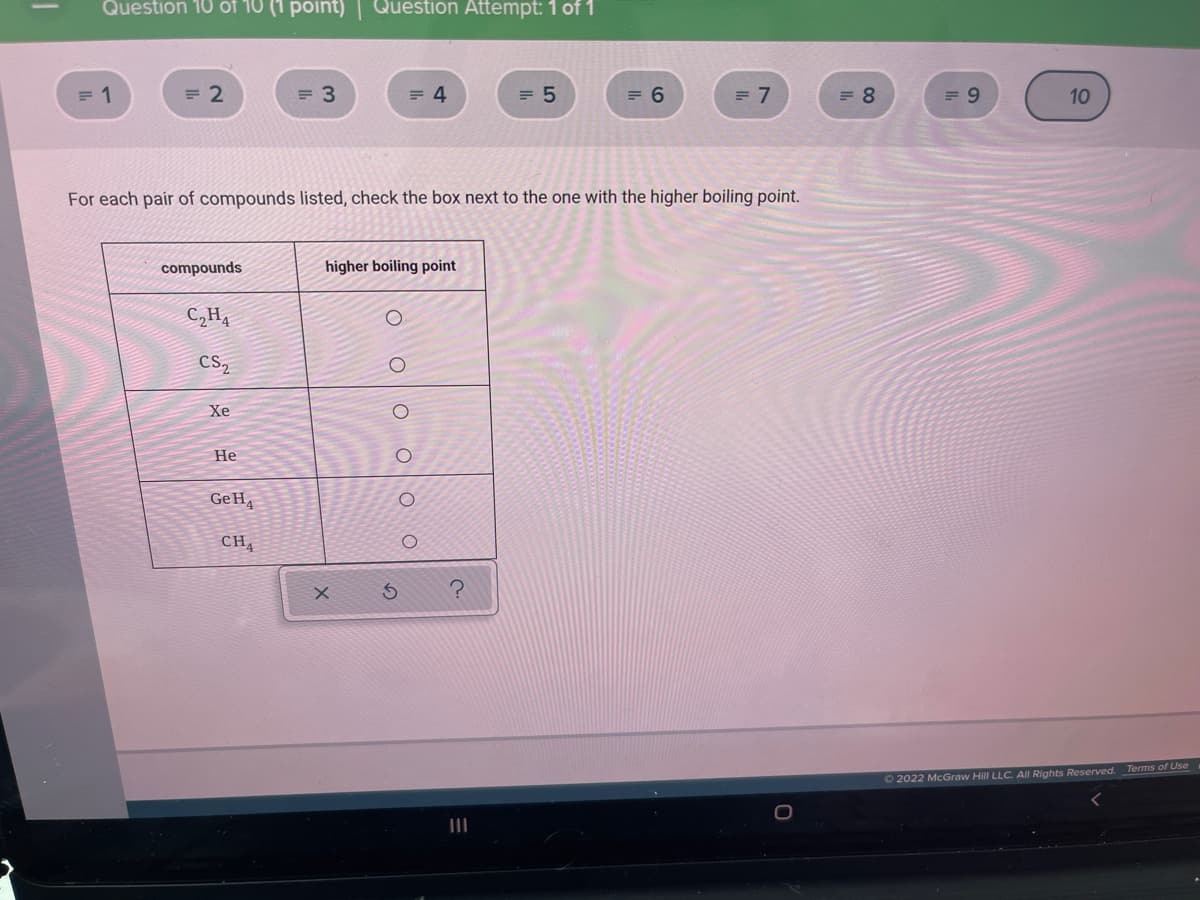
For each pair of compounds listed, check the box next to the one with the higher boiling point. compounds higher boiling point C,H4 CS, Хе Не GeH, CH Terms of Use O2022 McGraw Hill LLC. All Rights Reserved. II O oo oo o
Q: Ses an isotope produced from atmospherc testing of maclear bombs. lf nucler testing was stopped in…
A: Given: Half-life of isotope = 28.5 years. The testing was stopped in 1960. And the analysis is being…
Q: Consider the titration of 20.0 mL of a sample of 0.105 M HC2H3O2 with 0.125M NaOH. (Ka = 1.8 x 10-5)…
A:
Q: Ponatinib HCI, an antitumor drug: H F
A: The hybridisation of the following atoms in the molecule is: i) one of the fluorine atom - sp3
Q: Which of the following is not a characteristic of metals? Group of answer choices basic oxides…
A: 1) Basic oxides Metals have basic oxides.
