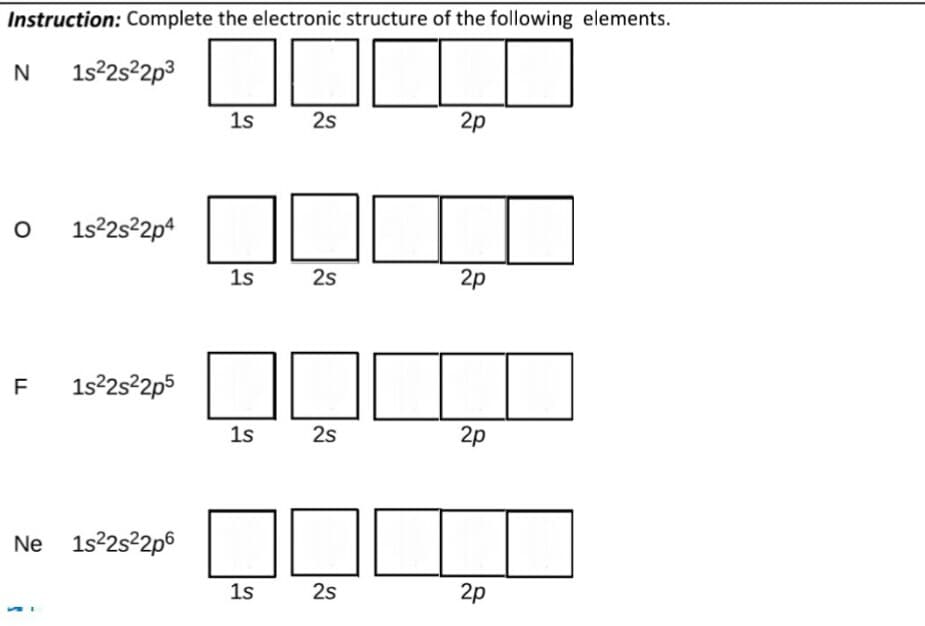
Instruction: Complete the electronic structure of the following elements. N 1s22522p3 1s 2s 2p 1s22s22p4 1s 2s 2p F 1s25²2p5 1s 2s 2p Ne 1s22s22p6 1s 2s 2p
Q: 6.9.2 What element is represented by the following electron
A: The element is represented by the following electronic configuration is [Kr]5s24d105p5, a) Tc b) Br…
Q: Question 3 a) The Schrödinger equation for Li²+ is a2 + 87²m ax² ' ay² ' az² h? 3e? Ý = Eµ Briefly…
A: Given:- The Schrodinger equation is:…
Q: QUESTION 3 Based on the Hermite polynomials given in Table 11.1, which wavefunction equation below…
A: The wavefunction equation which is suitable to describe ideal harmonic motion for n= 3 is
Q: 6 of 11 M Review | Constants Periodic Table Part E Now, find the value of 3 2 16 5 7 3 4 2 Finding…
A: Numerator which comes above Denominator which comes below Example :- 5/6 Here 5 is numerator and…
Q: Part A Choose the electron configuration for Y²+. O [kr]5s²4d6 O [Kr]4d¹ O [Kr]5s24d¹ O [kr]5s²4d² O…
A:
Q: QUESTION 2 What is the sample absorbance of a solution having a transmittance of 65.2%? O a. 0.186 O…
A: Given is, %T =65.2%
Q: 100 90 80 70 60 50 40 30 20 10 0 4000 3000 2000 1500 wave number, cm-1 WY 1000 500
A: The given spectra is for IR spectroscopy. IR spectroscopy useful for identifying functional groups.…
Q: Period # of Electrons Boiling Point Group Compound -162°C CH, -112°C SiH, -90°C GeH, -50°C SnH,…
A: We will determine the period in which the centre atom belong as well as calculate the valence…
Q: Select the correct electron configuration for Cu. Group of answer choices [Ar]4s23d9 [Ar]4s13d10…
A: Electron configuration is referred to as the distribution of electrons of an atom or a molecule, in…
Q: 28.9 nm to μm. could I get a step by step, please?
A: 1 nanometre = 0.001 micrometre 1 nm = 0.001 μm By using this conversion factor we can convert the…
Q: Part B Use the data from the table above to provide estimates for the first ionization energy of an…
A: Ionization energy (EI): When an electron is removed from the outermost shell of an isolated gaseous…
Q: Using the spdf form, write out the expected electron configuration for chromium. The actual electron…
A: Some atoms have different electronic configurations to get stability.
Q: Assume there are two pairs of harmonic oscillatorcs A-A and B-B. If A and B are isotopes, given…
A: In this question, two pairs of harmonic oscillators A-A and B-B are given. It is given that A and B…
Q: Problem • What is the frequency of light emitted from an electron in the hydrogen atom going from…
A: The frequency of this light emitted is to be calculated.
Q: a. Circle the particle that is NOT neutral (1) p-26 e-26 n-30 (2) p-30 e-28 n=35 (3) p=16 e-16 n=18…
A:
Q: Identify the smallest atom. Group of answer choices Be Li Xe C
A: In the given question we have to identify the smallest atom in the group of atoms mention. we have…
Q: Match the species on the left (a-e) with their corresponding colors on the right (1-5):
A: Answer: Hg2Cl2 is white in color. PbCrO4 is yellow in color .It is used in paints and pigments. NiS…
Q: There are .equivalent positions in 2/m Select one: O 4 O 1 O 8 O 2 The term 'Dimensions is equal to…
A: solution - (a); There are 2 equivalent position in 2/m (b); The term 'dimensions ' is…
Q: Q2))What do you infer from the figure on the left? 1) Hund's rule violation. 2) Violation of the…
A: Hund's Rule Hund's rule states that the electrons are singly occupied in orbitals and then it gets…
Q: 7. Quantum number that describes the size of the orbital a) spin b) magnetic c) orbital d) main
A:
Q: Calculate the lambda max of the given compound using Woodward-Fieser Rules. R AcO Select one: O 303…
A: Woodward–Fieser rules: The Woodward–Fieser rules are used for the calculation of the wavelength of…
Q: 1. Explain nuclear magnetic moment does not align do not immediately align with applied magnetic…
A: The nuclear magnetic moment is the moment of an atomic nucleus and arises from the spin of the…
Q: Classify these atomic orbitals as dxy, dyz, dxz, d2-y, or d2. d2 d2-y2 dyz dry Answer Bank
A: The shape of dxy, dyz, and dxz are shown below.
Q: Which of the following represent impossible combinations of nn and ℓℓ? Check all that apply. Check…
A: The maximum permitted value of l is n-1. For 2d: n=2 l=2-1=1 So, only s and p subshell is present…
Q: 12. Which one of the following is the electron configuration for ground state Cr(II)? a. [Ar]3d4 b.…
A: Which one of the following is the electron configuration for ground state Cr(II)?
Q: 1: Answer 1 question of the following 1- For "Co calculate:- A) magnetic state (term symbol) B)…
A: acc. to bartleby we can solve only 3 parts
Q: The spin and parity of J/psi meson J/y : JP О а. 1 O b. 1* Oc. 0* 33 d. 0
A: Concept: The J/ψ(J/psi) meson or psion is a type of subatomic particle. It is a flavor-neutral meson…
Q: (MULTIPLE CHOICE) This dot structure represents which of the following answers: - [Ne] 3s^2 3p^3…
A:
Q: what is the lambda of this picture? Choices: 227nm ,263 nm, 234 nm ,273nm
A: Woodward–Fieser rules: The Woodward–Fieser rules are used for the calculation of the wavelength of…
Q: 18.) Which value: n (Principal), l (Angular), ml (Magnetic) or ms (Spin) would you change to make…
A: n=2 valid l= 1 valid m= -1 valid ms= 0 not valid Spin value is either +1/2 or -1/2 but can't be 0 so…
Q: Choose the electron configuration for Fe6+. O [Ar]4s2 O [Ar]4s23d4 O [Ar]3d² O[Ne]3s23p6 O…
A: Since, Electronic configuration represent the electron present in orbital in which way. Arrangement…
Q: nce started, this test must be completed in one sitting. Do not leave Remaining Time: 31 minutes, 29…
A: Infrared spectroscopy:Infrared spectroscopy also known as vibrational spectroscopy.This method is…
Q: Write the full ground-state electron configuration for the following. (Type your answer using the…
A:
Q: Solve ASAP R = 1.12 × 10⁷ m-¹ 1) Find λ = ? (From n = 5 to n = 2) 2) ΔE --> Z = 6 find the…
A:
Q: Why pi(π) bond needed a longer wavelength than the sigma bond? Please shortly answer at your own…
A:
Q: Compute the empherical formula of the following: a. 19.1% Sn and 80.9% I b. 25.9% Fe and 74.1% Br
A: Given, a). 19.1% Sn and 80.9% I b). 25.9% Fe and 74.1% Br Determine the empirical formula of the…
Q: Indicate if the following electron configurations are correct or not. Identify and correct any…
A: The following electronic configuration is correct or not has to be indicated The errors has to be…
Q: MCQ 16: The electronic configuration of clectrons involves their addition starting from lowest…
A: In case of entry of electrons into the orbitals it maintains Aufbau principle, which says that…
Q: Part A A;H° = - 87 kJ mol 1, A,S = – 150 JK-' mol ', T = 295 K Express your answer as an integer. ?…
A:
Q: 10. What is the ground state configuration for the element vanadium (V)? a. 1s2s2p63s²3p 4s? b.…
A: Ground state electronic configuration of V (vanadium) = ?
Q: A local FM radio station broadcasts at an energy of 6.26 x 10-29 kJ/photon. (1 MHz = 106 s-¹)…
A:
Q: Direction: Cite 5 elements that can be magnetized and 5 that can nullify magnetism. MAGNETIZERS…
A: Magnetisers can be defined as the substances that can impart magnetism to other substances.
Q: Consider the d4 electron configuration of a free ion
A: Formula to calculate spin multiplicity is (2S + 1) where s is the spin of an electron
Q: C. How many electrons in the carbon atom (C) with the value of ml = 0?
A: The electronic configuration of an atom is understood on the basis of quantum number: The four…
Q: Compute the empherical formula of the following: a. 19.16 Sn and 80.9% I b. 25.9% Fe and 74.1% Br
A: With the help of percentage of composition of elements and their atomic masses we can determine the…
Q: QUESTION 15 Match the following elementslions with the correct ground state electronic…
A: According to the question, we need to write down the electronic configuration of the following…
Q: 36R) Which one of the following configurations depicts an excited oxygen at A) 1s22s22p2 B)…
A: Oxygen atom having atomic number 8 having 8 electrons.
Q: MCQ 24: CI has the electronic configuration of A. 1s, 2s, 2p, 3s2 B. 1s2, 2s, 3s, 3p C. 1s, 2s², 2p,…
A: We know that, Atomic number of Cl is 17. So,
Q: Calculate the transition dipole moment, umn = | m (T)Hz n (T)dT where uz = -er cos(0) for a…
A:
Q: Identify the elements (two of these are irregular, so pay close attention). (a)…
A: In the periodic table, elements are arranged as the function of atomic numbers. Electrons are filled…
Step by step
Solved in 2 steps with 1 images

- In class, we learned why chromium (Cr) has a 4s13d5 electronic configuration instead of 4s23d4. By the same reasoning, shouldn’t carbon (C) have a 2s1 2p3 configurationand not the 2s2 2p2 configuration that it actually has? Explain why the 2s2 2p2 configuration ismore stable for carbon while the irregular 4s1 3d5 electronic configuration is more stable forchromium.What is the structure of the unknown? Determine it by analyzing the spectroscopic data given. thank you!Pls give me the answer to this one so I can study
- Show work with explanation needed of each options with structures...don't give Handwritten answer..don't use Ai for answering thisC12H13O3 (find structure )The problem is asking how many Sp3, Sp2, and Sp atoms there are and the answer key says 16 Sp3, 8 Sp2 and 2 Sp but I'm not even counting 26 carbons on this molecule so I'm a little confused.

