Q: Which C5H12 isomer will have the highest boiling point? O Y
A: Alkanes are non-polar molecules and the interactions between molecules are the relatively weak van…
Q: Increasing the temperature of an exothermic reaction increases the Keq value. True False
A:
Q: Fill in the element flowchart for Si: HCI A Si NaOH CH3CI Na₂CO3 SiO₂ HF C H*
A:
Q: 42 g of a compound contain 36 g of Carbon and 6 g of Hydrogen. Which of the following is a plausible…
A: Empirical formula can be determined on the mass of each element present in a compound. Empirical…
Q: The Ka of the conjugate acid of a pesticide is 6.7 x 10-6. What is the Kb of the pesticide at 25°C?…
A: The product of the acid dissociation constant of an acid and the base dissociation constant of a…
Q: 95.24 mL 24.0 °C Barometric pressure 739 mmHg Vapor pressure of water at 24.0 °C 22.5 mmHg Calculate…
A: Total Volume of H2 gas collected = 95.24 mL or ( 95.24 /1000 ) L Temperature, T = 24°C or 297 K…
Q: Write the systematic name of each organic molecule: structure o & name 0 0 0 X
A: Select the longest chain. Please add prefix as cyclo if the compound is cyclic as main chain. Write…
Q: for the first order reaction which plot is linear (listed as Y,X) (see image)
A: Linear plot for 1st order reaction.
Q: Math Example Calculate In (74.10). In(74.10) = Calculate e7.98 €7.98= Chemistry Example A freshly…
A: ln(74.10) = 4.31 e7.98=2.92×103
Q: What is the total change in entropy of the universe (system + surroundings) if 3.5 g of CO(g) reacts…
A:
Q: When a lead acid car battery is recharged by the alternator, it acts essentially as an electrolytic…
A: Current = 10.0 A Time = 12 seconds
Q: the standard cell potential (E°cell) for the voltaic cell based on the reaction below is what?…
A: Oxidation: It involves loss of electrons or increase in oxidation number. It takes place at anode.…
Q: If the pOH of an acidic solution is 12.69, what is the [H3O+ (aq)], in mol/L?
A: A numerical problem based in equilibrium concept. An acidic solution with pOH prescribed, whose…
Q: How many valence d electrons does the Pd+³ ion have? Enter only a number for your answer. Do not…
A:
Q: Consider the reaction below and the proposed mechanism. X2 + Y + Z → XY + XZ Step l) X2 +…
A: The relation between the concentration of the reactants and the rate of reaction is expressed by the…
Q: Calculate ∆G (in kJ) at 35.4oC for a reaction that has Keq = 119
A: Using Keq we can calculate ∆G at particular temperature.
Q: at elevated temps dinitrogen pentoxide decomposes to nitrogen dioxde and oxygen 2N2O5(g) -->…
A:
Q: What is the percent strength (% w/v) of a 40 mOsmol/L solution of MgCl₂? (MW 95.21) *Report your…
A: Molarity of NaF = 8 mmol/l Volume of NaF = 3 ml = 3/1000 = 0.003 lts (1000 ml = 1 lt) Number of…
Q: Which of the following Bronsted-Lowry conjugate pairs is/are CORRECT? I. CH3NH3+ – CH3NH2 II.…
A: Conjugate acid base pair: The acid-base pair which differ by one ionizable hydrogen is called…
Q: G) R) ОН LIAI(OC(CH3)3)H -78°C 'H
A: Organic Reactions: G) Acid halides can be reduced to aldehydes through use of the bulky reducing…
Q: 2. Provide the reagent(s) necessary to achieve the following reactions. لسلة a. b. سلام HO' OH ستر…
A:
Q: Consider the apparatus shown in the figure. The container on the left is 22.4 L in volume and…
A: The concept used in this question is from chemical thermodynamics of physical chemistry. Here we…
Q: Consider the ethylene molecule C2H4. This molecule is planar and its Lewis structur is shown X C=C…
A: Given molecule is ethylene . Ethylene is two carbon alkene . IUPAC name of ethylene is ethene.