Q: Why one should swirl vigorously during the process of titration?
A: If you tirate a base into an acid with phenolphtaleine as an indicator, the indicator will go from…
Q: A patient with emphysema is breathing pure O2 through a face mask. The cylinder of O2 contains…
A:
Q: D. None of the above 11. A 315mL of water was added to 2100 mL of 19M NaCI solution. What is the new…
A:
Q: • Observe the following reactions carefully: H2(g) + F2(g) 2HF(g) AH = -537 kJ > C(s) + 2 F2(g)…
A: The reactions given are, 1) H2 (g) + F2 (g) → 2 HF (g) ΔH = -537 KJ 2) C (s) +…
Q: 6. When determining the molecular/electron geometry of a molecule, what information is required?
A: Information required to determine molecular / electron geometry = ?
Q: n the analysis of 0.8972 g of an impure Cl- sample (35.45 g/mol), 1.399 g of AgCl (143.32 g/mol)…
A: Here we are required to find the % of Cl in the impure sample and also find the mass of AgI formed…
Q: What is true about the heated solution upon cooling? A. As energy is released, the concentration of…
A: As per bartleby guideline, an expert is allowed to answer one question at a time. Please re upload…
Q: At a certain temperature, the reaction 2HF(g) H2(g) + F2(g) has K 1.2 x 10 13. Does this reaction…
A: Reaction: 2HF(g) <=> H2(g) + F2(g) Kc = 1.2 × 10-13 Moles of HF = 0.013 mol Volume of…
Q: NO(g) + SO3(g) has K = 5.94. Suppose 0.189 mol of NO2 and 0.189 mol of SO2 are placed At 1000°C, the…
A: At equilibrium- Kc = [Product]/[Reactant]
Q: A multivitamin sample has a label claim of 500 mg ascorbic acid (176.12 g/mol). According to quality…
A: A question based on mole concept that is to be accomplished.
Q: 6) Which one has the weakest conjugate base? a) CH;CH;OH b) HCECH c) NH3 d) HCI e) H;0
A:
Q: Which among the alkanes below has the highest boiling point? A. n-hexane B. isohexane C.…
A: Greater the chain length of hydrocarbon, higher will be the boiling point. Because as chain length…
Q: Refer to the attached picture for the following questions Which among the five aromatic molecules…
A: We know that molecular stacking is decreased when there is steric repulsion in structure 3 there is…
Q: Balance the following equation: 8Be - - > 4He + X 4 O 8He 2 O 4Li 3 O 12C O 4He
A: ->For balancing nuclear reaction we see proton number both side must be equal .Also (proton…
Q: If a positron is to be emitted spontaneously, how much more mass (as a minimum) must an atom of the…
A: As in positron emission, the nuclear positive charge and hence the atomic number decreases by one…
Q: increases, the solubility of alcohol decreases. As the number of branch point increases, the…
A: Solubility is depend on intermolecular forces and branches. In alcohol As carbon chain is non-polar…
Q: 4. A second-order reaction of the type A+ 2B P was carried out in a solution that was initially…
A: Given: A + 2 B -----> P Initial concentration of A = 0.085 mol/dm3 Initial concentration of B =…
Q: For the formation of Carbonyl Chloride: 2Cl, + CO --> COCI, + 2CI The given 3 step mechanism has…
A: Given chemical Reaction :- 2Cl2 + CO ---> COCl2 + Cl Rate = K [ reactant ]order
Q: O3 → O2 + O (fast) O3 + 0 → 202 (slow)
A: O3 ---> O2 + O. (Fast) O3 + O ---> 2O2. (slow)
Q: Name the following compound H2N. Br 0 2E45)-4-bromo-3-vinylhept-2-en-5-yn-2-amine O…
A:
Q: A voltaic cell has the following initial state: Cr (s) | Cr (aq, 2.0 M)|| C* (aq, 9.0 M) | Cr (s)…
A: The cell given is, Given: Volume of cell = 1.00 L And current = 26 A
Q: Which substance is the primary standard used for the accurate determination of HCl titrant…
A: Primary standard which can be weighed easily, and which is so pure that its weight is truly…
Q: Refer to tne reactiOH Below 1o ans Items HBr- -> --- CIH CIM CIH
A: ->One double bond required one mole of HBr .
Q: Determine whether the statements are true or false and could you please leave a short explanation in…
A: 1. Vitamins are useful in the body in great amounts. -----True Vitamins and minerals boost the…
Q: lodine-131 is used to treat Graves disease, a disease of the thyroid gland. The amount of 131 used…
A: As per statement, 86 microcuries required per gram of thyroid gland, so for 23g of thyroid gland =…
Q: ? O3 a. b. Zn, H*
A:
Q: Refer to the equation below to answer questions 26 to 30. Br HII CIN
A: For saturation of one double bond , one mole of halogen is required . Detail solution is given below
Q: 3C. Identify the element or ion from the following information. X*t: [Ar] 4s? 3d10 4p® 4d? а. b. x2:…
A: The cation is formed when any electron is removed from the neutral atom. The anion is formed when…
Q: 6. Calculate the E° at 25 °C for the following reaction: + H,0 CO, +2H +2e AGP (kJ/mol) -137.3…
A: ∆G° and E° are related as : ∆G° = - nFE° Here , n = number of electrons involved in reaction F =…
Q: Study the following phase diagram of Substance X. B E D' F G temperature (K) O A O B Which is the…
A: The phase diagram given is,
Q: Consider the combustion of compressed methane, and liquid hydrogen. Which one will release more…
A: A question based on chemical thermodynamics that is to be accomplished.
Q: Silver nitrate reacts with nickel to form Ni(NO3)2 and silver. To produce 0.7kg of silver, how many…
A:
Q: At a given temperature, osmotic pressure of a concentrated solution of a substance A. is lower than…
A: Equation for osmotic pressure ( π ) is, π = i × C × R × T i is the van't hoff factor, C is the…
Q: Muriatic acid is between 20-32% hydrochloric acid. A particular batch is determined to have a…
A:
Q: B. Calculating Average Molecular Mass 3. Diboron heptoxide – B2O7
A: Given compound ---> Diboron heptoxide = B2O7 Average molecular mass = ?
Q: Substance AH: Substance AH kJ mole mole |-189 216 206 |-177 |-229 IF B G D 61 Please calculate the…
A:
Q: statements is FALSE about alkanes and cycloalkanes? Choices: All alkanes produce CO2 and water…
A: Correct statements are All alkanes produce CO2 and water vapour upon complete combustion. Cyclic…
Q: What approximation is used to determine the rate law of linear chain reactions? Justify your answer.
A:
Q: Organic compounds that contain large proportions of nitrogen and oxygen tend to be unstable and are…
A:
Q: What is the importance of making sure that the correct process is done in the process of…
A: Because Two molecules of ATP are produced in fermentation if the process is not carried out properly…
Q: Which of the following compounds will result from the reaction between propene and HBr?…
A:
Q: NO2–1 (aq) + I -1 (aq)® NO (g) + I2(s) Oxidation numbers for every atom Oxidation and…
A: Some rules to be followed while calculating oxidation number: 1) Oxidation number of oxygen atom is…
Q: What is the vapor pressure at 25°C of a solution made by adding 50 mL of C3H80 to 500 mL of…
A: Given: Volume of C3H8O = 50 mL Volume of water = 500 mL Density of C3H8O = 1.26 g/mL. And the…
Q: A 0.25g of a certain substance is dissolved in 60g of water. The freezing point of the solution is -…
A: Here we are required to find the molar mass of the substance where we see depression of freezing…
Q: QUESTION 4 Explain the meaning of the term critical mass in relation to the use of a chain reaction…
A:
Q: Bleaching Chlorine atom to chloride anion Corrosion O O O
A: Answer - You have posted multipart of question as per the guidelines i have solved first three…
Step by step
Solved in 2 steps with 2 images

- I asked this question earlier but I think I forgot to include a conversion wht we know : mm of citrix acid = 192.14g/moldensity of citrix acid =166g/cm3 1 lime = 2tbsp of juice = 6 tsp 1tsp = 4.929x10-3L Q: assume citrix acid makes up 7.96% by volume of the lime juice . How many moles of citrix acid are in the recipe ( calls for 1 lime juice)Chemicals AmountBenzophenone 0.523 g (2.87*10^-3mol)Sodium borohydride 0.12 g (3.2*10^-3 mol)Benzhydrol 0.357 g (1.94*10^-3mol)Observed melting point 65-66 degree C Theroretical yield of Benzophenone ________ Percent yield _______________ Theoretical melting point __________--Cyclohexanol 10 ml Mass of cyclohexanol 9.62 g – 0.053 mol Theroretical yield cyclohexene–7.89g - ? mol Therotical mass of cyclohexene- Actual yield- 6.8ml –4.055 g –? mol Percent yield- 51.04% Boiling point of cyclohexene -83 degree Celsius Theroretical boling point -83 degree Celsius can i get some clarification pls
- does 50:50 hexane-toulene or 25:75 hexane-toulene have higher boiling point?A student measured the melting point range of an unknown solid to be 72.4 to 78.6 degrees celsius. a) Is the compound pure? b) Can you identify the compound from Table 2.2? c) If yes, what is the compound? If no, what compounds from Table 2.2 can be excluded? Table 2.2: Solid - Melting Point (Celsius) Benzoic Acid - 122 Acentanilide - 114 Acetamide - 82 Biphenyl - 71 p-Dimethoxybenzene - 59 p-Dichlorobenzene - 54 Benzophenone - 48The alcohol content of hard liquor is normally given in terms of the “proof,” which is defined as twice the percentage by volume of ethanol (C2H5OH) present. Calculate the number of grams of alcohol present in 1.00 L of 75-proof gin. The density of ethanol is 0.798 g/mL. Round off to 4 sig. figures for all calculations made prior to final answer and the final answer should be in 3 sig. figures
- 1. How to collect main batch of crystals using vacuum filtration (Set-up, things to remember)• Why use small amounts of cold water for washing? What is being washed away (2things)?2. Why measure the volume of mother liquor in a graduated cylinder?3. How to let the main batch of crystals dry/how will they be stored until next week?4. How to collect the small amount of crystals from step 4 (those that were not subjected tocrystallization)• What can we expect of this small batch of crystals compared to the main batch?What is the boiling point of the automobile radiator fluid prepared by mixing 1.04 L of ethylene glycol (HOCH2CH2OH, density = 1.114 g/mL) with 1.08 L of water (density = 1.000 g/mL)?The Kb of water is 0.520°C/m.Phys chem unit 4 . part 1 Kindly properly answer this? thank you so much :) 2. Will water inside a sealed spherical glass boil (see bubble formation?Explain.
- A student was trying to prepare acetaminophen in the laboratory. She found a melting point range of 169-173. The melting of acetaminophen is reported to be 168-172c. How could the student have verified that the produce is acetaminophen? Group of answer choices. Retake the sample melting point with more material in the sample. Ask the lab partner to measure the melting point of the sample. Take a mixed melting point with an authentic sample of acetaminophenA student measured the melting point range of an unknown solid to be 81.9 to 82.3 degrees celsius. a) Is the compound pure? b) Can you identify the compound from Table 2.2? c) If yes, what is the compound? Table 2.2: Solid - Melting Point (Celsius) Benzoic Acid - 122 Acentanilide - 114 Acetamide - 82 Biphenyl - 71 p-Dimethoxybenzene - 59 p-Dichlorobenzene - 54 Benzophenone - 48Properties of Liquids and IMF example andbrief discussion

