Q: of (a) vs. (b) vs. H
A:
Q: B. CIRCLE (CROSS-OUT) the FASTEST (SLOWEST) Nucleophilic Substitution Br + H3C-O + H3C-O .CI
A: Rate of Nucleophilic substitution depends on the nature of leaving group. Better the leaving group…
Q: Draw a detailed step-wise mechanism for the following reactions. Be sure to show all steps, formal…
A:
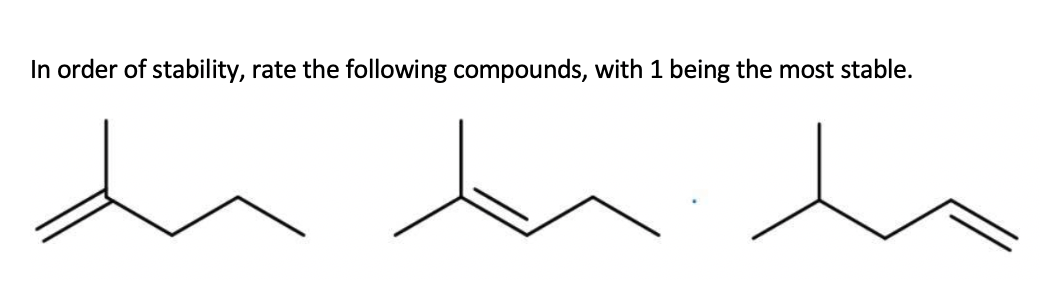
Q: Rank the following compounds from most to least reactive in an SN2 reaction
A:
Q: 1. In following pairs of nucleophilic substitution reaction, which one have higher reaction rate?…
A: Note: According to our guidelines we are supposed to answer only first three subpart. Kindly repost…
Q: c) What is the "order of stability"(most to least) of carbocations? Use the following list. methyl…
A: Tertiary Benzyl > Secondary Benzyl > Primary Benzyl > Secondary Diallyl > Primary Allyl…
Q: Draw curved arrows for the following reaction step. Arrow-pushing Instructions CH3 CH3 H-0-H H3C-C+…
A: This is SN1 reaction When tertiary butyl chloride reacts with water forms carbocation and gives…
Q: c) Circle the most electrophilic carbon in the structure below and explain your choice.
A:
Q: Follow the curved arrows and draw the products of the following reaction. Include all lone pairs and…
A:
Q: By following the curved red arrows, draw the product(s) of each of the following reaction steps.…
A: a. In accordance with the movement of electrons in the above reaction, bromine atom was detached…
Q: 2-iodopentane
A:
Q: 1. Show how to carry out the following transformation. N ? N.
A: Note: Bromination of benzene. Acetylation of bromo benzene. Azo coupling. Sonagashira coupling.…
Q: Rank the following substrates in order from slowest SN1 reaction rate to fastest. Br Br Br Br A В
A: The stability of carbocation is given as substituted with Hetero atom > Tertiary > Secondary…
Q: The rate of bromination of the following three alkenes is very different: 2-methyl-1-pentene,…
A: Bromination of alkene or alkene halogenation reaction means addition of dihalide on the…
Q: 4. Propose a reasonable mechanism, using curved arrow notation, for the following reaction. Be sure…
A:
Q: Show how a 1,2-shift forms a more stable carbocation from attached intermediate.
A:
Q: Draw a step-wise mechanism for each of the following reactions. Show all arrows, proton transfers,…
A:
Q: y12. Which reaction in each of the following pairs would you anticipate being faster?
A: We have given that Which reaction in each of the following pairs would you anticipate being faster?…
Q: Which of the following is expected to be a major product of the reaction shown in the box? Br, or…
A:
Q: [H] N-H
A:
Q: Rank the attached carbocations in order of increasing stability?
A: The order of stability of carbocations is: Tertiary carbocation > secondary carbocation >…
Q: 3. Show the series of step(s) to perform the following transformations. а.
A:
Q: Starting from intermediate X, complete the mechanism. Provide curved arrows for both steps and draw…
A: The carbocation formed in the intermediate X is unstable because it is present at bridge head…
Q: For each pair of aromatic compounds, determine which will undergo electrophilic aromatic…
A: Since your question has multiple sub-parts, we will solve first three sub-parts for you. If you want…
Q: ) Rank the following compounds in order of their reaction rates in an Sn1 reaction with NaF with 1…
A:
Q: whats the product name from reaction below?
A: This is a question from organic reaction.
Q: (i) which reaction among the pair, is fastest? Explain your (ii) predict the products of faster…
A:
Q: Rank the following radicals in order of decreasing stability. most stable least stable (CHдС-снсн,…
A: Free radicals are electron deficeint. the stability of tertiary radicals is more than that of…
Q: i) (CH3)3N: (CH3)2ö: W ii)
A:
Q: Rate the below nucleophiles in terms of INCREASING nucleophilicity. Explain why. CH3OH CH;0 t-BuO-…
A: As the name suggests, nucleophiles are those reagents that possess attraction for the positively…
Q: Given the compounds below, which is the more stable one?
A:
Q: Circle the better nucleophile in polar protic solvent. Below each answer, BRIEFLY explain your…
A: Polar protic solvents are capable of forming hydrogen bonding because they have atleast one hydrogen…
Q: which compound would form the most stable carbocation, either initially or after rearrangement…
A: Note: Allylic carbocations are more stabilized than secondary carbocations due to allylic…
Q: 2) Follow the flow of electrons indicated by the curved arroWs in the reaction and predict the…
A:
Q: Using Curved Arrow Formalism, draw the reaction of 1-methylcyclopentanol treated with aqueous…
A:
Q: Determine the position that is most likely to be the site of an EAS reaction in the compound below:…
A:
Q: The electrophilic addition of Br, to several alkenes was examined. Explain why the relative reaction…
A: In the reaction of Br2 with an alkene, an alkene is a nucleophile and Br is an electrophile.
Q: Copy the given reaction and show the mechanism by supplying the appropriate curved arrows. Also,…
A:
Q: Question is attached
A: The structure of 1-butene is drawn as, It is an isomer of butene.
Q: Rank the following radicals in order of increasing stability.
A: Since the stability of free radicals depends on two things 1) resonance => more is the resonance…
Q: What happens to the rate of an SN2 reaction under the following conditions? [RX] is halved, and…
A: Nucleophilic substitution bimolecular reaction or the SN2 is a type of reaction in which bonds are…
Q: For the following intermediates , please draw as many resonance contributors that you can come up…
A: Resonance structures are the canonical structures or two or more Lewis structures that describe the…
Q: lect which molecule is the better nucleophile in e following pair: Br- or Cl- in H20 СНЗО-or CНЗОН…
A:
Q: In the given condensed formula, I understand that there will be a carbocation rearrangement due to…
A: The E1 mechanism of conversion of secondary alcohol to alkene is given in step two.
Q: For each pair of compounds determine which will undergo electrophilic substitution faster. (a) (b)…
A: As there are more than three parts of this question, we have solved the first three parts for you,…
Q: The free-radical bromination of the following compound introduces bromine primarily at the benzylic…
A: Free radical bromination occurs exclusively at the benzylic position. The benzylic position is next…
Q: CH3 CH, H3C-¢* + :Br: H,C Br : CH3 CH,
A: The movement of electrons are shown by curved arrows. There are two types of arrows to show the…
Q: How do I rank these from best to poorest leaving group? Do I base it in the size of the…
A: We have to rank leaving group tendency :-
Q: On the left-hand (reactants) side of the reaction, provide the curved arrows (using the formalism…
A:
Q: Rank the following radicals in order of increasing stability.
A: The stability of free radical is governed by various factors such as: Allylic / benzylic radical 3°…
Step by step
Solved in 2 steps with 2 images


