Q: 50.0 mL of 0.010M NaOH was titrated with 0.50M HCI using a dropper pipet. If the average drop from…
A:
Q: Please do not round off intermediate calculations. Thank you.
A: Given that : The mass of lead nitrate is 26.4g. The volume of ammonium sulfate solution is 150 mL.…
Q: What is the most likely product resulting from the reaction shown? Н 0= OH НО OH NaBH4 ? од
A: NaBH4 is a good reducing agent. It can reduce aldehydes , ketones , acid chlorides and anhydrides .…
Q: Show how you might synthesize this compound from an alkyl bromide and a nucleophile in an SN2…
A: For the synthesis of the following compound, we have first identify the bond that can be easily…
Q: Consider the decomposition reaction: XY2(s) X(g) + 2 Y(g) where X and Y are some chemical groups. At…
A: In the equilibrium constant expression, the concentration of pure solids and liquids are considered…
Q: Predict whether AS for each reaction would be greater than zero, less than zero, or too close to…
A: Entropy is a measure of the randomness of the system. The entropy change in a chemical reaction can…
Q: or the reaction: AgI(s) + 1/2Br2(g) → AgBr(s) + 1/2I2(s), ΔH° = –54.0 kJ…
A: Answer: Enthalpy of reaction is always equal to the difference between enthalpy of formation of…
Q: What is A and B? H3C НО :0 A P H3C Br Br О B H3C H3C О Br :0
A: Since you have posted a question with multiple sub-parts, we will solve the first three subparts for…
Q: Which of the following two molecules is more basic? Justify your answer A B
A: basicity is directly proportional to electron donation, the compound which can donate electron…
Q: Suppose 42.5 g of copper(II) nitrate is dissolved in 250. mL of a 0.70 M aqueous solution of sodium…
A:
Q: 100 80 60 40 20 0 25 50 75 100 m/z 125 150 M+ (80 175 3
A: We have find out the answer.
Q: The second-order rate constants for the reaction of oxygen atoms ·with aromatic hydrocarbons have…
A: Given: k1=1.44×107 dm3/mol s (1 L = 1 dm3)=1.44×107 L/mol s Initial temperature (T1) =300.3 K…
Q: Draw a structural formula of the R configuration of the compound shown below OH
A: GIVEN The R and S designations are based on the Cahn-Ingold-Prelog priority rules, which assign a…
Q: Use the following vapor pressure data to answer the questions: A B Liquid CH3COOC₂H5 C2H5NH2 Vapor…
A: Given that, We have to answer the following questions
Q: What is the correct value of the equilibrium constant Kc for the reaction: 2 NH3(g) N₂(g) + 3 H₂(g)…
A: Since,Equilibrium constant or Kc is the ratio of the equilibrium concentrations of product over…
Q: Light fuel oil with an average chemical composition of C10H18 is burned with oxygen in a bomb…
A: This problem is based on Thermodynamics. The solution is given below.
Q: The equilibrium constant, Ka, for a monopratic acid (propionic acid) is 1.3 ×10^-5. Which of the…
A:
Q: a. Magnesium chloride and sodium hydroxide. OMagnesium hydroxide, Mg(OH)2. Rule 5: Most hydroxide…
A: We need to use the given solubility rules to answer the questions.
Q: In a reaction where a carboxylic ester and water react to produce an alcohol and carboxylate (or…
A: In a reaction where a carboxylic ester and water react to produce an alcohol and carboxylate (or…
Q: For a generic equilibrium: HA (aq) H*(aq) + A¯(aq) which of the following statements are true? (A)…
A: Since the reaction given is HA(aq) ⇌ H+ (aq) + A-(aq) Hence the equilibrium constant expression…
Q: 2. Assign the R or S configuration to each chiral center in the molecules below. (H3C)3C. HO CH3…
A: Rules of R/S configuration i) Assign the priority to the directly attached atom based on their…
Q: 7. When investigating the boiling point of a liquid, a capillary tube is used to: a. Signal the…
A: The capillary rise method is a technique to determine the boiling point of a liquid. The boiling…
Q: A 0.430 g sample of a monoprotic acid is dissolved in water and titrated with 0.110 M KOH. What is…
A: Monoprotic acid is that acid which give only one proton upon dissociation. For example HCl is a…
Q: We use an ICE table to solve for equilibrium concentrations when given initial concentrations and…
A: Answer: When we have to find out the value positive change x for a system in equilibrium, we solve…
Q: Macmillan Learning What is the IUPAC name for the compound? H₂C-CH₂-CH-CH₂ IUPAC name: CH, OH…
A: IUPAC naming rules :1. Find the longest chain of carbon atoms and number them.2. Identify the…
Q: Part B Enter the ions formed when (NH4)2S dissolves in water. Express your answers as ions separated…
A: Part BAmmonium sulfide, NH42S is dissolved in water.We have to enter the ions formed when NH42S is…
Q: Gaseous ethane (CH₂CH3) will react with gaseous oxygen (O₂) to produce gaseous carbon dioxide (CO₂)…
A:
Q: What quantity in moles of NaOH need to be added to 200.0 mL of a 0.200 M solution of HF to make a…
A:
Q: Consider the reaction 3 Fe2O3(s) + H₂(g) → 2 Fe3O4(s) + H₂O(g) Using the standard thermodynamic data…
A: The balanced chemical equation given: 3 Fe2O3 (s) + H2 (g) → 2 Fe3O4 (s) + H2O (g)
Q: Of the following list of five compounds, select ALL that will form a PRIMARY ALCOHOL as the major…
A: We have been given five compounds and we have been asked all that will form a primary alcohol as the…
Q: A 12.7 mL solution of 0.100 mol L-1 HOCI is titrated using 0.150 mol L-1 NaOH. What is the pH of the…
A:
Q: How many O₂ molecules are needed to make 6 H₂O molecules? Explain how you know or show how you…
A: A question based on stoichiometry. An unbalanced equation is prescribed for which the appropriate…
Q: Acid-Base Definitions Match each term to its correct definition. Bronsted-Lowry acid 0 0 0 0 0 0…
A:
Q: B Current Attempt in Progress X Incorrect Draw the major product(s) for the following reaction. If a…
A: We have been given an organic reaction and we have been asked to draw the major product of given…
Q: 2 zdy NaOH NaOH 1) NaOEt; HOEt 2) H₂O
A: In cyclisation reaction formation of enolate takes place which further attacks carbonyl carbon of…
Q: Jon walked 89 feet in 11.5 seconds. What is his speed in miles per hour? (1mi)=(5280 ft)
A: Given , Jon walked = 89 feet Time = 11.5 seconds
Q: molecule proposed Lewis structure PO³ BH, XeO :0: | :0: [H-B-H]* :0: - | :0 Xe - I :0: .. Is this a…
A: The Lewis structure represents the valence electrons of each atom in the molecule. The bonded…
Q: The equivalence point in the titration of this weak acid is 50.80 mL. At 25.40 mL, the pH was…
A:
Q: David are cooking a dinner and the recipe calls for chicken broth. David realize that you don’t have…
A: David have the dried cube form of chicken broth that can be dissolved in water now we have to find…
Q: Show which peaks associate with which carbons and hydrogens in the benzaldehyde
A: The question is based on organic spectroscopy. we need to analyse the spectra and identify the…
Q: What is Kb for the conjugate base of HCN (Ka = 4.9 × 10-¹⁰)?
A: Ka is the acid dissociation constant. It is the equilibrium constant for the dissocation reaction of…
Q: hello i need help with this question Assuming standard conditions, answer the following questions.…
A: “Since you have posted multiple subparts questions, we will provide the solution only to the first…
Q: Suppose 2.89 g of zinc iodide is dissolved in 350. mL of a 46.0 m M aqueous solution of potassium…
A:
Q: The oxidation of aqueous iodide by arsenic acid to give 13 and arsenous acid proceeds via the…
A: We need to write the rate expression for (a) decrease of initial rate for I3-, Δ[I3-]/Δt (b) [H+],…
Q: 2. On the basis of charge stability, rank species A-G from weakest base to strongest base. ia NO HO.…
A:
Q: Calculate the change in pH when 7.00 mL of 0.100 M HCl(aq) is added to 100.0 mL of a buffer solution…
A: To solve this problem, we need to first determine the initial pH of the buffer solution. The buffer…
Q: Give IUPAC names for the following compounds. a) b) CH₂ CH3CCO2H CH3 0 ОН
A: -CO2H represent carboxylic acid (-COOH) functional group. Numbering starts from functional group…
Q: Aqueous sulfuric acid (H₂SO4) will react with solid sodium hydroxide (NaOH) to produce aqueous…
A:
Q: Strong-field ligands (such as CN-) stabilise the M2* oxidation state. Reflect on the reason.
A: Strong-field ligands, such as CN-, stabilize the M2+ oxidation state of a transition metal ion by…
Q: D. E. F. _OH НО. нази зи OCH₂CH₂CH3 НО.
A: The question is based on organic synthesis. we need to synthesize the product from the reactants…
Step by step
Solved in 3 steps with 1 images

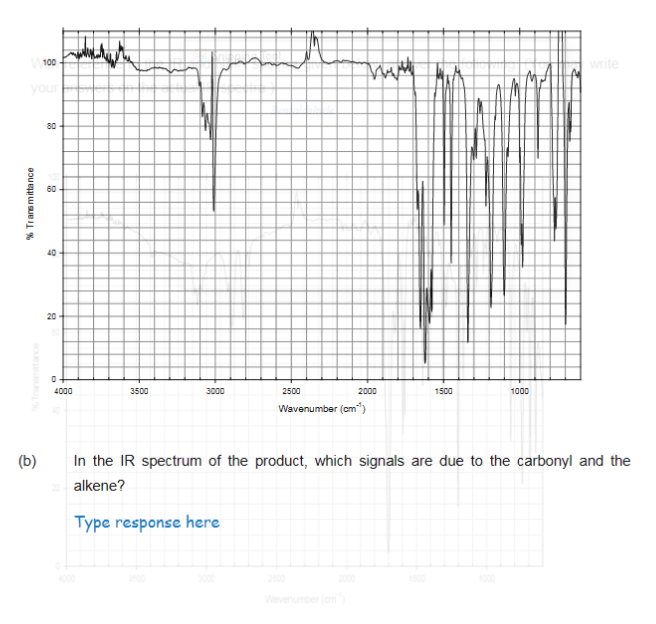
- PLEASE FOLLOW THE INSTRUCTIONS AND DO THIS TYPEWRITTEN ONLY. I WILL UPVOTE FOR YOUR GREATEST EFFORT. KINDLY SKIP THIS IF YOU HAVE ALREADY ANSWERED. THANK YOU SO MUCHShown is an IR spectrum of Xylene, determine what couild be wrong with the spectra?Fast pls solve this question correctly in 5 min pls I will give u like for sure Label functional groups and chiral carbons in the molecule below:
- Solve correctly please. PMMA, PCL, PLLA Which one of the polymers matches each FTIR spectra? One can be left out.Find the Ihd and label each spectra as the type of spectra it is , label the significant peaks in each spectra and determine the structure thank uExplain through a fragmentation mechanism the formation of theion rightly m/z appearing next to the following compounds.

