Improve the reaction efficiency of chemicals and pollutants High potential at interface MNBS have ability to generate hydroxyl radicals by MNBS collapse Degrade organic substances that are difficult to degrade in normal circumstances MNBS are combined with ozone for treating wastewater The enhanced performance of ozone oxidation with azo Uses an MNBS generator and a conventional bubble contactor. dye-containing wastewater Weak acidic conditions The amount of total organic carbon removed per gram of ozone consumed is higher in the MNBS Better efficiency Increase in salinity Higher internal pressure Smaller bubble size results in Larger specific area Oxidants convert contaminants to Follows pseudo-first-order kinetics CO2 in water
Improve the reaction efficiency of chemicals and pollutants High potential at interface MNBS have ability to generate hydroxyl radicals by MNBS collapse Degrade organic substances that are difficult to degrade in normal circumstances MNBS are combined with ozone for treating wastewater The enhanced performance of ozone oxidation with azo Uses an MNBS generator and a conventional bubble contactor. dye-containing wastewater Weak acidic conditions The amount of total organic carbon removed per gram of ozone consumed is higher in the MNBS Better efficiency Increase in salinity Higher internal pressure Smaller bubble size results in Larger specific area Oxidants convert contaminants to Follows pseudo-first-order kinetics CO2 in water
Chemistry: Principles and Reactions
8th Edition
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:William L. Masterton, Cecile N. Hurley
Chapter11: Rate Of Reaction
Section: Chapter Questions
Problem 99QAP: The gas-phase reaction between hydrogen and iodine H2(g)+I22HI(g)proceeds with a rate constant for...
Related questions
Question
Briefly explain about
degradation of azo dyes in wastewater using micro-nano bubbles (MNBs) treatment.
Explain how the process is done.
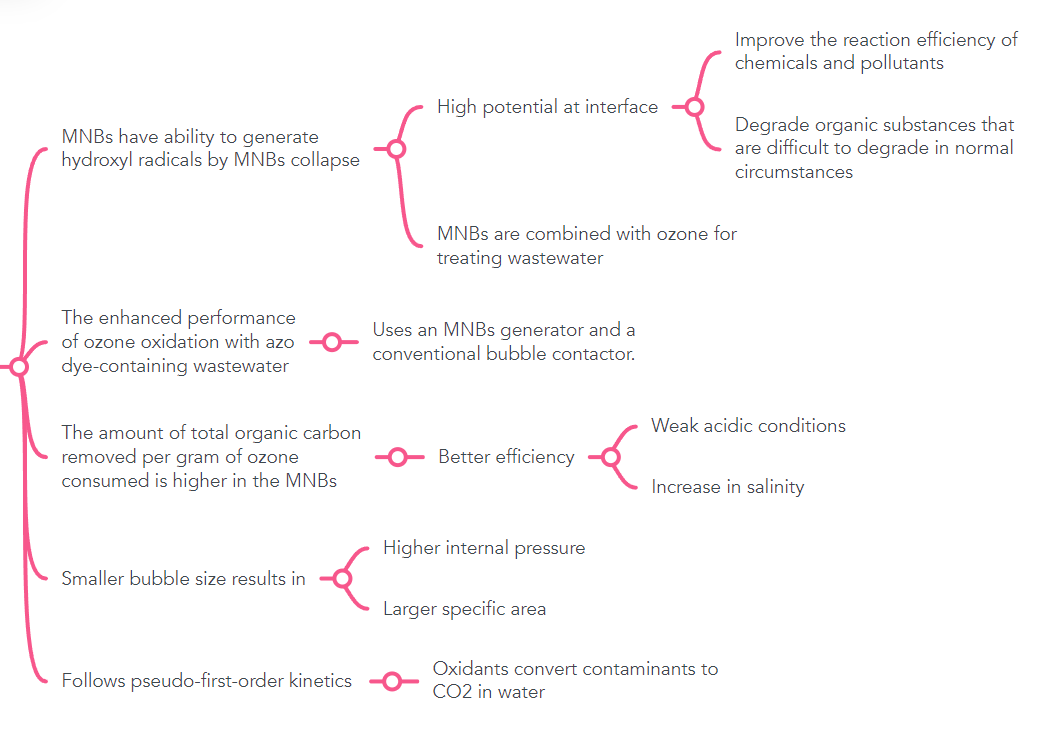
Transcribed Image Text:Improve the reaction efficiency of
chemicals and pollutants
High potential at interface
MNBS have ability to generate
hydroxyl radicals by MNBS collapse
Degrade organic substances that
are difficult to degrade in normal
circumstances
MNBS are combined with ozone for
treating wastewater
The enhanced performance
of ozone oxidation with azo
Uses an MNBS generator and a
conventional bubble contactor.
dye-containing wastewater
Weak acidic conditions
The amount of total organic carbon
removed per gram of ozone
consumed is higher in the MNBS
Better efficiency
Increase in salinity
Higher internal pressure
Smaller bubble size results in
Larger specific area
Oxidants convert contaminants to
Follows pseudo-first-order kinetics
CO2 in water
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning