Q: Additional Activities This part will intermolecular forces present between and among species to the ...
A: a.In case of HF, there is weak intermolecular hydrogen bonds present which lead to increase in the m...
Q: The rate-determining step for a chemical reaction that proceeds by multiple mechanistic steps, is th...
A: A chemical reaction proceeds via multiple steps is depends only on one step, which also decides the ...
Q: for the following electrophilic substitution reaction, choose the resonance form that will NOT be ob...
A: Given here a electrophilic substitution reaction and we are asked to choose the resonance form that ...
Q: Find AH for CaCO3 (s) → CaO (s)+ CO2 (g) given: 2 Ca (s) + O2 (g) → 2 CaO (s) Ca (s) + CO2 (g) + 1/2...
A: Given: 2 Ca (s) + O2(g) -----> 2 CaO (s) .......(1) ∆H1 = -1269.8 kJ Ca(s) + CO2 (g) + 1/2 O2...
Q: The Henderson-Hasselbalch equation relates the pH of a buffer to the relative concentrations of the ...
A: Henderson-Hasselbalch equation is a mathematical equation that provides the relationship between pH,...
Q: Evidencefor the intermediate carbocations in the acid-catalyzed dehydration of alcohols comes from t...
A: a) Mechanism for the formation of 2,3-dimethyl-2-butene from 3,3-dimethyl-2-butanol.· 1- The first ...
Q: Chemistry Question
A:
Q: What minimum amount of voltage would you need to apply to successfully run the electrolysis of molte...
A: anode: 2H2O(l) ⟶ O2(g) + 4H+(aq) + 4e− E∘anode = +1.229 V cathode: 2H+(aq) + 2e− ⟶ H2(g) ...
Q: 33 degrees Celsius is what temperature in Kelvin? O 298 K О 33 к O 273 K 306 K
A: We have given simple conversion of temperature in degree celsius to kelvin
Q: Im confused. Because I think it could be c or d
A: STP stands for standard temperature and pressure.
Q: How many grams of glucose (C6H12O6, 180.16 g/mol) would be required to prepare 2500.0 mL of a 0.265 ...
A: The volume of the solution is = 2500.0 mL The molarity of the solution is = 0.265 M The molar mass o...
Q: A solution is made from 0.51 M NH3 and 0.48 M NH4Cl. What is the pH if the Kb for NH3 is 1.8x10-5? ...
A:
Q: Predict the mode of decay begin 65^ Ni undergoes. Positron Beta It is stable and does...
A: The heavier isotopes having mass number more then 200 under go alpha decay. Light isotopes( mass num...
Q: Why is the equivalence point pH necessarily below 7 when a weak base is titrated with strong acid?
A: Equivalence point of a acid-base titration is a point when equivalent amount of acid and equivalent ...
Q: QUESTION 29 For each of the following substances, enter an A if it is Acidic, an N if it is Neutral ...
A: If compound has a proton attached to N,O,S, Se, a halide or has a positive charge it's an acid and i...
Q: 6
A:
Q: Compare the two values below: A number of lone pairs on central atom in NH, B number of lone pairs o...
A: Correct answer is (a) A is larger than B. Number of lone pairs on central atom in NH3 = 1 A = 1 Numb...
Q: Which compound on treatment with Nal in acetone form 2iodopropane?
A: Racemization is a process by which an optically active substance either Dextro or Levorotatory is di...
Q: 9. three standard cel potential of the reaction below is 0.375 V. calculate the value off delta G in...
A: The provided standard cell potential (E°cell)= 0.375V The provided reaction is- A(s) + 2B+(aq) -->...
Q: If a reaction produces fewer gas molecules than it consumes, ΔSº <0. True or False
A: The given statement is, If a reaction produces fewer gas molecules than it consumes, ΔSº <0.
Q: 7
A: First of all by using half life period, we can calculate the the rate constant k and then by using r...
Q: The Kp for the following reaction is 0.036 at 385 ºC, what is Kc? PCl5(g)PCl3(g)+Cl2(g)
A:
Q: A zinc bar weighing 3000g is attached to a buried iron pipe to protect the pipe from corrosion. An a...
A:
Q: Solve all
A:
Q: Calculate the moles of 02 that will be prepared through the decomposition (see balanced reaction) of...
A: Given: Number of moles of potassium chlorate are 4.7 moles. Introduction: Write the balanced reactio...
Q: According to the second law of thermodynamics, the change in the entropy of the universe (detla S un...
A: According to the second law of thermodynamics, The change in the entropy of the universe (detla S un...
Q: QUESTION 16 Choose all of the statements that are false. O When an intermediate compound is formed, ...
A: The false statement is, The rate determining step is the rate of the fastest elementary step in the...
Q: What is the concentration of X in a 0.150 M solution of the diprotic acid H,X ? For H,X K = 4.5 x10 ...
A: Given : Concentration of H2X = 0.150 M Ka1 of H2X = 4.5 X 10-6 And Ka2 of H2X = 1.2 X 10-11 ...
Q: When the d-orbitals of the central metal ion are split in a square planar crystal field, which orbit...
A:
Q: 21. Which of the following is the correct reaction intermediate? HCI, CH3OH Meo OMe intermediate НО ...
A: This is a cyclic acetal reaction. The ketone reacts with alcohol in acidic medium to form the cyclic...
Q: What volume of 8.50 M NaCl solution needs to be diluted to produce 400.0 mL of 2.00 M NaCl solution?
A: In case of dilution of any electrolyte, the following relation is used : MiVi = MfVf
Q: Closest-packed structures have a packing efficiency of 74% and a contact number of 12 and are thus m...
A:
Q: Solve all
A: Alkynes on treatment with H2 and Pd/BaSO4 produces cis alkene. A compound containing two or more chi...
