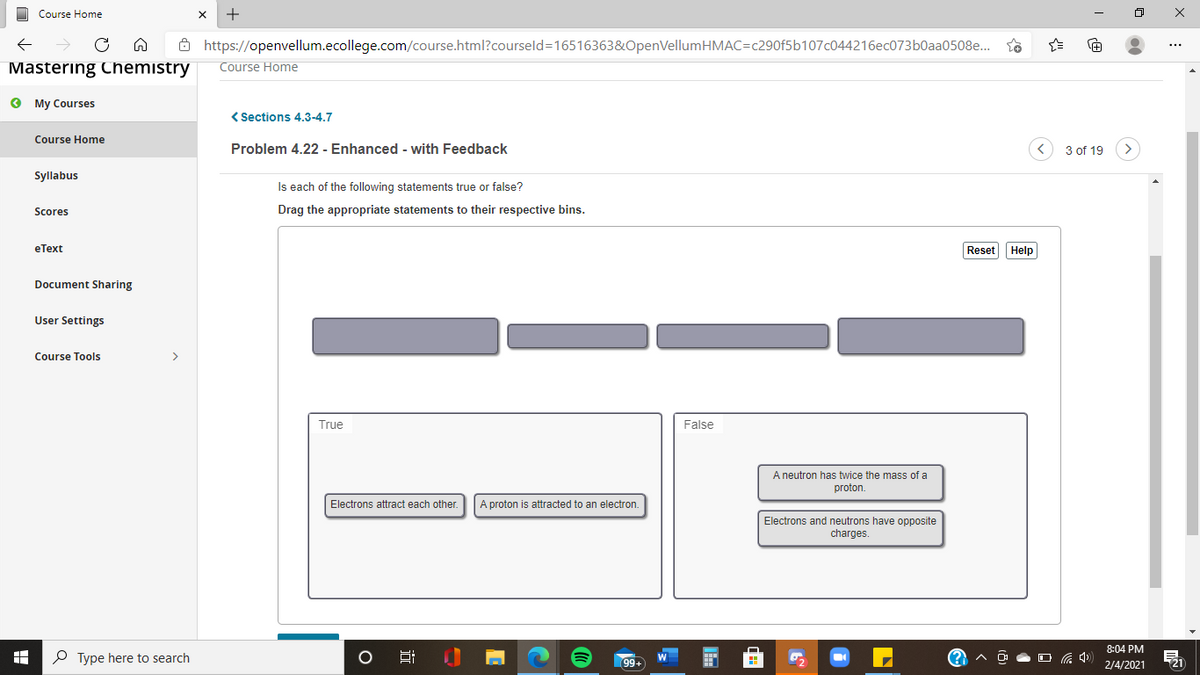
Is each of the following statements true or false? Drag the appropriate statements to their respective bins. Reset Help True False A neutron has twice the mass of a proton. Electrons attract each other. A proton is attracted to an electron. Electrons and neutrons have opposite charges.
Is each of the following statements true or false? Drag the appropriate statements to their respective bins. Reset Help True False A neutron has twice the mass of a proton. Electrons attract each other. A proton is attracted to an electron. Electrons and neutrons have opposite charges.
Chapter8: Sampling, Standardization, And Calibration
Section: Chapter Questions
Problem 8.20QAP
Related questions
Question
help?
Transcribed Image Text:Course Home
8 https://openvellum.ecollege.com/course.html?courseld=16516363&OpenVellumHMAC=c290f5b107c044216ec073b0aa0508e.
Mastering Chemistry
Course Home
O My Courses
< Sections 4.3-4.7
Course Home
Problem 4.22 - Enhanced - with Feedback
3 of 19
Syllabus
Is each of the following statements true or false?
Scores
Drag the appropriate statements to their respective bins.
eText
Reset Help
Document Sharing
User Settings
Course Tools
>
True
False
A neutron has twice the mass of a
proton.
Electrons attract each other.
A proton is attracted to an electron.
Electrons and neutrons have opposite
charges.
8:04 PM
P Type here to search
99+
2/4/2021
近
Expert Solution
Given
A question about atomic structure, which is to be accomplished.
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

