H20 f) OH Chemistry Steps Chemistry Stes Br Chemistry Steps Chemistry Steps Chemistry Steps Br h) Chemistry Steps Chemistry Steps Chemistry Steps Br i)
H20 f) OH Chemistry Steps Chemistry Stes Br Chemistry Steps Chemistry Steps Chemistry Steps Br h) Chemistry Steps Chemistry Steps Chemistry Steps Br i)
Biology (MindTap Course List)
11th Edition
ISBN:9781337392938
Author:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. Berg
Publisher:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. Berg
Chapter2: Atoms And Molecules: The Chemical Basis Of Life
Section2.2: Chemical Reactions
Problem 1C: What enables a radioisotope to substitute for an ordinary (nonradioactive) atom of the same element...
Related questions
Question
Predict the mechanism as SN1, SN2, E1 or E2 and draw the major organic product formed in each reaction. Consider any regioselectivity and stereoselectivity where applicable:
I’d be grateful if you helped with all of these but if not can you please help me answer f,g,h & j ?
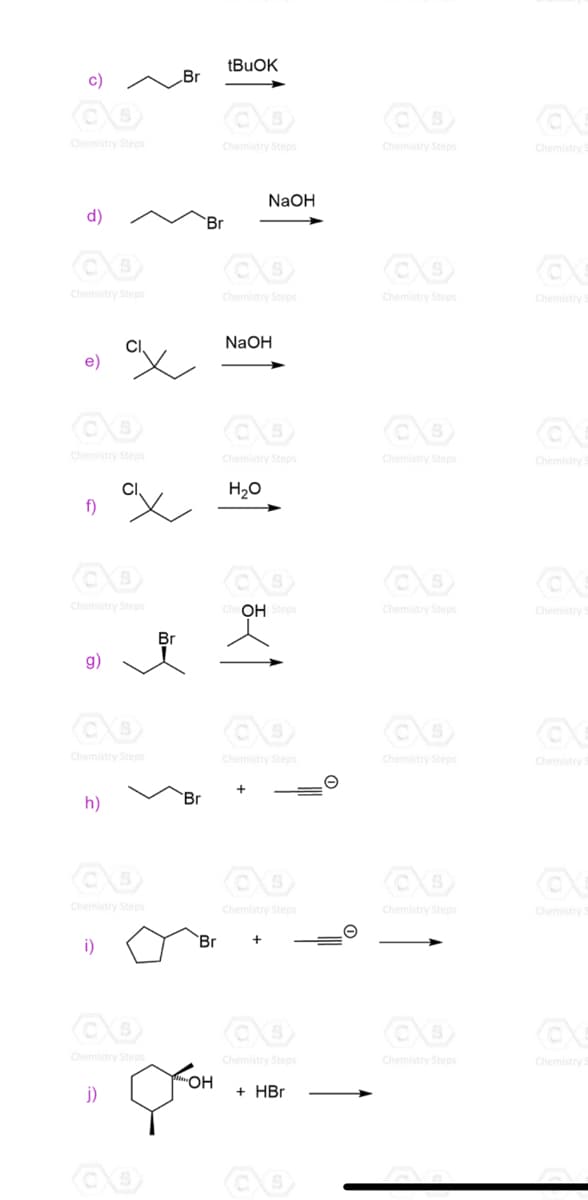
Transcribed Image Text:c)
Br
Chemistry Steps
Chemistry Steps
Chemistry Steps
Chemistry
NaOH
d)
'Br
Chemistry Steps
Chemistry Steps
Chemistry Steps
Chemistry
NAOH
Chemistry Steps
Chemistry Steps
Chemistry Steps
Chemistry
H20
f)
Chemistry Steps
OH Steps
Chemistry Steps
Chemistry
Br
g)
Chemistry Steps
Chemistry Steps
Chemistry Steps
Chemistry
Br
h)
Chemistry Steps
Chemistry Steps
Chemistry Steps
Chemistry
i)
`Br
Chemistry Steps
Chemistry Steps
Chemistry
Chemistry Steps
ОН
j)
+ HBr
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you

Biology (MindTap Course List)
Biology
ISBN:
9781337392938
Author:
Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. Berg
Publisher:
Cengage Learning

Human Biology (MindTap Course List)
Biology
ISBN:
9781305112100
Author:
Cecie Starr, Beverly McMillan
Publisher:
Cengage Learning

Biology (MindTap Course List)
Biology
ISBN:
9781337392938
Author:
Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. Berg
Publisher:
Cengage Learning

Human Biology (MindTap Course List)
Biology
ISBN:
9781305112100
Author:
Cecie Starr, Beverly McMillan
Publisher:
Cengage Learning