Q: What is the volume occupied by 3.01×1023 atoms of Helium a temperature of 0°C and a pressure of 1.00…
A: mole is equal to number of atom divide by avagadro number
Q: Acetic acid’s pKa is 4.75 and nitrous acid’s pKa is 3.16. If you made one buffer of equal parts…
A: Given: PKa of acetic acid = 4.75 PKa of nitrous acid = 3.16
Q: A galvanic cell at a temperature of 25.0 °C is powered by the following redox reaction: 2+ 2+ Cu²+…
A: Temperature = 25°C = 298 K Concentration of Cu+2 = 2.40 M Concentration of Zn+2 = 0.238 M Cell…
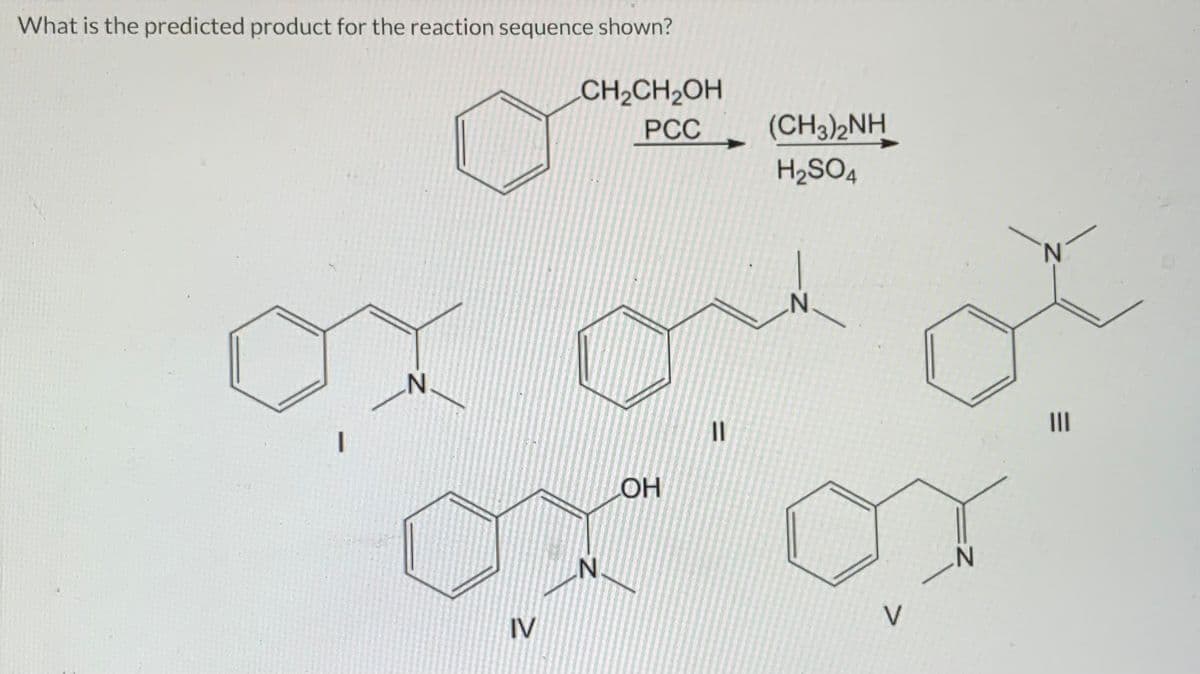
Q: What is the predicted product of the reaction sequence shown? AICI3 OCH3 IV NaBH4/CH3OH ОН || V ОН
A: NABH4 is reducing agent and it reduces ketone functional group into secondary alcohol
Q: What is the best technical name for a covalent compound having the chemical formula, SCI?
A: Covalent compound is formed by sharing of electron between atoms to complete their octet.
Q: Consider the mixing of 25.00 mL of 0.0500 M Pb(NO3)2 with 25.00 mL of 0.0500 M NaCl and the…
A:
Q: 3. Regarding the dissociation constant of electrolytes: Pk a = 7 A. The higher the pK, of a weak…
A: As per Arrhenius concept, acids produce the hydrogen ion when dissolved in water and bases produce…
Q: 4. Identify all types of intermolecular forces that are presents in the following substances. a. NH3…
A: We have been given four molecules and we have been asked to identify all types of intermolecular…
Q: the half life of a first order reaction is 13 min if the initial concentration of reactant is 0.085…
A:
Q: Write the equation for the beta-minus decay of phosphorus-32.
A: There are 2 types of beta decay- 1- beta plus decay- here positron is released 2- beta minus decay…
Q: Draw the structure of the following compounds: (1) (2S,4S)-2-ethyl-4-hydroxycyclopentan-1-one (2)…
A:
Q: Write a balanced equation for the positron emission of the following: a. 150 8 b. 67Ga 31 e. 67Zn 30…
A: Positron emission :Positron is the anti-particle of electron, in other words an electron having +1…
Q: Define atomic size. Give its unit of measurement. In the modern periodic table what trend is…
A: We have been asked to define atomic size and its unit of measurement.We have also been asked about…
Q: For the following organic reaction identify the mystery reactant R, its stoichiometric coefficient…
A: In reaction when atom or group of atoms are added to form large molecules are said to be addition…
Q: Give a reasonable mechanism for this reaction. Explain why it is exothermic. Н+ Дон H2O
A: Organic reactions are those in which organic reactant react to form organic products. In the given…
Q: Calculate the solubility at 25 °C of CuBr in pure water and in a 0.0090M CoBr₂ solution. You'll find…
A:
Q: 2. Calculate the number of moles and grams from the following: (use correct sig figs in answer)…
A: The molar mass of atoms of an element is given by the standard relative atomic mass of the element…
Q: In basic medium, cyanamide reacts with itself (it dimerizes) to form dicyanamide whose Lewis…
A: Levis dot structure represent the diagram of covalent compound (polar and nonpolar) where electrons…
Q: Is cholesterol an inherently "bad" biomolecule? a. No, as it is an essential major component of the…
A: Since you have asked multiple question, we will solve the first question for you.If you want any…
Q: A 25.0g block of iron is heated from 25.0 C to 85.0 C. How much heat (in J) was added?
A: Given : m = 25 g T1 = 25°C T2 = 85°C Specific heat of iron = c = 0.45 J/g°C Heat added = q = ? J
Q: When you perform a certain reaction, 5.780 g of product is made. If you calculate a percent yield of…
A:
Q: Which of the following statements is/are TRUE regarding solubility equilibria? I. It is an…
A: Solubility equilibrium is the equilibrium that exist between a solid compound and it's saturated…
Q: Arrange the following alkyl halides based on their reactivity (from fastest to slowest) towards…
A: According to the question, The compound I → 1-bromobutane The compound II→ 2-bromobutane Compound…
Q: The following is a diagram of energy states and transitions in the hydrogen atom. ENERGY A B C D E…
A: Here, we have to find the emission line with the longest wavelength, the absorption line with the…
Q: 12. H₂C H₂C 1000 4000 11 10 CH₂ H₂C HSP-03-214 CH3 3000 NMR Spectrum CH₂ 3000 T 9 H₂C 8 2008 CH3OH…
A:
Q: eats of formation given in the table: ! Formula C₂H2(g) 226.7 NH3(g) -46.19 C6H6(1) 49.0 ate…
A:
Q: What volume of a 0.600 M HCl solution is required to react completely with 2.50 g of sodium hydrogen…
A: Given : Mass of sodium hydrogen carbonate = 2.50 g Concentration of HCl = 0.600 M Mass of CO2…
Step by step
Solved in 2 steps with 1 images

- (b) Consider the reaction of 1-bromobutane with a large excess of ammonia (NH3). Draw the reactants, the transition state, andthe products. Note that the initial product is the salt of an amine (RNH3+ Br - ), which is deprotonated by the excess ammonia to give the amine.Provide the mechanism for the conversion shown below. CH3Li is effectively H3C-Li+. What type of mechanism is occuring?Which product is the final product in the reaction below, shown as numbered?
- What kind of mechanism would this reaction follow?Draw the reactant RCHDBr with S absolute configuration, the draw the product of R’OH + RCHDBr (with S-configuration) through an SN2 reactions.What happens to the rate of an SN1 reaction under the following conditions? [RX] is halved, and [:Nu−] stays the same

