It is believed that the reaction responsible or he depletion 0201e in the atohosphere 's Oalg)'t NoG)OgG) tMO, G) 17 46-41 q Q rearts with excess MO, how man will be produced?
Q: 39. The emissions for the series Kr-93→Rb-93→Sr-93→Y-93 are β, β, β β+, β+, β+ α, β+, α α,…
A: The given series is : Kr-93→Rb-93→Sr-93→Y-93 OR 36Kr93 → 37Rb93 → 38Sr93 → 39Y93 Now, β-particle…
Q: The concentration of H2O in the stratosphere is about 5ppm. It undergoes photodissociation according…
A: The photochemical dissociation of water molecule.
Q: It requires 364 kJ/mol to break the chemical bond in HBr molecules. 1. What is the longest…
A: 1.It requires 364 kJ/mol to break the chemical bond in HBr molecules.
Q: Average ozone concentrations in Jakarta, Indonesia have been reported to be 0.015 mg m^-3 and in…
A: Since ppbv i.e parts per billion by volume is given by ppbv = mass of solute in g X 109 / volume of…
Q: Can you help with question 3? Thanks!
A: (a)Heat required to increase the temperature of coffee is calculated as followsSpecific heat…
Q: What is the volume of 3.4 gram of annomia at R.T.P? [1 mole of any gas at R.T.P has a volume of 24…
A: Given data contains, Grams of ammonia is 3.4 grams. The formula of ammonia is NH3. The atomic mass…
Q: (1) (a) (b) (c) Identify the three main products resulting from the OH initiated oxidation of…
A: 2,2-dimethyl butane are volatile organic compounds that are constituent of air pollution.
Q: Puffer Lake is a natural mountain lake located at an elevation of 9700 feet. If Torriceli's…
A: Answer: Option D. 515 mmHg is correct
Q: Define the term troposphere?
A: To find: The definition of troposphere
Q: The degradation of CF3CH2F (an HFC) by OH radicals in the troposphere is first order in each…
A:
Q: 2. In a Mars Direct mission, the astronauts would return home in an Earth Return Vehicle (ERV)…
A:
Q: The degradation of CF3CH2F (an HFC) by OH radicals inthe troposphere is first order in each reactant…
A: As there are two reactants and the reaction is first order in each reactant, the rate of the…
Q: 32,07+16.00(3)= 8. What is the density of SO3 at STP? P=dRT/
A: Since you have posted multiple questions, we are entitled to answer the first only.
Q: Use the following information for as needed for questions 4,5 and 6 :for the following reaction at…
A: Note : since the reduction potential of Al is -1.676 V and not the oxidation potential. Hence in the…
Q: Is Mechanism I or Mechanism II of ozone destruction considered responsible for the ozone hole over…
A:
Q: At a constant external pressure of 43.6 bar, a mole of XX reacts via the reaction X(g)+4Y(g)→2Z(g),…
A:
Q: A 2.5 L tank can withstand a maximum pressure of 800 kPa. Calculate the temperature that the tank…
A:
Q: O3 is produced in the troposphere through the photochemical oxidation of NO2. The photolytically…
A: O3(g) + NO(g) → NO2(g) + O2(g)
Q: Combustion of a 1.450 g sample of glucose yielded 23.01 KJ of heat. The calorimeter contained 1.900…
A: The final temperature will be 24.18°C.
Q: A reaction at 25.0 °C evolves 166. mmol of carbon monoxide gas. Calculate the volume of carbon…
A: Applying ideal gas equation. PV = nRT
Q: Asphalt is composed of a mixture of organic chemicals. Does an asphalt parking lot contribute…
A: The chemical reactions that lead to the formation of smog are: Nitrogen dioxide breaks down in…
Q: A reaction at – 19.0 °C evolves 177. mmol of carbon dioxide gas. Calculate the volume of carbon…
A: Temperature = -19.0 ° C .... Or because °C + 273 = k ....so, -19.0°C + 273 = 254 k Number…
Q: A reaction at 20.0 °C evolves 333. mmol of boron trifluoride gas. Calculate the volume of boron…
A:
Q: A reaction at 8.0 °C evolves 420. mmol of carbon dioxide gas. Calculate the volume of carbon dioxide…
A:
Q: Suppose you have an 8-hour ozone value of 0.088 ppm, and a PM2.5 value of 35.7 ug/m3. The AQI value…
A: AQI stands for Air Quality Index which serves as a means to measure the extent of Air Pollution.
Q: A test of an automobile engine’s exhaust revealed that 3.4 g of NO2was emitted in 10. minutes of…
A: Given that 3.4 g NO2 was emitted in 10 miniutes.Number of moles of NO2 emitted = Given mass Molar…
Q: ample human blood was measured to pe 7.52 at 25°C. Calculate pOH, CH+], and [OH-] for the sample.…
A: pH +pOH = 14 pH = -log[H+] pOH = -log[OH-]
Q: How many collisions does a single Ar atom make in 1.0 s when the temperature is 25oC and the…
A:
Q: Why are CHF2Cl and CH2ClCF3 important for stratospheric ozone depletion? pls consider its lifetime.
A: Life time of CHF2Cl and CH2ClCF3 are 11.9 and 13.4 years respectively.
Q: Find YW V. W 19 Y 31
A:
Q: A reaction at 19.0 °C evolves 193. mmol of chlorine pentafluoride gas. Calculate the volume of…
A:
Q: The average translational kinetic energy for a molecule (Erans) is given by the following equation:…
A:
Q: The following data is from a prospective cohort study examining the association between air…
A:
Q: Which of the following expressions is NOT equivalent to nR? as dP н as - P dP as 1 T ov dP
A: Option (3) is correct
Q: A reaction at 28.0 °C evolves 451. mmol of sulfur tetrafluoride gas. Calculate the volume of sulfur…
A: Given :- No.of moles of SF4 = 451. mmol Temperature = 28.0°C Pressure = 1 atm To determine :-…
Q: What happens to the rates of ozone formation and destruction in the absence of outside Intervention?…
A: In the Stratosphere the ozone gas is formed by the oxygen gas in the presence of solar ultraviolet…
Q: A reaction at 30.0 °C evolves 520. mmol of sulfur tetrafluoride gas. Calculate the volume of sulfur…
A: For sulfur tetrafluoride gas, Temperature, T = 30°C = 30 + 273.15 K = 303.15 K Number of moles, n =…
Q: Given f(x) = x- 7, what will f(12) be equal to? O 12 19 O 5
A: Variable: The term whose value is not fixed. e.g. x is the variable is the given equation. Constant:…
Q: Argon diffuses at a rate of 1.45 meters in 8.65 minutes. How long will it take helium to diffuse…
A: Rate of diffusion of Argon (Ar) = distance / time = 1.45 / 8.65 = 0.16763 m/min Since according to…
Q: A reaction at 34.0 °C evolves 174. mmol of carbon monoxide gas. Calculate the volume of carbon…
A:
Q: When we study photochemical processes in the upper atmosphere, we need to know how frequently atoms…
A: The mean free path of molecules is provided by the average distance that the molecule moves between…
Q: The greenhouse effect is a natural process that maintains the Earth's temperature within a range…
A: We have to tell which option is correct from given choices of options
Q: 15. If the concentration of ClO. in the South Pole air is 2.0 x 10° molecules/cm3 at 1 atm and…
A: The expression of ideal gas equation is shown below: PV = nRT Where; P = pressure V = volume n =…
Q: List two types of chemical compounds that must be present in the air for photochemical smog to from.…
A: Photochemical smog is a mixture of pollutants that are formed when nitrogen oxides and volatile…
Q: How many collisions does a single Ar atom make in 1.0 s when the temperature is 25 °C and the…
A:

The reaction is given as follows:
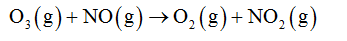
The mole of O3 is calculated as follows:
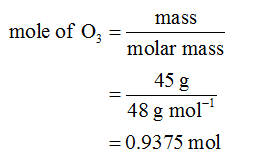
According to the reaction,
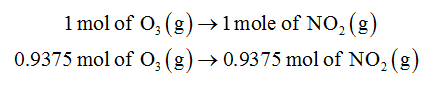
Step by step
Solved in 3 steps with 4 images

- Halons are fluorocarbons that contain bromine, such asCBrF3. They are used extensively as foaming agents forfighting fires. Like CFCs, halons are very unreactive andultimately can diffuse into the stratosphere. (a) Based onthe data in Table , would you expect photodissociationof Br atoms to occur in the stratosphere? (b) Propose amechanism by which the presence of halons in the stratospherecould lead to the depletion of stratospheric ozone.The coal whose composition is given in the table below is burntwith 100% excess air. Calculate (a) the theoretical oxygenrequirement per 100 kg of coal (b) the theoretical air requirementper 100 kg of coal (c) the Orsat analysis of the gases when thecoal is burnt with 100% excess air. Component Weight % C 70.32 H2 3.91 S 0.52 O2 25.25Assume that a single cylinder of an automobile engine hasa volume of 524 cm3. (a) If the cylinder is full of air at 74 °Cand 0.980 atm, how many moles of O2 are present? (Themole fraction of O2 in dry air is 0.2095.) (b) How manygrams of C8H18 could be combusted by this quantity ofO2, assuming complete combustion with formation ofCO2 and H2O?
- its also not 4.57e-13The ultraviolet spectrum can be divided into three regionsbased on wavelength: UV-A (315–400 nm), UV-B (280–315nm), and UV-C (100–280 nm). (a) Photons from whichregion have the highest energy and therefore are the mostharmful to living tissue? (b) In the absence of ozone, whichof these three regions, if any, are absorbed by the atmosphere?(c) When appropriate concentrations of ozone arepresent in the stratosphere, is all of the UV light absorbedbefore reaching the Earth’s surface? If not, which region orregions are not filtered out?33a, no need to explain too much.
- This is PV=nRt? But what is n then?Assume that a single cylinder of an automobile engine hasa volume of 524 cm3. (a) If the cylinder is full of air at 74 °C and 0.980 atm, how many moles of O2 are present? (Themole fraction of O2 in dry air is 0.2095.) (b) How manygrams of C8H18 could be combusted by this quantity ofO2, assuming complete combustion with formation ofCO2 and H2O?The gas-phase reaction between hydrogen and iodine H2(g)+I22HI(g)proceeds with a rate constant for the forward reaction at 700C of 138 L/moI s and an activation energy of 165 kJ/moI. (a) Calculate the activation energy of the reverse reaction given that H f for HI is 26.48 kJ/moI and H f for I2(g) is 62.44 kJ/mol. (b) Calculate the rate constant for the reverse reaction at 700C. (Assume A in the equation k=AeE a /RT is the same for both forward and reverse reactions.) (c) Calculate the rate of the reverse reaction if the concentration of HI is 0.200 M. The reverse reaction is second-order in HI.
- The amount of anthropogenic SO2 emission from Pakistan was 180 kilo tonnes(kt) in 20191.How many grams of SO2 were emitted to the atmosphere?How many grams of Sulfur was emitted to the Atmosphere?Pakistan population was 216.6 million in 2019. How many kg of solidSulfur/Head was emitted?Refer to the following equation: Mg (s) + 2HNO3(aq) -> Mg(NO3)2 (aq) + H2 (g) A reaction between 0.359 grams of Mg and 21.12mL of a 0.450M HNO3 solution was conducted in a room witht he following conditions: 27.5 degrees Celius, 723mmHg of atospheric pressure. If .15mL of H2 were collected over water (by the water displacement method), calculate the percent yeild of the reaction. (Pressure H2O at 27.5 degrees Celius = 27.82 mmHg)[B] *refer tot he (2) photos below*

