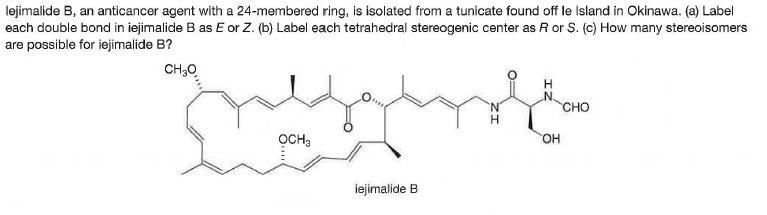
lejimalide B, an anticancer agent with a 24-membered ring, is isolated from a tunicate found off le Island in Okinawa. (a) Label each double bond in iejimalide B as E or Z. (b) Label each tetrahedral stereogenic center as R or S. (c) How many stereoisomers are possible for iejimalide B? CH;0 CHO OCH, HO, iejimalide B
Q: Neglecting stereoisomers, four different a,ß-unsaturated aldehydes are produced when the aldehydes…
A:
Q: Draw a stepwise mechanism for the reduction of epoxide A to alcohol B using LiAlH4. What product…
A: Stepwise mechanism for the reduction of epoxide A to alcohol B: In the first step, the hydride…
Q: What product is formed when (CH3C=CHCH,COCH2CH,CO,CH,CH3 is treated with each reagent: (a) H2 (1…
A: a) Catalytic hydrogenation of alkene gives alkanes. b) two mole of hydrogen in the presence of…
Q: Hydroxy aldehydes A and B readily cyclize to form hemiacetals. Draw the stereoisomers formed in this…
A: Hemiacetal is a compound which is generated by the addition of alcohol to the aldehyde. Another…
Q: Consider the para-substituted aromatic ketones, NO,CeH,COCH3 (p-nitroacetophenone) and…
A: The structure of p-nitro acetophenone is; The structure of p-methoxy acetophenone: Carbonyl…
Q: Iejimalide B, an anticancer agent with a 24-membered ring, is isolated from a tunicate found off Ie…
A: How to find E and Z-configuration? First of all look out for the double bonds in the structure and…
Q: The shrub ma huang (Section 5.4A) contains two biologically activestereoisomers—ephedrine and…
A: Since you have posted a question with multiple sub-parts we will solve the first three sub-parts for…
Q: Rank the anions in order of increasing nucleophilicity in acetone: CH3S−, CH3NH−, I−, Br−, and…
A: Among CH3NH-, CH3S-, CH3O- , I- and Br- acetone.
Q: Draw the product(s) produced (only constitutional isomers, disregard stereoisomers) when the…
A:
Q: Orsellinic acid, a common constituent of lichens, is synthesized from the condensation of acetyl…
A:
Q: What product is formed when the esters in ginkgolide B, the chapter-opening molecule, are hydrolyzed…
A: The hydrolysis of esters in the presence of acid leads to the formation of carboxylic acids and…
Q: What product is formed by the [3,3] sigmatropic rearrangement of eachcompound?
A:
Q: One step in the synthesis of the nonsteroidal anti-inflammatory drug rofecoxib (trade name Vioxx)…
A: One step in the synthesis of the nonsteroidal anti-inflammatory drug rofecoxib (trade name Vioxx)…
Q: Ammonia and amines react with epoxides with the same stereospecificity as anionic nucleophiles. Draw…
A:
Q: Heating compound X with aqueous formaldehyde forms Y (C17H23NO), which has been converted to a…
A: The above reaction indicates that lone pairs of electron present on the nitrogen atom of given…
Q: Treatment of compound A (C8H17Br) with NaOCH2CH3 affords twoconstitutional isomers B and C.…
A:
Q: (a) Draw all products formed by treatment of each dibromide (A and B) with one equivalent of NaNH2.…
A: (a). The products formed by treatment of each dibromide (A and B) with one equivalent of NaNH2:…
Q: Ethanal is treated with CyclohexylMgCl/H2O and resulting product is dehydrated by Conc.H2SO4.
A: Answer:
Q: When (ft)-6-bromo-2,6-dimethylnonane is dissolved in CH3OH, nucleophilic substitution yields an…
A: NOTE : there is a typing error in the question its (6R)-6-bromo-2,6-dimethylnonane and…
Q: What reagent is needed to convert (CH3)2CHCH2COCl to each compound?
A: a. The given compound is aldehyde. Acid chlorides on reaction with mild reducing agent like lithium…
Q: Atorvastatin (Lipitor) is used to decrease patient serum cholesterol levels. It works by inhibiting…
A:
Q: When (R)-6-bromo-2,6-dimethylnonane is dissolved in CH3OH, nucleophilic substitution yields an…
A: The structure of given compounds are shown below:
Q: Draw all of the substitution and elimination products formed from thegiven alkyl halide with each…
A: a.
Q: Consider the tetracyclic compound with rings labeled A–D. (a) Which ring is the most reactive in…
A: Benzene ring is activated towards electrophilic substitution reaction if there is an electron…
Q: Draw the products formed when p-methylaniline (p-CH3C6H4NH2) is treated with each reagent.a. HClb.…
A: a.
Q: When attached halohydrin is treated with NaH, a product of molecular formula C4H8O is formed. Draw…
A: In the given reaction, halohydrin is treated with NaH. When a halohydrin is treated with a base…
Q: When attached halohydrin is treated with NaH, a product of molecularformula C4H8O is formed. Draw…
A: In the given reaction, halohydrin is treated with sodium hydride (NaH). When a halohydrin is treated…
Q: The poison gelsemoxonine can be isolated from the leaves of a plant native to southeastern Asia…
A: Sigmatropic rearrangement: It is a pericyclic reaction in which a sigma bond migrates from one site…
Q: Explain each observation: (a) When (R)-2-methylcyclohexanone is treated with NaOH in H2O, the…
A:
Q: Q3\ The pKa of p-cyclopropylbenzoic acid is 4.45. Is cyclopropylbenzene likely to be more reactive…
A:
Q: An unknown compound A (molecular formula C7H14O) was treated withNaBH4 in CH3OH to form compound B…
A: The nuclear magnetic resonance spectroscopy or NMR deals with the study of magnetic properties of…
Q: (a) Draw all products formed by treatment of each dibromide (A and B) with one equivalent of NaNH2.…
A: (a). The products formed by treatment of each dibromide (A and B) with one equivalent of NaNH2:…
Q: Consider the para-substituted aromatic ketones, NO2C6H4COCH3 (p-nitroacetophenone) and CH3OC6H4COCH3…
A:
Q: 5-Hydroxyhexanal forms a six-membered cyclic hemiacetal, which predominates at equilibrium in…
A: Note: “Since you have posted a question with multiple sub-parts, we will solve first three sub-parts…
Q: When each halohydrin is treated with NaH, a product of molecular formula C4H8O is formed. Draw the…
A: A functional group where both a halogen group and a hydroxyl group are attached to the same carbon…
Q: b) OH Hemiacetal H3C D Compound D could be converted into a cyclic hemiacetal in the presence of an…
A: Aldehydes on treatment with an alcohol in the presence of an acid produces hemiacetal.
Q: When (R)-6-bromo-2,6-dimethylnonane is dissolved in CH3OH,nucleophilic substitution yields an…
A: The structure of given compounds are shown below:
Q: Which stereoisomer is obtained in the following intramolecular reaction? BrMg Br a. b. d. C. **** I,
A:
Q: When attached halohydrin is treated with NaH, a product of molecular formula C4H8O is formed. Draw…
A: When attached halohydrin is treated with NaH, a product of molecular formula C4H8O is formed. The…
Q: 3.1) Isomers A and B undergo elimination reaction when treated with NaOEt. OTS OTs H3CH,C "CH2CH3…
A:
Q: Quinapril (trade name Accupril) is used to treat high blood pressure andcongestive heart failure.…
A: The reaction mechanism along with the products is given below.
Q: What product is formed when each compound undergoes thermal electrocyclic ring opening or ring…
A: Incase of electrocyclic reactions, one of pi-bond of conjugated double bond is converted to ring…
Q: β-Vetivone is isolated from vetiver, a perennial grass that yields a variety of compounds used in…
A: a.
Q: The pKa of p-cyclopropylbenzoic acid is 4.45 . Is cyclopropylbenzene likely to be more reactive or…
A:
Q: Ibufenac, a para-disubstituted arene with the structure HO2CCH26H4CH2CH(CH3)2, is a much more potent…
A: The molecular formula of given compound is: Its structure is:
Q: MENH2 SO3ME MеHN. NEC- CI NEC a. Draw a reasonable arrow-pushing mechanism for this transformation.…
A: There will be removal of HCl.
Q: Give the IUPAC name for each compound, including the R,S designation for each stereogenic center. a.…
A: The IUPAC name of the following compounds are:
Q: A key step in the synthesis of the narcotic analgesic meperidine (tradename Demerol) is the…
A: The total transformation mechanism is given below -
Q: Consider the reaction scheme below NBS hv a. Draw the major regioisomer resulting from…
A:
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 5 images

- A key step in the synthesis of the narcotic analgesic meperidine (tradename Demerol) is the conversion of phenylacetonitrile to X. (a) What isthe structure of X? (b) What reactions convert X to meperidine ?(R)-Carvone, the major component of the oil of spearmint, undergoesacid-catalyzed isomerization to carvacrol, a major component of the oilof thyme. Draw a stepwise mechanism and explain why thisisomerization occurs.Treatment of compound A (C8H17Br) with NaOCH2CH3 affords two constitutional isomers B and C. Ozonolysis of B affords CH2=O and (CH3CH2CH2)2C=O. Ozonolysis of C affords CH3CH2CH2COCH3 and CH3CH2CHO. What is the structure of A?
- When (R)-6-bromo-2,6-dimethylnonane is dissolved in CH3OH,nucleophilic substitution yields an optically inactive solution. When theisomeric halide (R)-2-bromo-2,5-dimethylnonane is dissolved in CH3OHunder the same conditions, nucleophilic substitution forms an opticallyactive solution. Draw the products formed in each reaction, and explainwhy the difference in optical activity is observed.(-)-Hyoscyamine, an optically active drug used to treat gastrointestinal disorders, is isolated from atropabelladonna, the deadly nightshade plant, by a basic aqueous extraction procedure. If too much base is used during isolation, optically inactive material is isolated A) explain this result by drawing a stepwise mechanism(a) Explain how NaBH4 in CH3OH can reduce hemiacetal A to butane-1,4-diol (HOCH2CH2CH2CH2OH). (b) What product is formed when A is treated with Ph3P = CHCH2CH(CH3)2? (c) The drug isotretinoin is formed by reaction of X and Y. What is the structure of isotretinoin? Although isotretinoin (trade name Accutane or Roaccutane) is used for the treatment of severe acne, it is dispensed under strict controls because it also causes birth defects.
- The shrub ma huang (Section 5.4A) contains two biologically activestereoisomers—ephedrine and pseudoephedrine—with two stereogeniccenters as shown in the given structure. Ephedrine is one component ofa once-popular combination drug used by body builders to increaseenergy and alertness, whereas pseudoephedrine is a nasaldecongestant.a.) Draw the structure of naturally occurring (−)-ephedrine, which has the1R,2S configuration.b.) Draw the structure of naturally occurring (+)-pseudoephedrine, whichhas the 1S,2S configuration.c.) How are ephedrine and pseudoephedrine related?d.) Draw all other stereoisomers of (−)-ephedrine and (+) pseudoephedrine, and give the R,S designation for all stereogeniccenters.e.) How is each compound drawn in part (d) related to (−)-ephedrine?Friedel–Crafts alkylation of benzene with (R)-2-chlorobutane and AlCl3 affords sec-butylbenzene.a. How many stereogenic centers are present in the product?b. Would you expect the product to exhibit optical activity? Explain, with reference to the mechanism.Ibufenac, a para-disubstituted arene with the structureHO2CCH2C6H4CH2CH(CH3)2 , is a much more potent analgesic thanaspirin, but it was never sold commercially because it caused livertoxicity in some clinical trials. Devise a synthesis of ibufenac frombenzene and organic halides having fewer than five carbons.
- Although γ-butyrolactone is a biologically inactive compound, it isconverted in the body to 4-hydroxybutanoic acid (GHB), an addictive andintoxicating recreational drug. Draw a stepwise mechanism for thisconversion in the presence of acid.Ibufenac, a para-disubstituted arene with the structure HO2CCH26H4CH2CH(CH3)2, is a much more potent analgesic than aspirin, but it was never sold commercially because it caused liver toxicity in some clinical trials. Devise a synthesis of ibufenac from benzene and organic halides having fewer than five carbons.β-Vetivone is isolated from vetiver, a perennial grass that yields a variety of compounds used in traditional eastern medicine, pest control, and fragrance. In one synthesis, ketone A is converted to β-vetivone by a two-step process:Michael reaction, followed by intramolecular aldol reaction. (a) What Michael acceptor is needed for the conjugate addition? (See Problem 23.61 for another method to form the bicyclic ring system of β-vetivone.) (b) Draw a stepwise mechanism for the aldol reaction, which forms the six-membered ring.

