In a calorimetry experiment, two liquid substances X and Y with the same mass are placed in separate isolated containers. The specific heat of the substances are related by cx = 2cy. If the substances receive the same amount of heat, what will be the relationship between the change in temperature, ATx and ATy? Ο ΔΤΧ ATy Ο ΔΤΧ 4ΔΤΥ Ο ΔΤ 2ΔΤΥ 2ATY Ο 2ΔΤ ΔΤΥ
In a calorimetry experiment, two liquid substances X and Y with the same mass are placed in separate isolated containers. The specific heat of the substances are related by cx = 2cy. If the substances receive the same amount of heat, what will be the relationship between the change in temperature, ATx and ATy? Ο ΔΤΧ ATy Ο ΔΤΧ 4ΔΤΥ Ο ΔΤ 2ΔΤΥ 2ATY Ο 2ΔΤ ΔΤΥ
Chapter1: Temperature And Heat
Section: Chapter Questions
Problem 100P: One easy way to reduce heating (and cooling) costs is to add extra insulation in the attic of a...
Related questions
Question
please help i dont know the correct answer
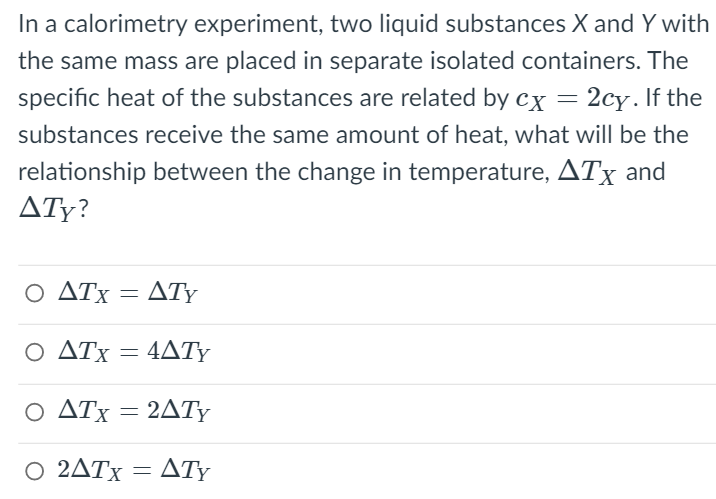
Transcribed Image Text:In a calorimetry experiment, two liquid substances X and Y with
the same mass are placed in separate isolated containers. The
specific heat of the substances are related by cx = 2cy. If the
substances receive the same amount of heat, what will be the
relationship between the change in temperature, ATx and
ATy?
Ο ΔΤΧ
ATy
Ο ΔΤΧ
4ΔΤΥ
Ο ΔΤ 2ΔΤΥ
2ATY
Ο 2ΔΤ ΔΤΥ
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you


College Physics
Physics
ISBN:
9781285737027
Author:
Raymond A. Serway, Chris Vuille
Publisher:
Cengage Learning

Physics for Scientists and Engineers: Foundations…
Physics
ISBN:
9781133939146
Author:
Katz, Debora M.
Publisher:
Cengage Learning


College Physics
Physics
ISBN:
9781285737027
Author:
Raymond A. Serway, Chris Vuille
Publisher:
Cengage Learning

Physics for Scientists and Engineers: Foundations…
Physics
ISBN:
9781133939146
Author:
Katz, Debora M.
Publisher:
Cengage Learning