Q: 75. Draw structure for (S,E)-4,5-dimethyl-4-hepten-3-ol:
A:
Q: Number of atoms in:a. nitrogen atoms in 25.0 g of TNT, C7H5N3O6
A: Trinitrotoluene, or 2,4,6-trinitrotoluene (TNT),is a chemical compound with the formula C7H5N3O6 is...
Q: QUESTION 17 The Kp for the following reaction is 0.035 at 387 °C, what is Ke? PCI (g) PCI (g) + Cl,(...
A: Given that the equation is PCl5 -----> PCl3 + Cl2 Kp = 0.035
Q: 37
A: Given: Initial moles of N2O4 = 0.0439 mol Volume of the flask = 1.91 L Equilibrium moles of N2O4 = 0...
Q: aon is necessary for these transformations.) a) HO- b) HO- OCH3
A: The answer to the following question is-
Q: Identify the correctly classified ionic compounds. Compound Solubility 1. Hg(NO3)2 insoluble ...
A: The correctly classified ionic compounds is given by following – Hg(NO3)2 – Soluble VBr3 – Soluble...
Q: Explain why real gases become less ideal at high pressures and low temperatures in the context of in...
A: We will explain which is required
Q: A sample of argon gas at a pressure of .924 atm and a temperature of 20.2 degrees Celsius, occupies ...
A: Given for argon gas P1 = 0.924 atm T1 = 20.2 °C = (20.2 + 273)K = 293.2 K V1 = 437 mL Temperature is...
Q: Solve all
A:
Q: Define the following terms:a. GSHb. mercapturic acidc. CSEd. CBSe. g-glutamyl cycle
A: The five basic types of chemical reactions occuring in chemistry are combination, decomposition, sin...
Q: Find the temperature (in K) above which a reaction with a ΔH of 170.8 kJ/mol and 64.5 ΔS J/K·mol bec...
A:
Q: Which one of the following reactions belongs to an exothermic reaction? A. The ionization of potass...
A: Exothermic reaction : In a reaction when products are more stable than reactants,the change in entha...
Q: When acetyl chloride, CH3COCl (shown below) dissolves in HF, what is the dominant solute-solvent mix...
A: It form hydrogen bonding with HF Hydrogen bonding is formed when Hydrogen is bonded with more electr...
Q: What are the molecular geometry, bond angle, and bond lengths of beta-carotene?
A: We will write the required information
Q: 5
A: Depression in freezing point can be determined as below : ∆ Tf = Kf. m Where ∆ Tf = depression in f...
Q: Briefly explain the concept of dynamic equilibrium using the relationship between a dissolved gas an...
A: explanation - the solubility of gases increases as the partial pressure of the gas above a solution...
Q: Please answer very soon will give rating surely
A: Moles of C6H6 = 35.2/78 = 0.4513 mol Moles of Br2 = 67.9/160 = 0.4244 mol By stoichiometry of react...
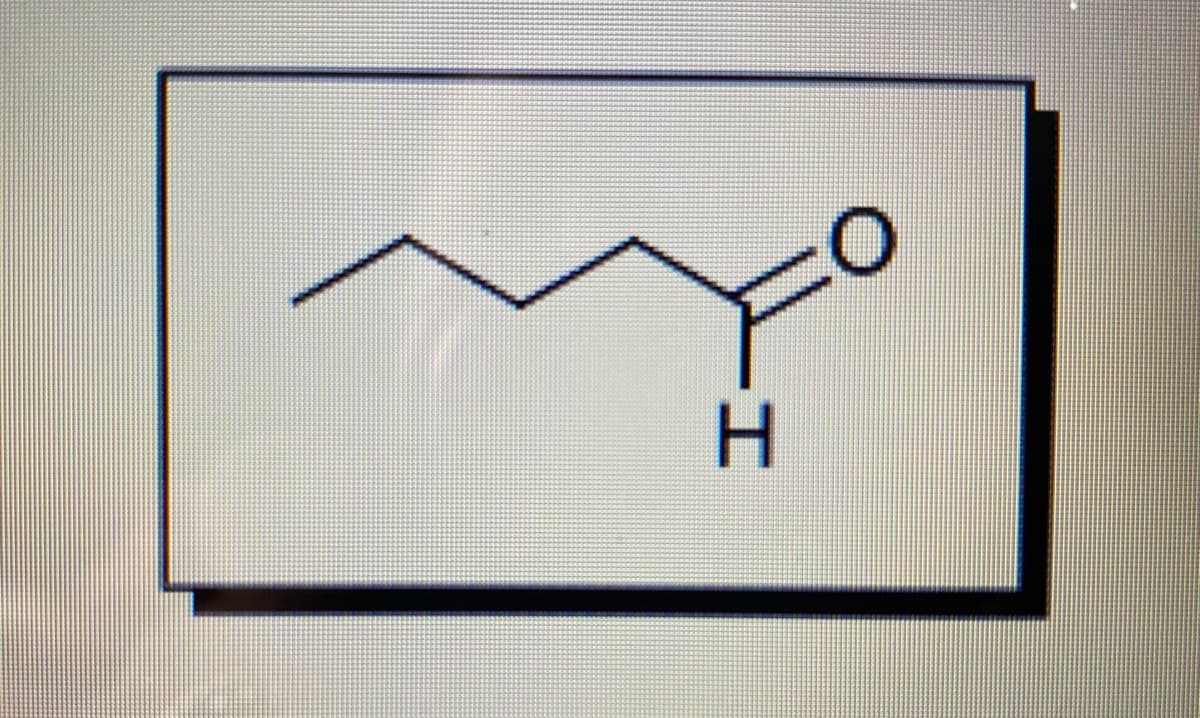
Step by step
Solved in 2 steps with 2 images


