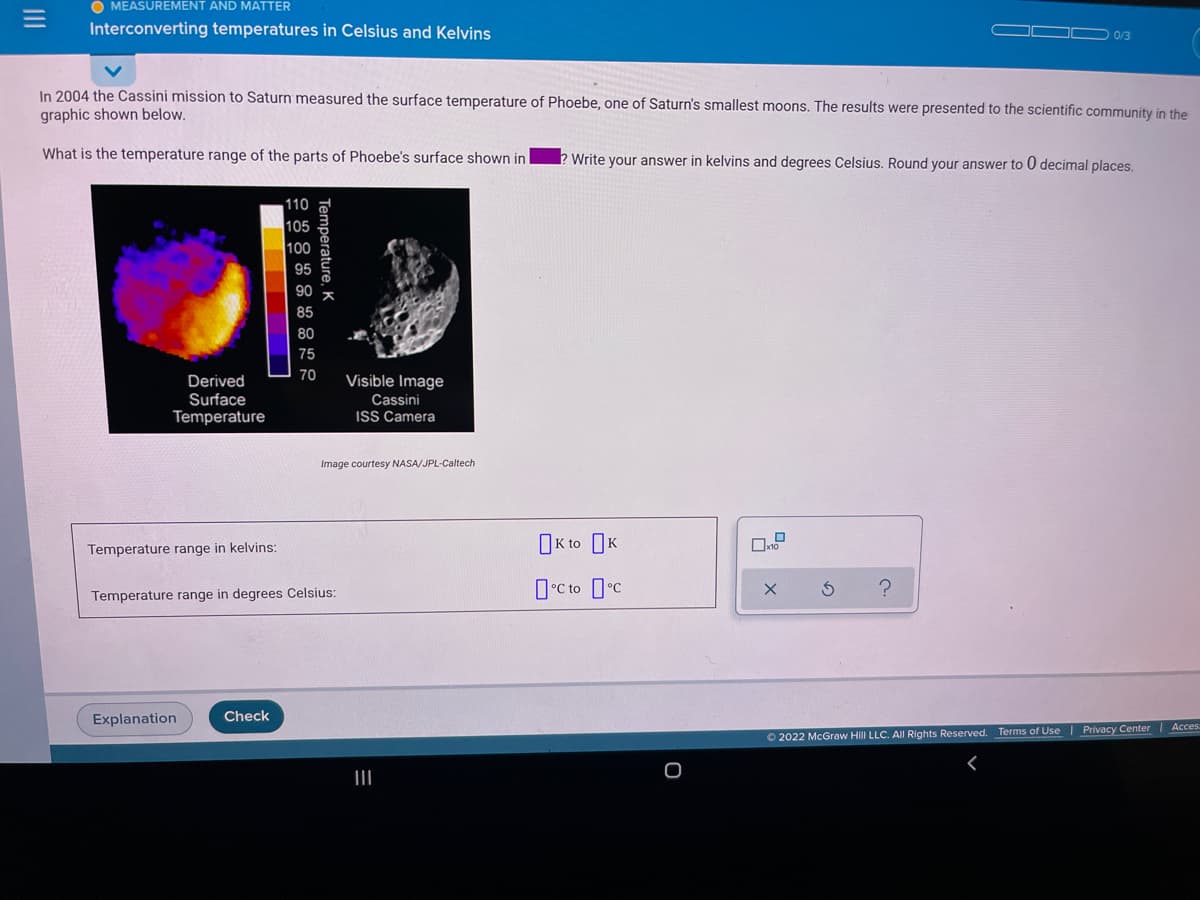
In 2004 the Cassini mission to Saturn measured the surface temperature of Phoebe, one of Saturn's smallest moons. The results were presented to the scientific community in the graphic shown below. What is the temperature range of the parts of Phoebe's surface shown in Write your answer in kelvins and degrees Celsius. Round your answer to 0 decimal places. 110 105 100 95 85 80 75 70 Derived Surface Temperature Visible Image Cassini ISS Camera Image courtesy NASA/JPL-Caltech OK to OK Temperature range in kelvins: O°C to c Temperature range in degrees Celsius: Temperature, K
In 2004 the Cassini mission to Saturn measured the surface temperature of Phoebe, one of Saturn's smallest moons. The results were presented to the scientific community in the graphic shown below. What is the temperature range of the parts of Phoebe's surface shown in Write your answer in kelvins and degrees Celsius. Round your answer to 0 decimal places. 110 105 100 95 85 80 75 70 Derived Surface Temperature Visible Image Cassini ISS Camera Image courtesy NASA/JPL-Caltech OK to OK Temperature range in kelvins: O°C to c Temperature range in degrees Celsius: Temperature, K
Chemistry: Principles and Reactions
8th Edition
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:William L. Masterton, Cecile N. Hurley
Chapter1: Matter And Measurements
Section: Chapter Questions
Problem 38QAP: A gasoline station in Manila, Philippines, charges 38.46 pesos per liter of unleaded gasoline at a...
Related questions
Question
Transcribed Image Text:O MEASUREMENT AND MATTER
Interconverting temperatures in Celsius and Kelvins
O 0/3
In 2004 the Cassini mission to Saturn measured the surface temperature of Phoebe, one of Saturn's smallest moons. The results were presented to the scientific community in the
graphic shown below.
What is the temperature range of the parts of Phoebe's surface shown in
Write your answer in kelvins and degrees Celsius. Round your answer to 0 decimal places.
110
105
100
95
90
85
80
75
70
Visible Image
Derived
Surface
Temperature
Cassini
ISS Camera
Image courtesy NASA/JPL-Caltech
OK to K
Temperature range in kelvins:
O°C to C
Temperature range in degrees Celsius:
Explanation
Check
O 2022 McGraw Hill LLC. All Rights Reserved.
Terms of Use Privacy Center | Acces
II
Temperature
II
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning


Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning