Q: Determine the product(s) formed when cyclohexene is treated with the following reagents. Br Br Br OH…
A: Cyclohexene + HBr ----> bromocyclohexane cyclohexane + Br2/H2O -----> halohydrin derivative of…
Q: Shown are the H NMR spectra for 2 isomeric compounds three and four of the formula C5H10O. The IR…
A: The question is based on the concept of organic spectroscopy . we need to analyse the spectral data…
Q: 214Bi decays to isotope A by alpha emission; A then decays to B by beta emission, which decays to C…
A:
Q: Tutored Practice Problem 7.3.1 COUNTS TOWARDS GRADE Apply the Pauli exclusion principle. (a)…
A: According to Aufbau principle, electrons will occupy the lowest available energy level or orbital…
Q: Select the balanced nuclear equation for each of the following changes. (a) beta emission from…
A:
Q: An aqueous solution contains 0.404 M K₂SO3 and 0.205 M HSO3™. The pH of this solution is Submit…
A:
Q: The fluids within cells are buffered by H₂PO4 and HPO42. For H3PO4. Kat = 7.5 x 10-³, K2 = 6.2 ×…
A: A buffer is a solution which resist any change in pH on adding a small amount of acid or base . it…
Q: report A. J. Mukhedkar and N. V. Deshpande (Anal. Chem., 1963, 35, 47, DOI: 10.1021/ac60194a014) on…
A: The equation for the Beer-Lambert Law is: A = εlc where A is the absorbance, ε is the molar…
Q: An aqueous solution contains 0.441 M hypochlorous acid. Calculate the pH of the solution after the…
A: Henderson-Hasselbalch equation gives the relation between the pH and pKa of the acid. It can be…
Q: A chemist carefully measures the amount of heat needed to raise the temperature of a 106.0 mg sample…
A:
Q: The preparations of two aqueous solutions are described in the table below. For each solution, write…
A: To determine the major species present at equilibrium, we need to write the chemical equation for…
Q: Cycloheptatrienone is an unusually stable etc Draw an aromatic resonance structure that exp the…
A: There are several conditions that must be met for a compound to be considered aromatic: The…
Q: In the laboratory a student finds that it takes 19.2 Joules to increase the temperature of 13.2…
A:
Q: For the reaction 2H₂O(l) → 2H₂(g) + O2(g) 461 kJ and AH° = 572 kJ at 338 K AG° and 1 atm. = This…
A: Answer: Decomposition of water into hydrogen and oxygen gas is taking place and sign of Gibbs free…
Q: Provide a synthesis for the given target molecule, starting with an alkyl halide of your choice.…
A: We have been given a target molecule and we have been been asked to provide complete synthesis of…
Q: Choose the most likely regiochemical outcome for the reactants and conditions shown below: 1.…
A: The reaction is an example of oxymercuration and demercuration.
Q: Find the standard enthalpy of reaction for the reaction between solid magnesium chloride and a…
A: This question belongs to Chemical Thermodynamics. The standard enthalpy of a reaction is the…
Q: An aqueous solution contains 0.417 M nitrous acid. Calculate the pH of the solution after the…
A: Answer: Buffer solution is a type of solution that resists the change in its pH on adding small…
Q: Draw the reactants that would react to form this cyclohexene derivative in a Diels-Alder reaction…
A: In Diels-Alder reaction, diene reacts with dienophile to form an adduct in which the stereochemistry…
Q: An aqueous solution contains 0.422 M dimethylamine ((CH3)₂NH). How many mL of 0.312 M hydroiodic…
A: Given data: The molarity of the dimethyl amine solution = 0.422 M The molarity of the hydroiodic…
Q: Which of the following transformations represent an increase in the entropy of the system. Choose…
A: We have to tell among the below given options, on which case the entropy of the system is increased.…
Q: d. e. "OH NH3 D NH₂ (NH, is basic so you end with the carboxylate)
A: Nucleophilic substitution is a type of organic reaction where a nucleophile replaces a leaving group…
Q: does the N have to be there or can i write it without in (1)
A: It is a follow up question.
Q: 29 eBook Print References What product results from this reaction? HC с HC A 2 c=c H3C H₂C CH3 CH3…
A: We have been given an organic reaction and we have been asked the product of given organic…
Q: Draw the Michael adduct and the final product of the Robinson annulation reaction. Ignore inorganic…
A:
Q: MeO. MeO 19 Br MeO₂C CO₂Me R TMP, n-BuLi, THF, -78 °C, 1 h R OMe OMe 20, R = H 24, R = OMe, 65%
A: Organic reaction mechanism.
Q: are OP H aroma
A: If a species is aromatic, It should be: Molecule or species should be planner in shape. Molecule…
Q: 14.40 The equilibrium-constant expression for a reac- tion is K = [NH3]4[0₂]5 [NO] [H₂O]6 the What…
A: The equilibrium expression for the reaction is: Kc=NH34O25NO4H2O6 ......(1) Equilibrium constant is…
Q: 11 Determine the mass of chloride (MW= 35.45 g/mol) in grams present in 100mL of a 0.163 M solution…
A: Molarity is defined as number of mole of substance divide by volume of solution in liter
Q: Name: CH3CHCH=CHCH₂CH3 CH3 Name: 4-bromo-3-chloro-1-heptyne Name: 3-methyl-1-butyne Name: CH3- CH3 с…
A: We have to draw the hydrocarbon and give the name of the hydrocarbons. The given question is
Q: Visited A buffer solution is made that is 0.340 M in H₂S and 0.340 M in KHS. preview rupica…
A:
Q: Suppose i want to manufacture 200 litters of Acid free shampoo for bike or car washing. What is the…
A: To manufacture 1 liter of acid-free shampoo for bike or car washing, you can use the following…
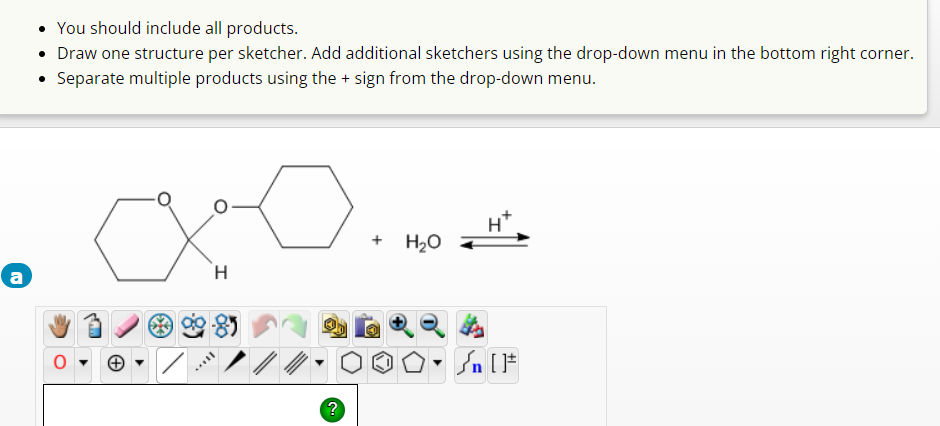
Q: Determine the products of the following reaction of alcohol. Give the name of the product that will…
A:
Q: How would you make a 9 μM solution of NaCl from a 90 mM stock solution?
A: We know that, 1 μM = 0.001 mM or 1 mM = 1000 μM Thus we have to prepare 0.009 mM solution of NaCl…
Q: Which explains why solutions of HCl in benzene (a nonpolar solvent) are nonconductive, while…
A: We need to select best option among given four options for the question:- Which explains why…
Q: Predict the major product of the following reaction. B Br₂ CH3CH₂SH All Br S H com 4 S 4 Br 'S +…
A: This reaction is based on electrophilic addition of alkenes.
Q: The standard cell potential E°(cell) is +0.77 V for the redox reaction 2 Fe³+ (aq) + H₂(g) → 2 H(aq)…
A: The standard cell potential E°(cell) is +0.77 V for the redox reaction 2 Fe³+(aq) + H2(g) → 2 H+(aq)…
Q: Draw structures for amino acids, recognize the different R groups.
A: The basic building blocks for the formation of proteins are called amino acids. In nature, there are…
Q: The hydroxide ion concentration, [OH-], of an aqueous solution of 0.560 M morphine (a weak base with…
A: we have to calculate the hydroxide ion concentration for the given solution
Q: 9:13 PM Tue Mar 28 Draw the major product of this Dieckmann condensation reaction. Ignore inorganic…
A: We know sodium ethoxide acts as a base and abstract the acidic proton of ketone leading to…
Q: Consider the reaction: CO(g) + Cl₂(g) → COC1₂ (9) Using standard thermodynamic data at 298 K,…
A:
Q: *O* Convert the following CH3 CHOR = b) Describe the mechanism of this reaction: PENS саза снаси…
A: The organic reactions takes place by adding various reagents, to get the desired final product.
Q: Draw the amide formed when 1-methylethylamine (CH3CH(CH3)NH2) is heated with each carboxylic acid.
A:
Q: Give the major product(s) of the following reaction. CH3CH₂CH₂CH₂CI (1 mole) MeO MeO MeO- -OMe AICI…
A: We have been given an organic reaction and we have asked to choose major product from given…
Q: What is the pH of a 0.168 M aqueous solution of ammonium perchlorate, NH4CIO4? This solution is O pH…
A: Given data,Molarity of NH4ClO4=0.168Mkb for weak base NH4OH=1.78×10-5 The relation between Ka and Kb…
Q: 1. The molecule below is ascorbic acid, also known as vitamin C. a. As drawn below, ascorbic acid is…
A:
Q: The molecule XH3 has a dipole moment. Is X boron or nitrogen? Question 7 options: a)…
A: The dipole moment of a molecule is a measure of the separation of positive and negative charges…
Q: Identify the best reagents to achieve the following transformation: HO. 1. HBr, ROOR; 2. NaOEt; 3.…
A:
Q: There is a gain in binding energy per nucleon when light nuclei fuse to form heavier nuclei.…
A: There is a gain in binding energy per nucleon when light nuclei fuse to form heavier nuclei.…
Q: Which functional group was introduced as a result of the transformation shown below? O halogen O…
A: The transformation of the given cycloalkene to the ketoaldehyde is an example of ozonolysis…
Step by step
Solved in 2 steps with 2 images

- Hi, I'd appreciate help with b-e for this problem.Draw the structures of the products of the neutralization reaction between methylamine and propanoic acid. Include all valence lone pairs in your answer. Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner. Separate multiple products using the + sign from the drop-down menu.1st [Select] options include: 0,1,2,3 or 4 2nd [Select] options include: unaffected by, attracted to, or repelled by
- Please help me with (c)Please help with row 3 highlighted in yellow (column 1 and column 2); the instructions for the specific column in row 3 are above row 1: Follow the instructions in each column. Hint for the last column: draw it with a wedge and again with adash – which gives the correct configuration?Help on letter B please

