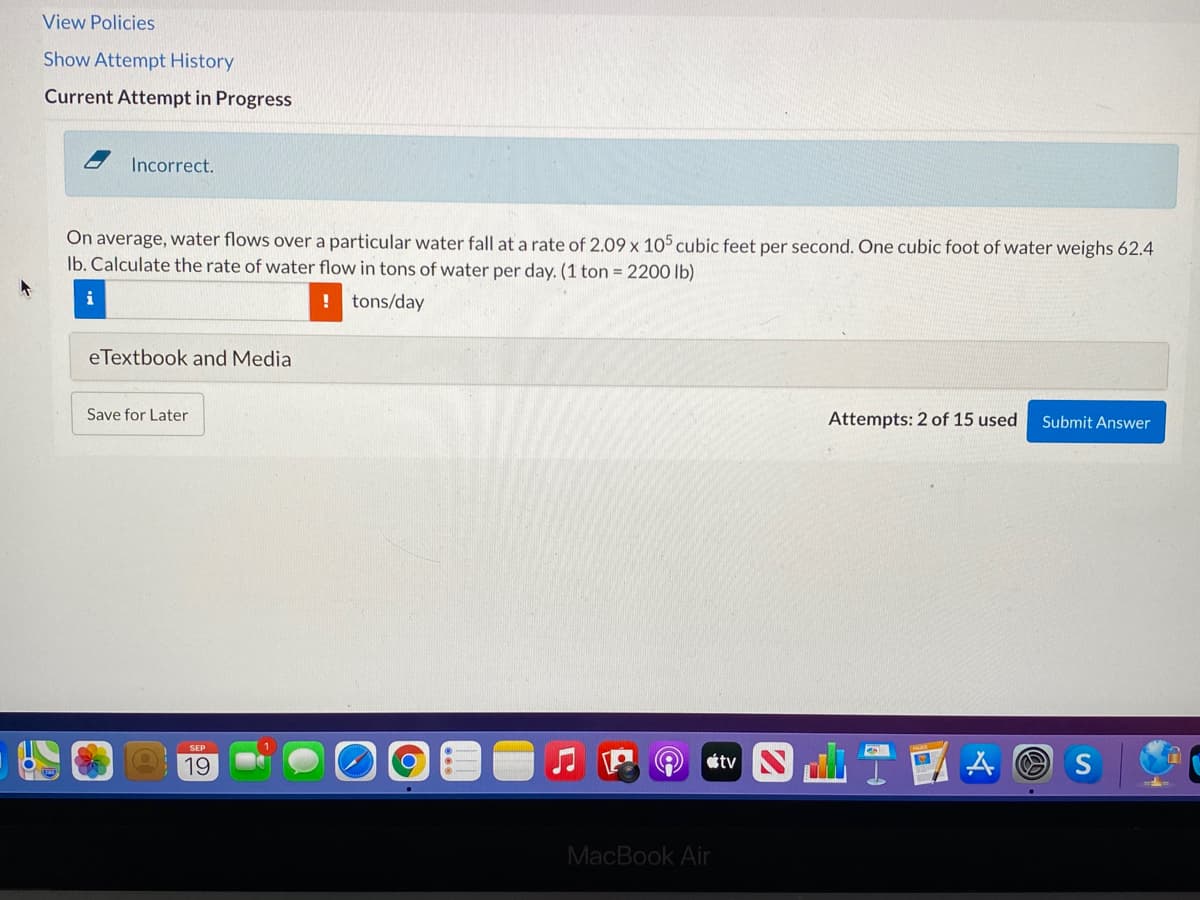
Incorrect. On average, water flows over a particular water fall at a rate of 2.09 x 105 cubic feet per second. One cubic foot of water weighs 62.4 Ib. Calculate the rate of water flow in tons of water per day. (1 ton = 2200 lb) i !tons/day eTextbook and Media Save for Later Attempts: 2 of 15 used Submit Answer
Incorrect. On average, water flows over a particular water fall at a rate of 2.09 x 105 cubic feet per second. One cubic foot of water weighs 62.4 Ib. Calculate the rate of water flow in tons of water per day. (1 ton = 2200 lb) i !tons/day eTextbook and Media Save for Later Attempts: 2 of 15 used Submit Answer
Chapter7: Statistical Data Treatment And Evaluation
Section: Chapter Questions
Problem 7.19QAP
Related questions
Question
Transcribed Image Text:View Policies
Show Attempt History
Current Attempt in Progress
Incorrect.
On average, water flows over a particular water fall at a rate of 2.09 x 105 cubic feet per second. One cubic foot of water weighs 62.4
Ib. Calculate the rate of water flow in tons of water per day. (1 ton = 2200 lb)
i
! tons/day
eTextbook and Media
Save for Later
Attempts: 2 of 15 used
Submit Answer
tv T
SEP
19
A O S
MacBook Air
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning



Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co