Biochemistry
9th Edition
ISBN:9781305961135
Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougal
Publisher:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougal
Chapter6: The Behavior Of Proteins: Enzymes
Section: Chapter Questions
Problem 60RE
Related questions
Question
Just type in the answers. Thanks
![Biol 3800
Virt Hr 3 Worksheet
Dr. Song
In Michaelis menton kinetics which variable is held constant? multiselect
a. substrate concentration
b.
enzyme concentration
c. reaction time
Which of the following binds to the active site multiselect
a. competitive inhibitor
b. non-competitive inhibitor
c. mixed inhibitor
An enzyme with a high/strong affinity for substrate has multiselect
a. low Km
b. high Vmax
c. x intercept close to the origin
d. y intercept close to the origin
The addition of an enzyme will make a non-spontaneous reaction spontaneous multiselect-just kidding
True or False
The addition of an enzyme can make a non-spontaneous reaction go
True of False
where would a second molecule be added during nucleic acid elongation (ie., syn-
Nucleotide structure
thesis)
NH2
Phosphate
Base
O=P-0-CH2
H
H
H
ОН Н
Sugar
In the reversible reaction A -
- B;
Keq (equilibrium constant) is
a. [B]/[A]
b. [A]/[B]
c. equal to 1
d. the difference between A and B
CS Scanned with CamScanner](/v2/_next/image?url=https%3A%2F%2Fcontent.bartleby.com%2Fqna-images%2Fquestion%2F51766312-77d6-4388-ba53-71601d5219eb%2F8b8311e7-6b5d-4ce7-a9eb-a262dd39270f%2Fixh8lxf_processed.jpeg&w=3840&q=75)
Transcribed Image Text:Biol 3800
Virt Hr 3 Worksheet
Dr. Song
In Michaelis menton kinetics which variable is held constant? multiselect
a. substrate concentration
b.
enzyme concentration
c. reaction time
Which of the following binds to the active site multiselect
a. competitive inhibitor
b. non-competitive inhibitor
c. mixed inhibitor
An enzyme with a high/strong affinity for substrate has multiselect
a. low Km
b. high Vmax
c. x intercept close to the origin
d. y intercept close to the origin
The addition of an enzyme will make a non-spontaneous reaction spontaneous multiselect-just kidding
True or False
The addition of an enzyme can make a non-spontaneous reaction go
True of False
where would a second molecule be added during nucleic acid elongation (ie., syn-
Nucleotide structure
thesis)
NH2
Phosphate
Base
O=P-0-CH2
H
H
H
ОН Н
Sugar
In the reversible reaction A -
- B;
Keq (equilibrium constant) is
a. [B]/[A]
b. [A]/[B]
c. equal to 1
d. the difference between A and B
CS Scanned with CamScanner
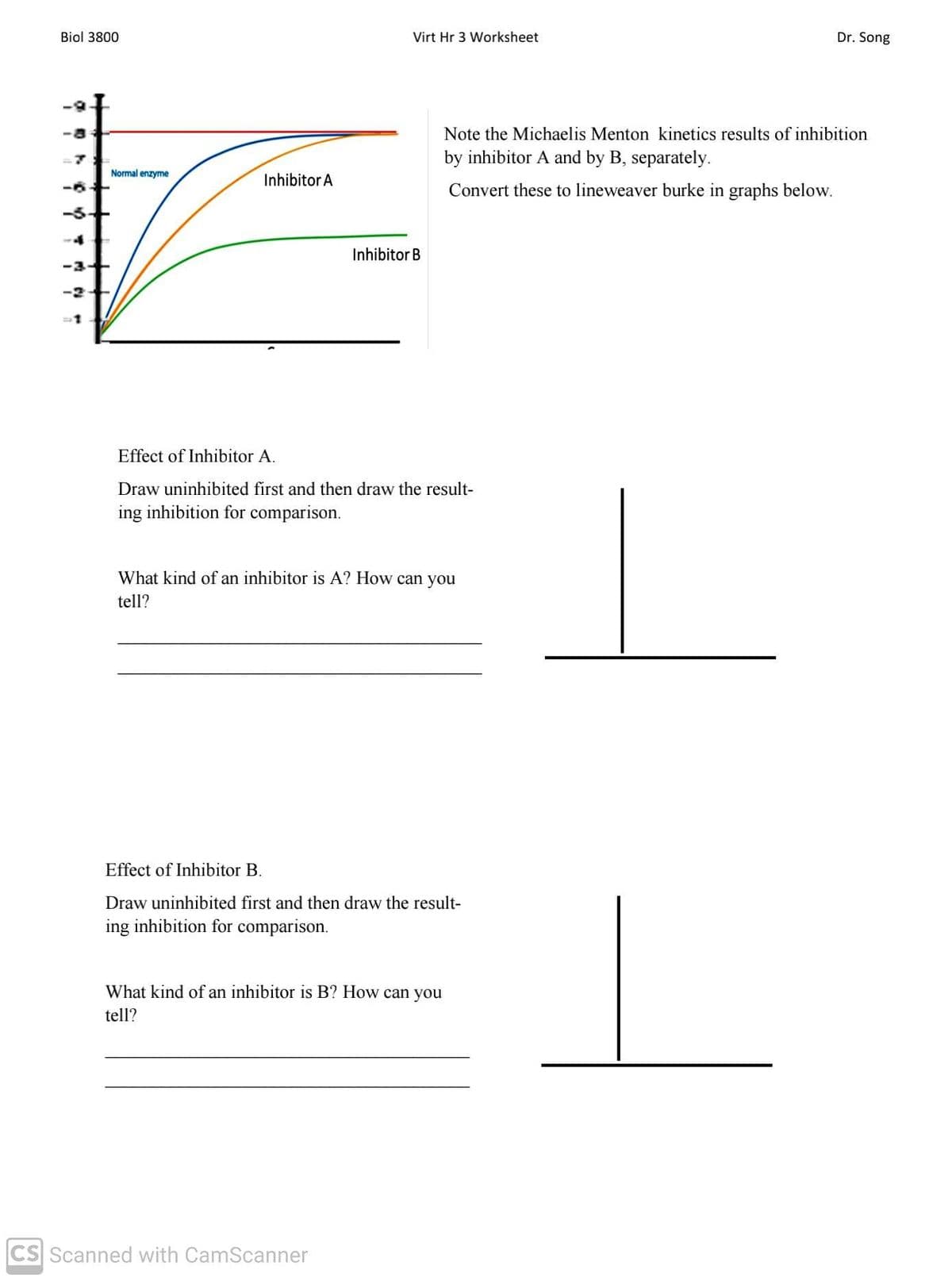
Transcribed Image Text:Biol 3800
Virt Hr 3 Worksheet
Dr. Song
Note the Michaelis Menton kinetics results of inhibition
by inhibitor A and by B, separately.
Normal enzyme
Inhibitor A
Convert these to lineweaver burke in graphs below.
-5-+
Inhibitor B
-3+
-2+
Effect of Inhibitor A.
Draw uninhibited first and then draw the result-
ing inhibition for comparison.
What kind of an inhibitor is A? How can you
tell?
Effect of Inhibitor B.
Draw uninhibited first and then draw the result-
ing inhibition for comparison.
What kind of an inhibitor is B? How can you
tell?
CS Scanned with CamScanner
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Recommended textbooks for you

Biochemistry
Biochemistry
ISBN:
9781305961135
Author:
Mary K. Campbell, Shawn O. Farrell, Owen M. McDougal
Publisher:
Cengage Learning

Biochemistry
Biochemistry
ISBN:
9781305961135
Author:
Mary K. Campbell, Shawn O. Farrell, Owen M. McDougal
Publisher:
Cengage Learning