In the following equilibrium, when Fe *3 ion is added, the concentration of Fe(SCN) +2 ion will Fe +3 + SCN +2 2 Fe(SCN) O a. Increase O b. Decrease Oc. Remain the same QUESTION 3 A 0.50 M solution of acetic acid, HC 2H 30 2, is 0.60% ionized. The ionization constant for acetic acid is O a. 7.2 X 10-3 O b. 1.8 X 10-3 O c. 1.2 X 10-2 O d. 1.8 x 10-5 QUESTION 4 The K of silver iodide is 8.3 X 10 17. What is the solubility of silver iodide in g/L? sp O a. 2.1 X 10-6 O b. 1.6 X 10-30 OC. 1.9 X10-14 O d. 6.1 X 10-21
In the following equilibrium, when Fe *3 ion is added, the concentration of Fe(SCN) +2 ion will Fe +3 + SCN +2 2 Fe(SCN) O a. Increase O b. Decrease Oc. Remain the same QUESTION 3 A 0.50 M solution of acetic acid, HC 2H 30 2, is 0.60% ionized. The ionization constant for acetic acid is O a. 7.2 X 10-3 O b. 1.8 X 10-3 O c. 1.2 X 10-2 O d. 1.8 x 10-5 QUESTION 4 The K of silver iodide is 8.3 X 10 17. What is the solubility of silver iodide in g/L? sp O a. 2.1 X 10-6 O b. 1.6 X 10-30 OC. 1.9 X10-14 O d. 6.1 X 10-21
Chemistry by OpenStax (2015-05-04)
1st Edition
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Chapter14: Acid-base Equilibria
Section: Chapter Questions
Problem 47E: Explain why equilibrium calculations are not necessary to determine ionic concentrations in...
Related questions
Question
Please answer all
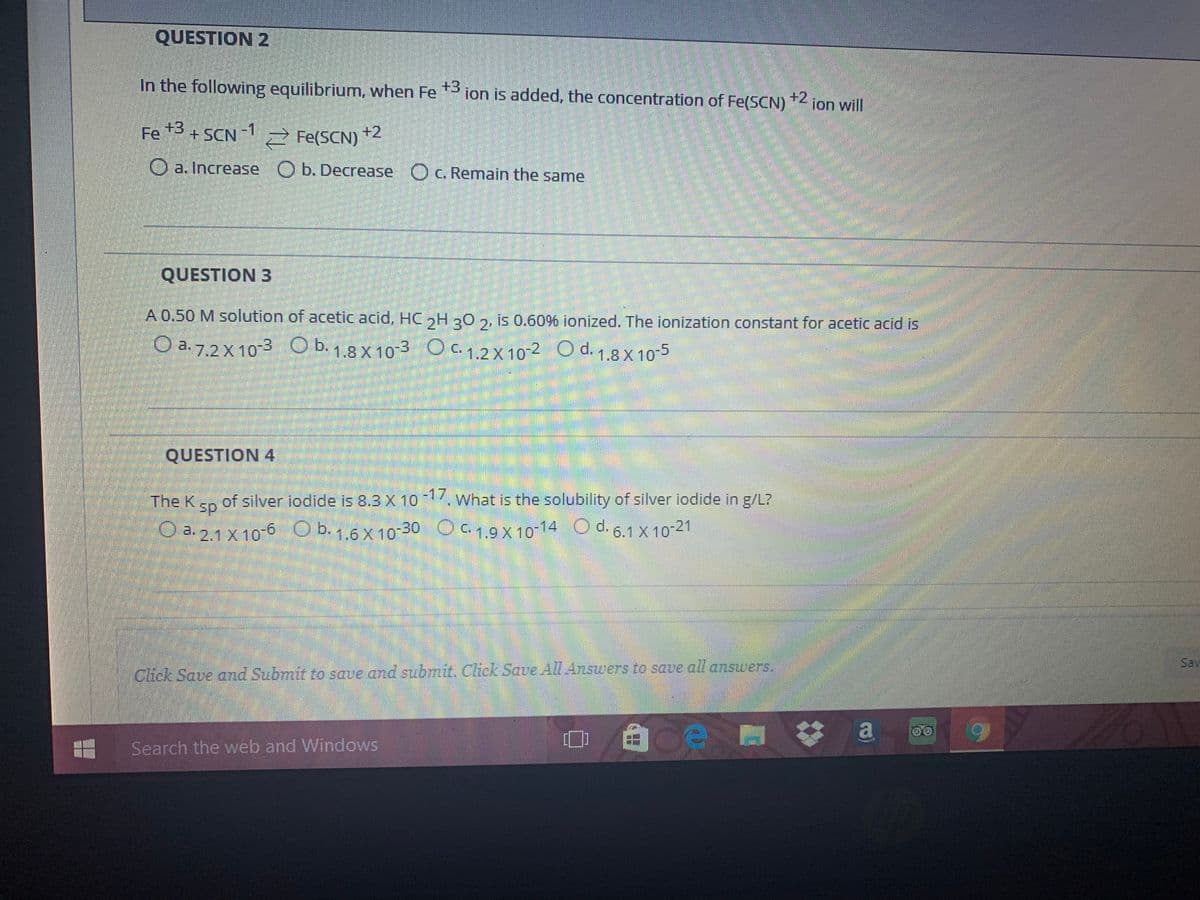
Transcribed Image Text:QUESTION 2
In the following equilibrium, when Fe *3 ion is added, the concentration of Fe(SCN)
) "< ion will
+2
Fe 3 + SCN
-SCN 1
-1
2 Fe(SCN)
+2
O a. Increase O b. Decrease O c. Remain the same
QUESTION 3
A 0.50 M solution of acetic acid, HC,H 20 2, is 0.60% ionized. The ionization constant for acetic acid is
O a. 7.2 X 10-3 O b. 1.8 X10-3 O c. 1.2 X 10-2 O d. 1.8 X 10-5
QUESTION 4
The K
of silver iodide is 8.3 X 10 What is the solubility of silver iodide in g/L?
sp
O a. 2.1 X 10-6 O b. 1.6 X 10-30 O C. 1.9 X 10-14 O d. 6.1 X 10-21
OC 1.9X 10 14
d. 6.1 X 10 21
Sav
Click Save and Submit to save and submit. Click Save All Answers to save all answers.
自
a
Search the web and Windows
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning


Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning