In a 6.00 L pressure cooker, water is brought to a boil. If the final temperature is 115 °C at 4.05 bar, how many moles of steam are in the cooker? (R = 0.08314 L-bar/mol.K) 1 4 7 +/- 2 3 5 mol 8 6 9 0 X x 100
In a 6.00 L pressure cooker, water is brought to a boil. If the final temperature is 115 °C at 4.05 bar, how many moles of steam are in the cooker? (R = 0.08314 L-bar/mol.K) 1 4 7 +/- 2 3 5 mol 8 6 9 0 X x 100
Introductory Chemistry: A Foundation
9th Edition
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Donald J. DeCoste
Chapter14: Liquids And Solids
Section: Chapter Questions
Problem 9ALQ: ook at Fig. 14.2. Why doesn't temperature increase continuously ever time? That is, why does the...
Related questions
Question
please help
Transcribed Image Text:Google Chrome isn't your default browser Set as default
Question 12 of 12
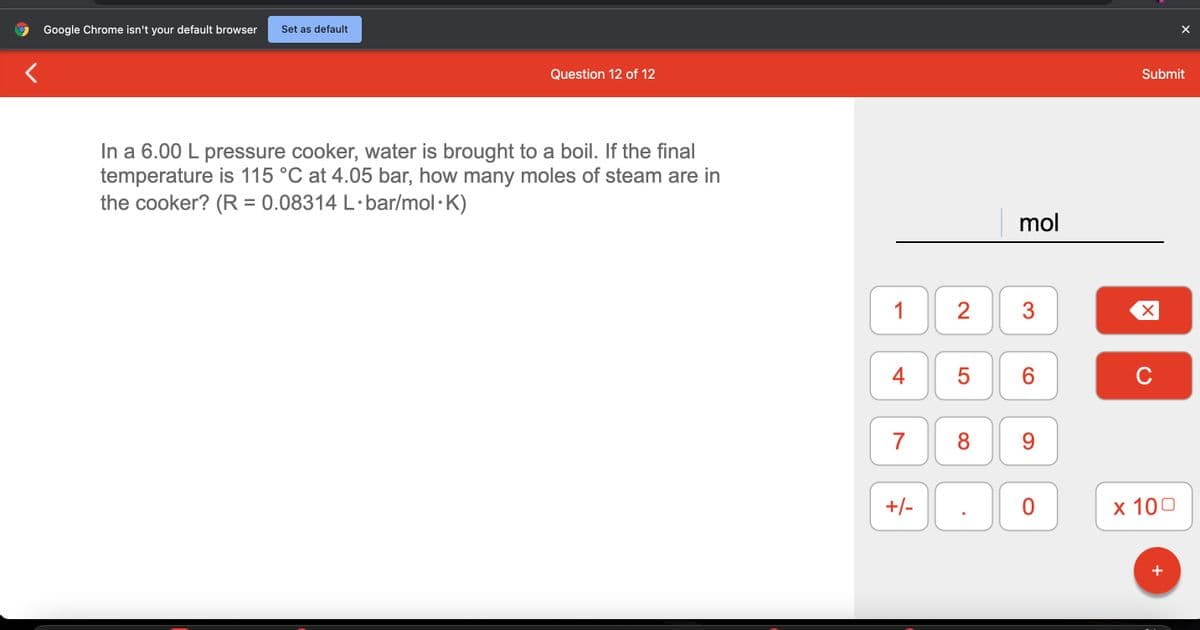
In a 6.00 L pressure cooker, water is brought to a boil. If the final
temperature is 115 °C at 4.05 bar, how many moles of steam are in
the cooker? (R = 0.08314 L·bar/mol.K)
1
4
7
+/-
2 3
5
| mol
8
6
9
0
Submit
X
C
x 100
+
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning


Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning