January 10, 2017 Jane Doe and John Doe Experiment 1: Density, Graphing, Accuracy and Precision Sample # 1 2 3 4 Initial Buret Reading 12.40 14.20 15.98 18.08 Final Buret Reading 14.20 15.98 18.08 20.08 mL Difference 1.80 1.78 2.10 2.00 Cumulative Volume 1.80 3.58 5.68 7.68 Dry Cap + Vial 4.935 g After Adding Sample 6.7081 8.5443 10.7027 12.8074 Difference 1.7731 3.6093 5.7677 7.8724
January 10, 2017 Jane Doe and John Doe Experiment 1: Density, Graphing, Accuracy and Precision Sample # 1 2 3 4 Initial Buret Reading 12.40 14.20 15.98 18.08 Final Buret Reading 14.20 15.98 18.08 20.08 mL Difference 1.80 1.78 2.10 2.00 Cumulative Volume 1.80 3.58 5.68 7.68 Dry Cap + Vial 4.935 g After Adding Sample 6.7081 8.5443 10.7027 12.8074 Difference 1.7731 3.6093 5.7677 7.8724
Chapter5: Errors In Chemical Analyses
Section: Chapter Questions
Problem 5.12QAP
Related questions
Question
100%
can you please explain how we get the cumulative volume and what is it? The image below is just for reference. It shows the cumulative volume. But do we obtain it
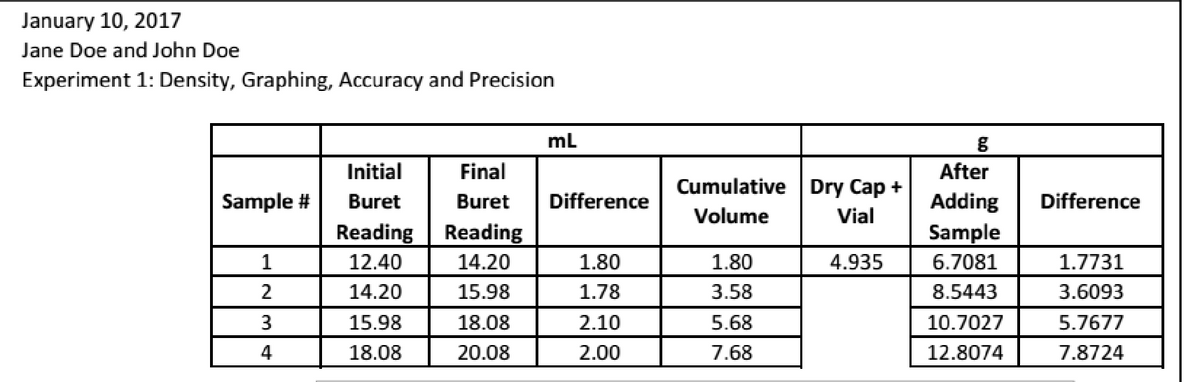
Transcribed Image Text:January 10, 2017
Jane Doe and John Doe
Experiment 1: Density, Graphing, Accuracy and Precision
Sample #
1
2
3
4
Initial
Buret
Reading
12.40
14.20
15.98
18.08
Final
Buret
Reading
14.20
15.98
18.08
20.08
mL
Difference
1.80
1.78
2.10
2.00
Cumulative
Volume
1.80
3.58
5.68
7.68
Dry Cap +
Vial
4.935
After
Adding
Sample
6.7081
8.5443
10.7027
12.8074
Difference
1.7731
3.6093
5.7677
7.8724
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

