the spontaneity of a reaction/process; and <<Question 20 of 32 provides…
A: Thermodynamics is branch of chemistry in which we deal with amount of heat evolved or absorbed by…
Q: Patty needs to measure the unknown concentration of Sn2+. A Galvanic cell is set up in a room with a…
A:
Q: Calculate the volume of 0.0407 M EDTA needed to titrate the Ca in a 0.1622 g CaCO3 (MM = 100.0869…
A: We knew that in complexometric titration EDTA use as titratrent .EDTA makes 1:1 complex with matel…
Q: A 1.50 L buffer solution is 0.250 M in HF and 0.250 M in NaF. Calculate the pH of the solution after…
A:
Q: volume of this solution
A:
Q: de, what are the products (=
A:
Q: What is the value of AG (kJ mol-1) for this reaction at 320.1 °C? The data refer to 25 °C: 2 PC13…
A:
Q: 3. Which polymer has higher Tg? 0 0 0 || || || || -C(CH₂)4CO(CH2)40- -C(CH₂)4CNH(CH₂)6NH- (A VS. B)…
A: Since you have asked multiple questions we will answer the first one fir you. To get the remaining…
Q: Which compound has the element X with the oxidation number of +5. (select all that are correct)…
A: Below attached file showing the details answer. All the best.
Q: A system consists of three energy levels: a ground level (&o= 0, 90 = 1); a first excited level (1 =…
A:
Q: To study the kinetics of the reaction between acetone and iodine N/10 NaHCO3 solution in the conical…
A: To study the kinetics of the reaction between acetone and iodine N/10 NaHCO3 solution.
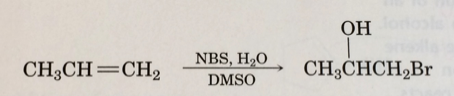
Step by step
Solved in 2 steps with 1 images

- Limestone rocks are composed mainly of calcium carbonate (CaCO3) that reacts easily with an acid, releasing carbon dioxide gas.Complete and balance the following equations. Write the entire equation, including the reactants and products, and include the physical state of each species. (a) K(s) + H2O(l)→ (b) NaH(s) + H2O(l)→Complete the balanced chemical reaction for the following weak base with a strong acid. In this case, write the resulting acid and base as its own species in the reaction. CH3NH2(aq)+HClO3(aq) ->

