In the thermodynamic cycle depicted in the PV-diagram below, the NET work done BY the system is P PA В D! 2 4 6. 8 10 V (L) positive negative zero either positive or negative, but not zoro
In the thermodynamic cycle depicted in the PV-diagram below, the NET work done BY the system is P PA В D! 2 4 6. 8 10 V (L) positive negative zero either positive or negative, but not zoro
Chapter4: The Second Law Of Thermodynamics
Section: Chapter Questions
Problem 15CQ: Is it possible for a system to have an entropy change if it neither absorbs nor emits heat during a...
Related questions
Question
4.
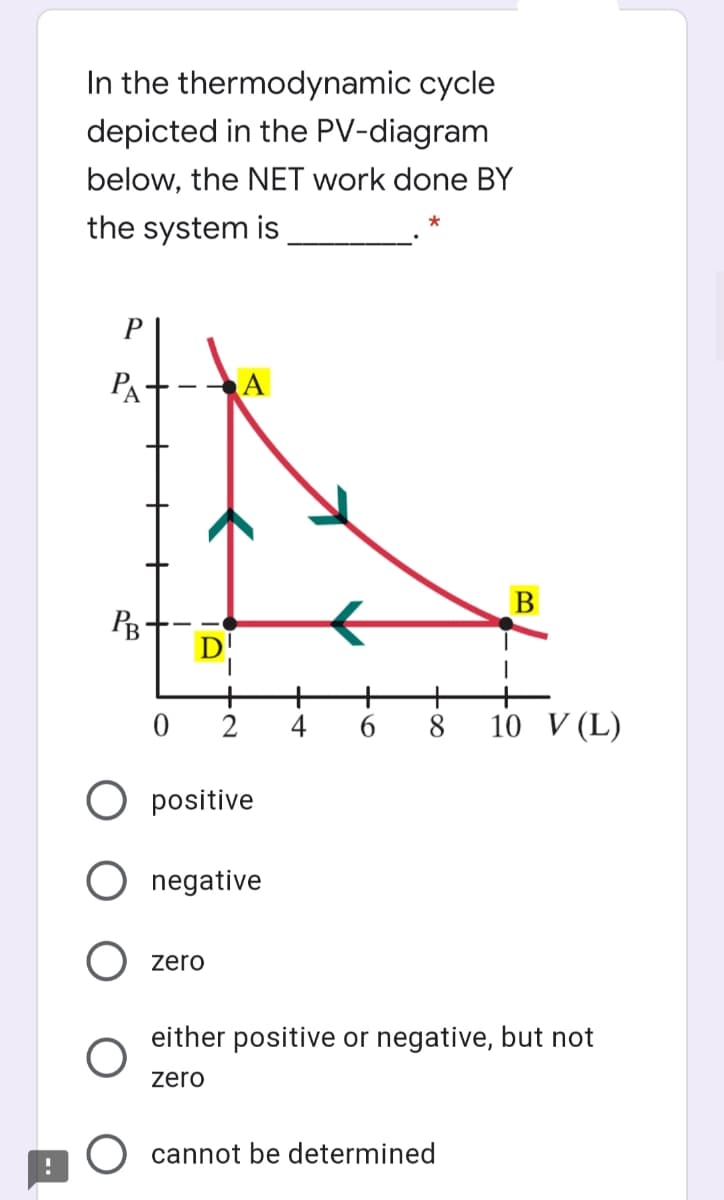
Transcribed Image Text:In the thermodynamic cycle
depicted in the PV-diagram
below, the NET work done BY
the system is
PA
B
4
10 V (L)
O positive
negative
zero
either positive or negative, but not
zero
O cannot be determined
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Recommended textbooks for you


Principles of Physics: A Calculus-Based Text
Physics
ISBN:
9781133104261
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning


Principles of Physics: A Calculus-Based Text
Physics
ISBN:
9781133104261
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning