Q: 16. A child takes 15 mg of elemental iron PO BID. How many milliliters of ferrous sulfate 500…
A:
Q: O2.16 atm Predict the products for the following reaction (ignore balancing): Zn (s) + O Zn(SO4)2…
A: Different types of reaction: a) Precipitation reaction: In precipitation reaction an insoluble salt…
Q: What would be the major product of the following reaction sequence? 1. HBr, ROOR, hv 2.BuOK, BuOH,…
A: The reactant is an alkene. #1. In the presence of peroxide(RCOOR), HBr and alkene undergo…
Q: Using Kay's method, what is the number of moles of this mixture in the tank?
A: As per Kay's method ; the no. of moles of a mixture in tank is 2.27 Kmoles ( option A is correct )
Q: 2-Stretching frequency of the O=C bond of a compound (3-butene-2-on) is less than the stretching…
A: We have to select the factor which explains the given statements based on IR spectroscopy
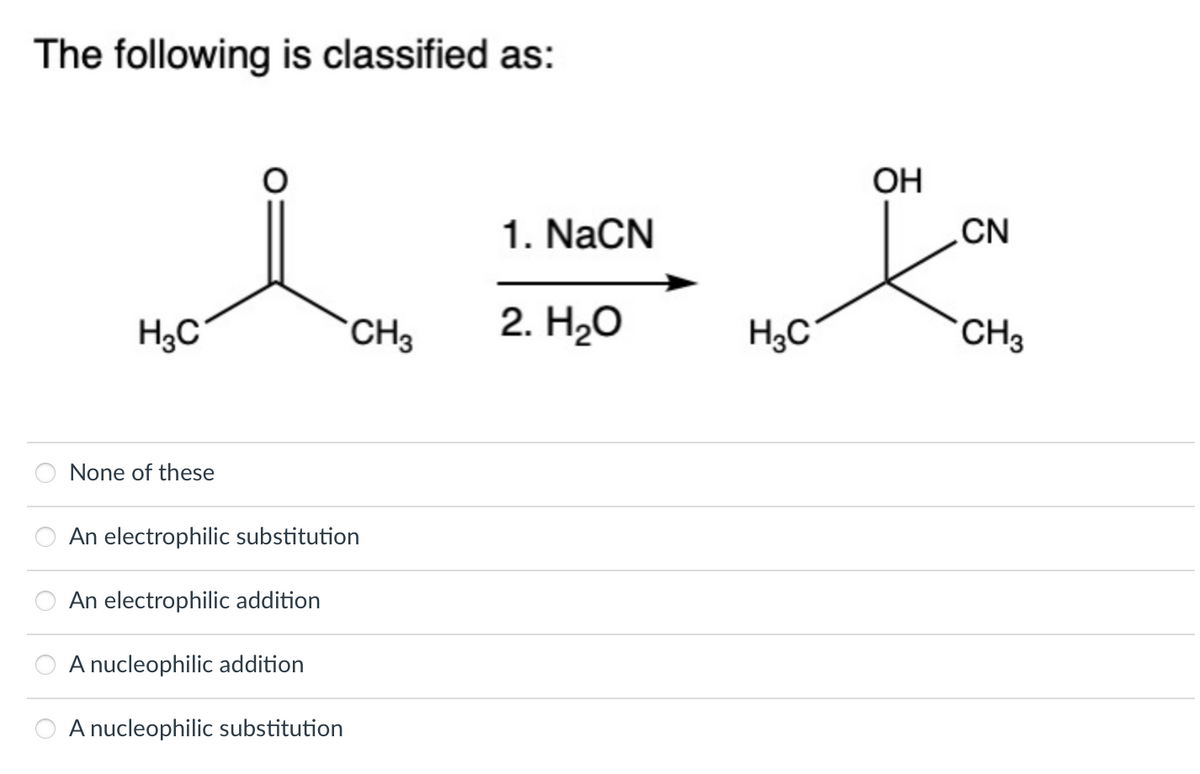
Q: What is the major product of this reaction? O H HCN ???
A:
Q: Before the use of CFCs, sulfur dioxide was used as a refrigerant. A sample of sulfur dioxide…
A:
Q: 1) Describe a simple chemical test to differentiate the following pairs of compounds. Write the…
A: #1: (i): 2-propanone and propanoic acid: 2-propanone(CH3COCH3) has terminal -COCH3 group that gives…
Q: Which of the following statements appropriately describes the colors in each diamond of the chemical…
A: Answer 2 is correct; that is C (c) Red indicates the fire (flammability) hazard, blue…
Q: What alkyl halide would be the starting reactant for this reaction? 1. Mg0 2. ??? B с 3. H+ None of…
A: Grignard reagents reaction with carbonyl compound is a good way to increase the number of carbon…
Q: Link a description with an equation - - . . Second-order integrated rate law First-order integrated…
A:
Q: . Express the sodium ion concentration for 500 mL of ½ NS as a ratio strength (w/v) AND parts-per-…
A: Hi there, as there are multiple questions posted. we are answering first question. if you need…
Q: Please send me the question in 30 minutes it's very urgent plz
A: According to henry's law: Henry's Law constant(Kh) for Helium in the blood = 4.44 x 10-4 M atm-1.…
Q: From the solubility data given for the following compounds, calculate their solubility product…
A:
Q: A basic drying agent should not be used to dry an acidic Which of the following is a basic drying…
A: Given statement is : A basic drying agent should not be used to dry an acidic. Which of the…
Q: 1. The symbol, , on organic glassware means O (c) standard taper joint O (b) standard size glassware…
A: 1) We have to write the meaning of the symbol given
Q: (B1) The following Compound can undergo Signifiant Kefo-enol + possesses enhan ced enot form.…
A:
Q: What pressure of gas is produced from the reaction of 78.3 g of Zn with 125 mL of 12 M HCI in a 2.6…
A: The reaction is ; 2HCl(aq) + Zn (s) ---> ZnCl2(aq) + H2(g) 78.3 g zinc react with 125 mL 12M…
Q: Chemistry Draw out a complete mechanism for imine formation from the reaction of 4-iodoaniline will…
A: Concepts of imine formation.
Q: Select the reagents from the following list which would perform the following transformations in the…
A:
Q: Write correct IUPAC names for the following structures. (a) CH₂CH₂CH=CHCHCH=CHCH, I CH,CHCH, (b) 3…
A: Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: Draw out a complete mechanism for imine formation from the reaction of 4-iodoaniline will be…
A: The mechanism of the reaction is as follows:
Q: and not the identity of the element) 17 X 2+ Protons 7 Neutrons 10 electrons name (identity) of…
A: In atoms, electrons are revolving around the nucleus. Nucleus is made up of protons and neutrons.
Q: How would the series of figures change in the presence of a catalyst? Would there be different…
A: Given:
Q: Calculate the change in energy (in Joules) when an electron transition from n=4 to n=2 in a hydrogen…
A: The electronic energy levels in an atom are quantized and when an electron undergoes a transition…
Q: If 20.15% of the atoms in a sample of tin are replaced by copper atoms without any change to the…
A:
Q: Which is the correct half reaction for the reduction of iodine to iodide ions?
A: Since, Reduction reaction means gain of electron. Thus,
Q: What is the name of a reaction that we have been studying that produces a 3-ketoester?
A: A question based on introduction to organic chemistry that is to be accomplished.
Q: The mean free path of a gas at a temperature T1 and a pressure P1 is 7x10-5 cm. At these temperature…
A: Mean free path (λ) = 7×10-5 cm Number of molecules per unit volume (N) = 2×1018 molecules/cm3 We…
Q: A voltaic cell is constructed in which the cathode is a standard hydrogen electrode and the anode is…
A:
Q: Which of the following bonds would produce the weakest absorption? B) O-H C) sp³ C-H A) symmetrical…
A: •ABSORPTION IN SPECTRUM:- Absorption in spectrum is depend upon the polarity of bond. • Relationship…
Q: 4) Which of the following compounds is least reactive in the nucleophilic aromatic substitution…
A: Given:
Q: Super fine 40-gauge copper wire has a diameter of only 0.080 mm and wei wire is pulled off to wind a…
A: Given: Diameter of the gauge is 0.080mm The weight of the gauge is 705.g The weight was reduce due…
Q: If CuCl2 has a Ksp of 1.9 x 10-7 and a solution of CuCl2 has a chloride ion concentration of 6.0 x…
A: We need to find the solubility of solution.
Q: Which of the two setups below is more appropriate for the separation of two liquids in the…
A: Separating funnel is used to separate immiscible liquids , when these tere separatory immiscible…
Q: What mass of Mg and what volume of 1.50 M HCI are required to make 62.3 L of H₂ gas at a temperature…
A:
Q: Sutur des formed by the catalytication of sute doe 250()+ 0) 250) All-1978 kl/mol AS= 188.0 J/K…
A: Given: T = 25°C or 298 K ∆H=-197.8 kJ ∆S=-188.0 J/K
Q: Propose routes to the following compounds from propyl bromide and any necessary organic or inorganic…
A:
Q: 5. Write forward and reverse reactions for the following: CH4 (g) + 2H2S (g) (g) ==== CS2 (g) + 4H2…
A:
Q: What is the general class of reaction of potassium cyanide? How much heat must be added or removed…
A:
Q: If AuBr has a Ksp of 5.0 x 10-17 and a solution of AuBr has a bromide ion concentration of 7.1 x…
A:
Q: Propose preparative routes to the following ketones using an enamine intermediate - you must make…
A: In this question, we have to synthesized the given product a) and b) by using enamine intermediate.…
Q: 6 8 O A OB OC What is the major product when cyclohexanone is reacted with ammonia and trace acid?…
A: It is given that cyclohexanone is reacted with ammonia and trace acid .Let's find the major product.
Q: The following information is given in a science supply catalog: 60,00 rubber stoppers = 500.00 grams…
A:
Q: Perform a retrosynthetic analysis of the target molecule shown below, from starting materials…
A: Please find the attachment.
Q: Chlorineoxide (ClO), which plays an important role in the depletion of ozone decays rapidly at room…
A: We can find the order of the reaction by the graphical method of order determination. If ln[A] Vs t…
Q: Use the standard reduction potentials given below to predict if a reaction will occur between Ag(s)…
A: Given data F2(g) +2e-→2F-(aq), E°red = 2.870 V Ag+(aq) +e-→ Ag(s), E°red = 0.799 V. The overall…
Q: Decide how the compounds below will partition during liquid-liquid extraction of water with an…
A: Polar compounds soluble in polar solvents( H2O) Non polar compounds soluble in non polar solvents
Q: electronic ure must be planar or nearly planar so that overlap of these port must be cyclic with…
A: Aromatic compounds are those which are cyclic, planar and have (4n + 2) pi electrons.
Q: An analytical chemist has been given the task of precipitating lead cation (Pb2+) out of solution…
A: Given: The Ksp of lead iodide = 9.8 x 10-9 The concentration of Pb2+ = 0.424 M We have to find the…
Step by step
Solved in 2 steps with 2 images

- 1 example of reaction for each of the types of organic reactions: substitution, elimination, addition, and rearrangement. Show:a. the overall reaction (reactants --> products)b. the reaction mechanism (indicate intermediate product)c. indicate which is the reactive species or intermediate in the reaction (radical? electrophile? nucleophile?)d. overall description of the reaction eg., radical substitution or SRWhich is the major product?a. Ab. Neither product would likely formc. BExplain Terminal Alkynes—Reaction as an Acid ?
- a) Predict the major product of the reaction and identify the type of reaction (substitution or addition). Explain the function of Pt used in the reaction. (with First image) b) Write the reaction of the mononitration of chlorobenzene and predict the major products with reasons. c) Identify all the carbon atoms in the following compounds that are sp2 (Circle the carbon(s) your selected on image two)Does the following SNAr reaction occur accordingto an addition-elimination or an elimination-addition mechanism? Motivate.Add curved arrows to show how carbocation A is converted tocarbocation B. Label each new σ bond formed. Similar reactions havebeen used in elegant syntheses of steroids.
- 1. When you add H-Br in a terninal alkene, the product has the rule od Markovnikov? 2. The type pf intermetary thag forms when we add HgSO4/H2SO4/H2O in an alkyl is? Please solve my both questions.thank you.Below is the equation for a nucleophilic substitution reaction and some experimental data. CH3CH2Br + CH3COO- ⇌ CH3CH2CO2CH3 + Br- Rate = k [CH3CH2Br][CH3COO-] Which mechanism would best fit the data?Each H↔H eclipsing interaction in ethane costs about 4.0 kJ/mol. How many such interactions are percent in cyclopropane? What fraction of the overall 115 kJ/mol (27.5 kcal/mol) strain energy of cyclopropane is due to torsional strain.



