In the discussion of poly(ether ether ketone), or PEEK, in Section 26.6a.2, we observed that the addition of a tert-butyl group to the aromatic ring increases the solubility of a PEEK in organic solvents. However, when researchers added a second tert-butyl group, as shown here, they found that the solubility of the PEEK decreased. Explain this observation. A PEEK with two tert-butyl groups per repeating unit
In the discussion of poly(ether ether ketone), or PEEK, in Section 26.6a.2, we observed that the addition of a tert-butyl group to the aromatic ring increases the solubility of a PEEK in organic solvents. However, when researchers added a second tert-butyl group, as shown here, they found that the solubility of the PEEK decreased. Explain this observation. A PEEK with two tert-butyl groups per repeating unit
Organic Chemistry
8th Edition
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Chapter23: Amines
Section23.5: Basicity
Problem EQ
Related questions
Question
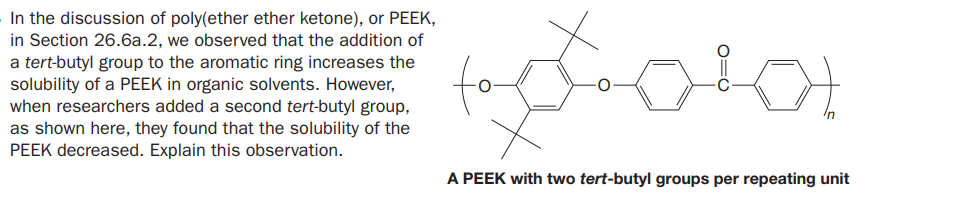
Transcribed Image Text:In the discussion of poly(ether ether ketone), or PEEK,
in Section 26.6a.2, we observed that the addition of
a tert-butyl group to the aromatic ring increases the
solubility of a PEEK in organic solvents. However,
when researchers added a second tert-butyl group,
as shown here, they found that the solubility of the
PEEK decreased. Explain this observation.
A PEEK with two tert-butyl groups per repeating unit
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning