Q: (3) The rate constants for the exchange reaction: [Cr(H2O)sX]? + [*Cr(H2O)6]?[*Cr(H2O)sX] +…
A:
Q: The acid-base indicator "Thymol Blue" has two transition ranges as listed below (with the…
A: The pKa values of the forms of thymol blue can be calculated as: pKa=-log Ka
Q: Predict products X and Y in the reactions shown below: 1) NaOH, H,0, heat 1) DIBAL, H,O, EN 2) H,0…
A:
Q: draw prduct of rxn
A:
Q: For the following reactions at equilibrium, identify which between the reactants and products is…
A:
Q: Please provide the reagents that will accomplish the following transformations. More than one…
A:
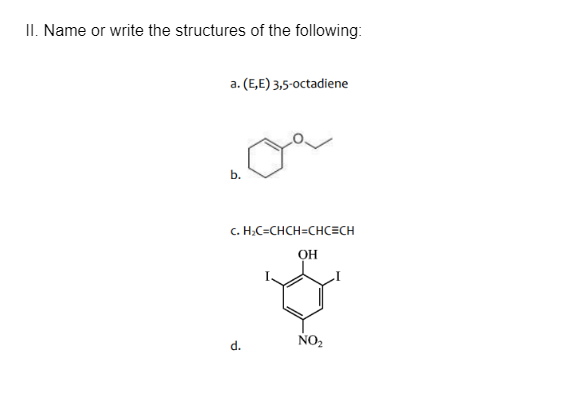
Q: II. Name or write the structures of the following: a. (E,E) 3,5-0octadiene b. c. H;C=CHCH=CHC=CH OH…
A:
Q: छोल्सद पह CHormie सपलtm तिम्यणा पप उ०. P৮N6 + ctmmp०৪ Pb3০५+ CU2०3+Mmd.tNo
A:
Q: In the addition reactions of alkynes, the reagents undergo transformation to form first. a. a…
A:
Q: The number of protons determines the element. True O False
A: To solve this we have to know about proton .
Q: Write the equilibrium expressions for the following balanced equations: a. 7H2 (e) + 2NO2 () 2NH3…
A: Write the equilibrium expressions of the given reactions----
Q: Name the following compounds: a. Br C.
A:
Q: TRUE OR FALSE: Allylic carbons can be a substrate for halogenation via electrophilic addition.
A: In this question, we will see that the given statement in the above question is true or false. You…
Q: molecular equation F, (g) + NaCl (aq) – net ionic equation 2. molecular equation K (s) + HBr (aq) →…
A:
Q: true or false. There is nothing wrong with the reaction below
A:
Q: What are the different shampoo ingredients and its desciption? you may also cite an example of it.
A:
Q: Which of the tollowing is a bufter system? Ο ΗΝΟ2 and KNO2 O S(OH)2 and Sr(Br)2 O HCl and LiCI O…
A: Buffer solution: The solution that resists the change in pH is known as a buffer solution. The…
Q: Given the unbalanced equation: C6H6 + O2 ---> H2O + CO2. When properly balanced, the coefficient…
A: Given, Unbalanced equation, C6H6 + O2 ---> H2O + CO2
Q: Write the half-cell equations, cell reaction equation and cell representation for the following…
A: Recall the given reaction, Cd s + NiOOH s → CdOH2 s + NiOH2 sWe have to…
Q: Calculate delta H for the following
A: To solve this problem we will use the given thermochemical equations.
Q: 1. What process is responsible for the increase in the total negative charge as soil pH increases?…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: но. 1. HO 2. 3.
A: Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: write an essay explaining bonding in water cohesion.
A: Cohesion means how much molecules like to stick to itself. For water molecule there operate very…
Q: Calculate biological standard potential of the NAD+/NADH couple at 25°C. The reduction half-reaction…
A:
Q: If 0.00864 mol neon gas at a particular temperature and pressure occupies a volánie of 228 ml, what…
A:
Q: Complete the following reactions (please be aware of regioselectivity): CH3. c=CH2 + a. HBr -> b.…
A: ->HBr give electrophilic addition reaction to double bond. ->Pd/H2 is reducing agent which…
Q: What is the molarity of a solution in which 7.1 g of sodium sulfate is dissolved in enough water to…
A: Weight of sodium sulfate = 7.1 gm Volume = 100 ml = 0.100 L (1L = 1000 ml)
Q: If 0.52kJ of heat is needed to raise the temperature of a 200g substance twenty degrees Celsius,…
A: The amount of energy needed to raise the temperature of unit mass of a substance by 1oC is known as…
Q: Calculate Eº for the process PbI6) + e- → (s) + e > Pbs) + 21,
A: Given reaction is : PbI2 + e- <---------> Pb (s) + 2I2 Find standard cell potential = E0 = ?…
Q: What are the units of the rate constant for a first ordereaction? M-sec M sec sec O M2
A: Which one of the following is correct answer
Q: 3. What happens when you treat unknown molecule A with lithium aluminum hydride (LIAIH.) and then…
A: Consider CH3CN is the substrate and whenever this is treated with LiAlH4 followed by Acidic…
Q: 3) The volume of a certain liquid varies with temperature as V=V'{0.75 +3.9 x10-4(T/K) +1.48…
A:
Q: CH;CH,CH2C=C-H ... A Н—С—с— Н and NaNHz, NH3, СH,CH,CH,Br B Н—С—С—H and NaNHz, NHз, CH,СH2CH-CH,Br…
A:
Q: To measure enthalpy of a reaction, we can use calorimetry under what condition? O constant…
A: In this question, we will see the condition for measuring enthalpy of a reaction by using…
Q: a solution contains 60.0 mL of 0.100 M HCl. let’s add 5.00 mL of the titrant, 0.200 M NaOH. What is…
A: When HCl react with NaOH it is acid base reaction and produced salt Nacl and water
Q: How much heat in kilojoules is released when 209.0 g hydrogen bromide (HBr) is dissolved if its…
A: Given: Molar heat of solution = -85.1 kJ/mol Mass of HBr = 209.0 g Known: Molar mass of HBr = 80.91…
Q: There are two benzylic bromides that give (Z)-3,5-dimethyl-2-phenyl-2-hexene on E2 elimination. Draw…
A:
Q: 2) When a certain freon used in refrigeration was expanded adiabatically from an initial pressure of…
A:
Q: CH2-C= CH
A:
Q: metallic character increases as you move to the right acrosss a row in the periodic table and…
A: The correct option is C)metallic character decreases as you move right to the periodic table and…
Q: How many amperes are required to deposit 0.291 grams of magnesium metal in 213 seconds, from a…
A: Given, Mass of Mg metal deposited = 0.291 g Molar mass of Mg = 24.305 g/mol Time = 213 second
Q: Explain the concept of Boyle's law in spray paint.
A: Boyle's Law:-According to Boyle's law at a fixed temperature, the volume of gas is inversely…
Q: 10. Applying the Michaelis-Menten Equation III A research group discovers a new version of happyase,…
A:
Q: A gas cylinder contains 2.10 mol He, 2.30 mol Ne, and 1.20 mol Ar. If the total pressure in the…
A: According to the Dalton’s law For a non-reacting gaseous mixture sum of partial pressure of…
Q: 6) Evaluate (dS/aV)† for a van der Waals gas. For an isothermal expansion, will AS be greater for an…
A: GIVEN & TO FIND:-
Q: A. At the composition of 61.9 %wt Sn, no solid phase exists. B. As more Sn is added the mixture…
A: For Pb-Sn phase system, 61.9 wt % is the eutectic composition, i.e. the liquid directly changes to…
Q: 1a) How long will it take for 30 g of 222Rn to decay to 7.5 g? 1b) How many grams of 1°N will be…
A:
Q: Evaluate (∂S/∂V)T for a van der Waals gas. For an isothermal expansion, will DS be greater for an…
A:
Q: TYPEWRITTEN ONLY PLEASE UPVOTE.
A: Given that μ 0.25 K/atm for nitrogen, calculate the value of its isothermal Joule-Thomson…
Q: 1. Zn + HCI — ZnClz + Н2 2. CH4 + 02 СО2 + Н20 3. Fe + NaBr → FeBr3 + Na
A: We are authorised to solve first three subparts of a question. Please post rest of the subparts…
Step by step
Solved in 3 steps with 3 images

- Organotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides. One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(CzHs)3). 3SnCl4 + 4Al(C2H5)3 → 3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.160 L of SnCl4 (d= 2.226 g/mL) was treated with 0.346 L of triethylaluminum (Al(C2H5)3); d = 0.835 g/mL). What is the theoretical yield in this experiment (mass of tetraethylstannane, Sn(C2H5)4)? If 0.257L of tetraethylstannane (d= 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?A mixture of 0.10 mol benzene and 0.10 mol p-xylene was allowed to react with 0.10 mol nitronium ion until all the nitronium ion was gone. Two products were obtained: 0.002 mol of one and 0.098 mol of the other. a. What was the major product? b. Why was more of one product obtained than of the other?Rank the species below in order of increasing nucleophilicity in protic solvent. I. H2O II. CH3S— III. CH3COO— IV. t-BuO— I, II, IV, III I, III, II, IV I, III, IV, II I, II, III, IV
- Describe how the product is purified. 4,4'-DIBROMOBIPHENYL [Biphenyl, 4,4'-dibromo-] Submitted by Robert E. Buckles and Norris G. Wheeler1. Checked by R. S. Schreiber, Wm. Bradley Reid, Jr., and Robert W. Jackson. 1. Procedure In a 15-cm. evaporating dish is placed 15.4 g. (0.10 mole) of finely powdered biphenyl (Note 1). The dish is set on a porcelain rack in a 30-cm. desiccator with a 10-cm. evaporating dish under the rack containing 39 g. (12 ml., 0.24 mole) of bromine. The desiccator is closed, but a very small opening is provided for the escape of hydrogen bromide (Note 2). The biphenyl is left in contact with the bromine vapor for 8 hours (or overnight). The orange solid is then removed from the desiccator and allowed to stand in the air under a hood for at least 4 hours (Note 3). At this point, the product weighs about 30 g. and has a melting point in the neighborhood of 152°. The crude 4,4'-dibromobiphenyl is dissolved in 75 ml. of benzene, filtered, and cooled to 15°. The…Which of the following are intermediates in the reaction of excess methylmagnesium bromide with ethyl benzoate (shown in Image18) to make 2-phenyl-2-propanol? A. I and II B. I and III C. II and III D. I, II, and III (they are all intermediates)In the reaction below, 3-Buten-2-one reacts with 2,3-Dimethyl-1,3-butadieneand 1,3-Butadieneat 140°C and 30°C, respectively. Explain why these two dienes react at different temperatures.
- give the reagents for parts a-pOrganotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides.One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(C2H5)3). 3SnCl4 + 4Al(C2H5)3 3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.230 L of SnCl4 (d = 2.226 g/mL) was treated with 0.396 L of triethylaluminum (Al(C2H5)3); d = 0.835 g/mL). If 0.335 L of tetraethylstannane (d = 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?Adjust the structures below to show the resulting intermediates.
- Tunicates are marine animals that are called "sea squirts" because when they are taken out of water, they tend to contract and expel seawater. Lepadiformine is a cytotoxic agent (toxic to cells) isolated from a marine tunicate. During a recent synthesis of lepadiformine, the investigators observed the formation of an interesting by-product (3) while treating diol 1 with a reagent similar in function to PBr3 (J. Org. Chem. 2012, 77, 3390–3400):Assign the stereochemical configuration (E or Z) for the alkene below. Show your work, indicating clearly which groups are assigned high priority (e.g., through assigning the groups numbers, circling only the high priority groups, or labeling groups as high or low).Chemistry Give the products of the reaction of 1 mole of 2-methy1-1,3-pentadiene with 1 mole of HBr. Whichproduct(s) will predominate if the reaction is under kinetic control? Which products) will predominateif the reaction is under thermodynamic control?

