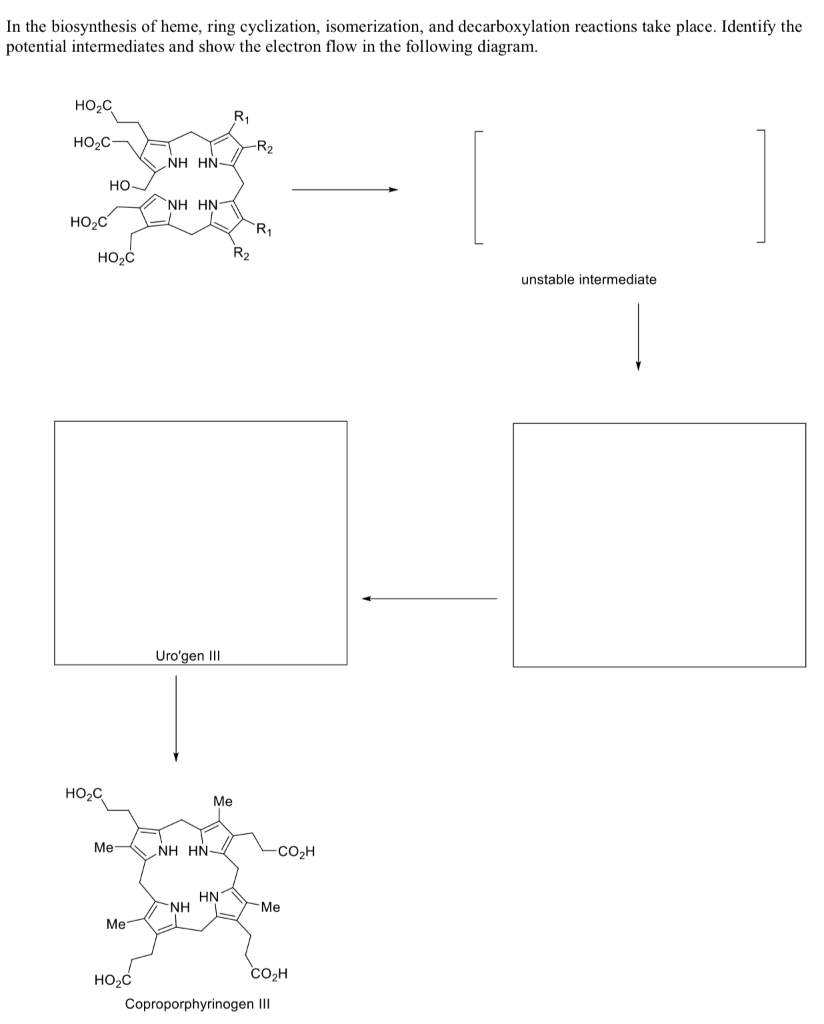
In the biosynthesis of heme, ring cyclization, isomerization, and decarboxylation reactions take place. Identify the potential intermediates and show the electron flow in the following diagram.
Q: Please explain what happened in the reaction. How did H2SO4 and 3H2O reacted with the glucose?
A: Carbohydrates are polyhydroxy aldehydes or ketones or compounds that yield them on hydrolysis.…
Q: Explain the chemical logic of dehydrating 2-phosphoglycerate before its phosphoryl group is…
A: 2-Phosphoglyceric acid , or 2-phosphoglycerate, is a glyceric acid which serves as the substrate in…
Q: Order the following compounds according to the glycolytic pathway: C D CHOPO Oro -o-ro HO OH HCOH ON…
A: Glycolysis is the biochemical process that occur in both aerobic and anaerobic organisms. In…
Q: Write the correct order of the following structures: To select the correct order start from the…
A: Shikimic acid or also known as shikimate is a cyclohexanecarboxylic acid. It is an important…
Q: charge on oxygen
A: the answer of the quastion is option 4. that is stabilize the charge on oxygen. its a process of…
Q: Write balanced equations for each of the reactions of thecitric acid cycle.
A: The citric acid cycle or the Kreb's cycle or the TCA cycle is the series of eight-step reactions…
Q: h. Give an outline of one example of an anaplerotic reaction.
A: Introduction Anaplerotic reactions are metabolic mechanisms that restore oxaloacetate after it has…
Q: Why would ether linkages be more stable in membranes than ester lipids? How would the presence of…
A: Archaea are unique in the lipid composition of the cell membrane.
Q: b) The above molecule may be further converted into a polymer with similar molecular structure to…
A: The molecule given in the question is named the molecule" 5-hydroxymethyl-2-furaldehyde or…
Q: In generating CO2, how can the citric acid cycle discriminate between the carbons in citrate given…
A: In the first phase of the cycle, acetyl CoA combines with oxaloacetate, a four-carbon acceptor…
Q: structure of ATP and label the phosphate ester linkage, the phosphate anhydride linkage, the…
A: ATP is the energy currency of the cell. It is a ribonucleotide which has Adenine as nitrogenous…
Q: H. C=0+NADH + H* H-C-OH + NAD+ CH3 Acetaldehyde CH3 Ethanol 7. Using the reaction above, describe…
A: Fermentation is a type of anaerobic respiration in which Glucose is converted into the ethyl…
Q: Write the structures for the molecule that best fits the answer to the following: d) the final…
A: Pyruvate is the end product of glycolysis, under aerobic conditions it is oxidized via citric acid…
Q: Consider the dextran sucrase reaction. Why do you suppose there is not an ATP requirement to…
A: Dextran sucrase is an enzyme that catalyses the chemical reaction.
Q: What metabolic strategies are employed to oxidize a saturated carbon? Dehydrogenation/elimination…
A: Saturated Carbon :- As carbon have 4 electron to share in its outermost shell so carbon form bonds…
Q: Consider MYRISTIC ACID CH3-(CH2)12 - COOH C. How many cycles of ß- oxidation are needed for complete…
A: It is given that, Myristic acid CH3 - (CH2)12 - COOH. How many cycles of beta-oxidation are needed…
Q: If you want to produce a sweet-tasting wine with low alcohol content, how will you attempt to…
A: Production of wines starts with harvesting of grapes at the right moment. Picking of the grapes…
Q: Write a balanced equation for the reaction catalyzed bynitrogenase.
A: Enzymes are biological catalysts that help in catalyzing or speeding up biological reactions by…
Q: Show, by a series of equations, the energetics of phosphorylation of ADP by phosphoenolpyruvate.
A: ATP is synthesized during the conversion of phosphoenolpyruvate to pyruvate. This is also known as…
Q: One of the following is most accurate about relative redox potential of different electron carriers.…
A: Electron carriers are molecule that is capable of accepting one or more electrons (acceptor) from…
Q: The fact that acid-anhydride bonds are less stable than ester bonds helps explain the fact that the…
A: Acid-anhydride bonds are less stable and more reactive than ester bonds due to the presence of the…
Q: I’m not sure which answer is true for all that answer this question can you please help?
A: ATP is called the energy currency of the cell. ATP's full form is adenosine 5' triphosphates. ATP…
Q: Suppose ribulose-5-phosphate, labeled with 14C in carbon 1, is used as the substrate in carbon…
A: The Calvin cycle also called as C3 cycle is the chemical reaction cycle that plants undergo to "fix"…
Q: What is a Hyrogel? Please design a hydrogel that photochemically cross-linked (explain everything…
A: Hydrogel is a 3D crosslinked, water loving (hydrophilic) polymer that doesnot dissolve in water.…
Q: In the Krebs Cycle (Citric Acid Cycle), A 4-carbon compound with a 2-carbon unit to yield a 6-carbon…
A: Kreb cycle/ tricarboxylic acid cycle/ citric acid cylce - occur in matrix of mitochondria.
Q: If you want to produce a sweet-tasting wine with low alcohol content, explain how you might attempt…
A: Production of wines starts with harvesting of grapes at the right moment. Picking of the grapes…
Q: The enzyme prenyl transferase is responsible for condensing isopentenyl pyrophosphate units during…
A: Prenyl transferases These are referred as class of enzymes that are involved in transfering allylic…
Q: Suppose ribulose-5-phosphate, labeled with 14C in carbon 1, is used as the substrate in dark…
A: Calvin cycle or the light-independent or dark reaction, the second stage of the photosynthesis…
Q: Write chemical equation intramolecular dezamination Serineу. Specify enzymes and co-enzymes for…
A: Amino acids Proteins are the polymers of nitrogenous compounds called amino acids. Each amino acid…
Q: Glucose fermentation in yeast produces ethanol as a wasteproduct. In contrast, Clostridium…
A: Fermentation is the process that produces organic compounds by a series of reactions with the use of…
Q: Write a balanced equation for the oxidation of ubiquinol by cytochrome c. Calculate ΔG°′ and Δℰ°′…
A: Ubiquinol and cytochrome c are the components involved in the electron transport chain in…
Q: Suppose ribulose-5-phosphate, labeled with "C in carbon 1, is used as the substrate in dark…
A: The dark reaction is the second stage of photosynthesis, during which the ATP and NADH are used to…
Q: Part A N- N. Which of the following is found in the coenzyme FAD? IN Check all that apply. two…
A: FAD { Flavin adenine dinucleotide } is a coenzyme that consists of flavin protein and involved redox…
Q: the RHR, OHR, and the balanced overall reaction of the voltaic cell
A:
Q: Plants convert carbon dioxide and water to glucose 1C6H12O62 and oxygen in the process of…
A: photosynthesis is the process of conversion of light energy into chemical energy in the presence of…
Q: Classification of flavonoids is based on: Select one: a. The oxidation state of the heterocyclic…
A: "Flavonoids" are very common polyphenols in the individual diet, accounting for almost two-thirds of…
Q: Draw the structure before the product of glucose react further, how does oxidation occur in glucose?…
A: Due to the conversion and oxidation of the C1 aldehyde group to the carboxyl functional group, the…
Q: Write the correct order of the following structures according to their biosynthetic pathway between…
A: Biosynthesis is a multi-step, enzyme-catalyzed mechanism in which living organisms transform simple…
Q: Arrange the following order of chemical events of how ribulose reacts in the light independent…
A: Photosynthesis is the process by which plants produce oxygen and prepare their own food by using…
Q: Describe each step of the metabolic pathway shown in the image below. Linoleic Acid Metabolism…
A: Arachidonic acid is a twenty-carbon chain long fatty acid with four unsaturations, while…
Q: a) 2-Hydroxy-carboxylic acids in the citric acid cycle and in glycolysis (3 answers): b) how do the…
A: Note : Hi ! Thank you for the question. We are authorized to answer one question at a time. Since…
Q: Write balanced equations for the three known reactions that transfer an amino group to a substrate…
A: Amino group : It are compounds and functional groups that contain a basic nitrogen atom with a lone…
Q: Write a series of balanced equations and a summary equation for the reactions of the glucose–alanine…
A: Glucose-alanine cycle: It is the series of reaction in which amino groups and carbons from muscle…
Q: Write balanced equations for the three known reactions that transfer anamino group to a substrate by…
A: IMP (Inosine monophosphate) synthesis (purine synthesis): - the 7th step in this process include…
Q: If a biochemist is trying to synthesis a 14C Valine at the a-Carbon using Pyruvate as a starting…
A: Pyruvate is a byproduct of glycolysis and L-valine is non-polar amino acids with side chain group of…
Q: Consider the reaction: malate + NAD+ → oxaloacetate + NADH + H+. Calculate ΔE°’ for the…
A: The potential/capacity of a molecule to gain an electron is known as its reduction…
Q: Write balanced equations for the three known reactions that transfer an amino group to a substrate…
A: Amino group : It are compounds and functional groups that contain a basic nitrogen atom with a lone…
Q: Specify the type of carbon unit that is transferred in one-carbon metabolism by each of the…
A: Note: Since you have posted a question with multiple subparts, we will solve the first three…
Step by step
Solved in 4 steps with 3 images

- :Write the correct order of the following structures according to their biosynthetic pathway between A and B : A B shikmic acid 1 Flavonoids 2 prephenic acid 3 Photosynthesis 4 Phenylalanine 5 Co2 6 phenylpyruvic acid 7 erthrose-4-phosphate 8 corismic acid 9 3-dehydroqunic acid 10Draw the structure of the products formed when Arabinose shown above undergoes the following reaction: Tollen's reagent (include side product) Benedict's reagent (include side product) Oxidizing enzyme to produce alduronic acid Reducing agent like NaBH4 with Ethylamine (CH3CH2NH2) do everything for upvote. no upvote if incompleteguanosine and a water bind in the active site, does not matter in which order. The scissile N-1 glycosidic bond between the anomeric carbon and N9 of the base is broken resulting in an oxocarbenium transition state. The leaving group electrons are not protonated, but instead swing up to form a double bond between N9 and C8. The double bond that is already present on the purine swings out on N7 and is protonated by an active site histidine acting as a general acid. An active site aspartate acts as a general base, and deprotonates the water substrate molecule. The deprotonated water is then free to attack the transition state in a manner that will generate an αribose. Draw the active site and show how the mechanism works. You will need to draw 3 or 4 different active site “snap-shots” to show the entire process. Be sure to clearly show the transition state and how it might be stabilized
- The enzyme prenyl transferase is responsible for condensing isopentenyl pyrophosphate units during the second stage of cholesterol biogenesis. Which of the following intermediates could still form if prenyl transferase were inhibited? (A) farnesyl pyrophosphate(B) dimethylallyl pyrophosphate (C) squalene(D) geranyl pyrophosphate(E) lanosterolThe photosynthetic process used by the green sulfur bacteria, under anaerobic conditions, is best represented by which of the following balanced equations? cell bio 6CO2 + 12H2S C6H12O6 + 6S2 + 6H2O C6H12O6 + C6H12O6 C12H22O11 + H2O 6CO2 + 12H2O C6H12O6 + 6O2 + 6H2O C12H22O11 + H2O C6H12O6 + C6H12O6 C6H12O6 + 6O2 + 6H2O 6CO2 + 12H2OWhich of the following statement is CORRECT? A Hydrolysis of starch into glucose in an elimination reaction B Group transfer reaction is a nucleophilic substitution reaction C Conversion of 3-phosphoglycerate to 2-phosphoglycerate is a group transfer reaction D Most biochemical reaction occurs via homolytic cleavage E Electrophiles are electron-rich, while nucleophiles are electron-deficient
- Suppose ribulose-5-phosphate, labeled with 14C in carbon 1, is used as the substrate in carbon reactions. In which carbon of 3PG will the label appear?The photosynthetic process used by the green sulfur bacteria, under anaerobic conditions, is best represented by which of the following balanced equations? 6CO2 + 12H2O C6H12O6 + 6O2 + 6H2O 6CO2 + 12H2S C6H12O6 + 6S2 + 6H2O C12H22O11 + H2O C6H12O6 + C6H12O6 C6H12O6 + C6H12O6 C12H22O11 + H2O C6H12O6 + 6O2 + 6H2O 6CO2 + 12H2OWould you expect DCPIP to become colorless if your chloroplast suspension is in the presence of the herbicide dichlorophenyldimethyl urea (DCMU)? Explain your Is the presence of light a direct requirement for starch synthesis? Why or why not?
- If photosynthesizing green algae are provided with CO2 containing heavy oxygen (18O), later analysis will show that all of the following molecules produced by the algae contain 18O EXCEPT a) ribulose bisphosphate (RuBP) b) glyceraldehyde 3-phosphate (G3P) c)O2 d)3-phosphoglycerate e)glucoseIf a biochemist is trying to synthesis a 14C Valine at the a-Carbon using Pyruvate as a starting material, the Pyruvate must be labeled with 14C at the Carbonyl carbon. True or False. The photosynthetic process used by the green sulfur bacteria, under anaerobic conditions, is best represented by which of the following balanced equations? 6CO2 + 12H2S g C6H12O6 + 6S2 + 6H2O C6H12O6 + C6H12O6 g C12H22O11 + H2O 6CO2 + 12H2O g C6H12O6 + 6O2 + 6H2O C12H22O11 + H2O g C6H12O6 + C6H12O6 C6H12O6 + 6O2 + 6H2O g 6CO2 + 12H2O



