Indicate the strongest force holding the crystals together in the following substances by putting a check on the appropriate box. The first five substances were done for you. TYPES OF CRYSTAL Metallic Molecular lonic Covalent Network Substance Dispersio Dipole- dipole Hydroge n bonds Metallic bonds lonic bonds Covalent Network forces attraction 1 Kr 2 HF NO Al K.S NH, HCI KMNO, so, CO, 10 11 Si N34 567 00
Indicate the strongest force holding the crystals together in the following substances by putting a check on the appropriate box. The first five substances were done for you. TYPES OF CRYSTAL Metallic Molecular lonic Covalent Network Substance Dispersio Dipole- dipole Hydroge n bonds Metallic bonds lonic bonds Covalent Network forces attraction 1 Kr 2 HF NO Al K.S NH, HCI KMNO, so, CO, 10 11 Si N34 567 00
Introductory Chemistry: An Active Learning Approach
6th Edition
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Mark S. Cracolice, Ed Peters
Chapter15: Gases,liquids, And Solids
Section: Chapter Questions
Problem 5CLE
Related questions
Question
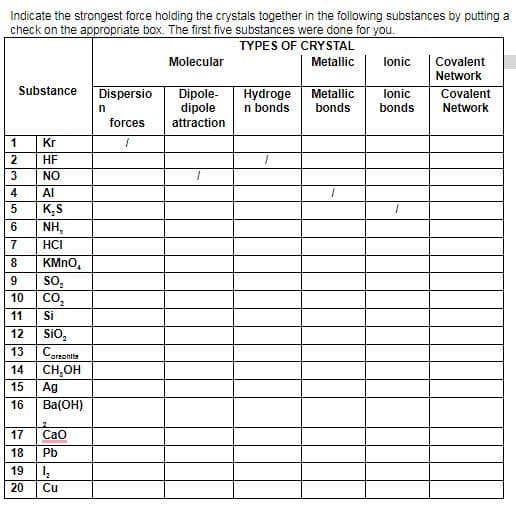
Transcribed Image Text:Indicate the strongest force holding the crystals together in the following substances by putting a
check on the appropriate box. The first five substances were done for you.
TYPES OF CRYSTAL
Molecular
Metallic
lonic
Covalent
Network
Substance
Dispersio
Dipole-
dipole
Hydroge
n bonds
Metallic
bonds
lonic
Covalent
n
bonds
Network
forces
attraction
1
Kr
HF
3
NO
4
AI
K.S
NH,
HCI
KMNO,
so,
co,
Si
10
11
Sio,
Coreonite
CH,OH
Ag
12
13
14
15
16
Ba(OH)
17
Сао
18
Pb
19
20
Cu
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning