Q: Explain why Jone's oxidation of 4-methyl-2-pentanol is faster than that of 3,3-dimethyl-2-pentanol.
A:
Q: How many unique products (including regioisomers and stereoisomers) could be produced with the…
A: Alkyl halide prefers E1 elimination under heating conditions in polar solvents. Three products were…
Q: ccording to Zaitsev's rule, the major product of an elimination is the, substituted alkene. O most D…
A:
Q: 8.46 Draw the structure (including stereochemistry) of an alkyl chloride forms each alkene as the…
A:
Q: Draw a stepwise mechanism for the attached reaction, which involves two Friedel–Crafts reactions. B…
A: Various types of mechanisms are studied in organic chemistry. One of them is electrophilic…
Q: Rank the attached alkenes in order of increasing stability.
A: The given alkenes are represented as follows:
Q: Draw the product(s) produced (only constitutional isomers, disregard stereoisomers) when the…
A:
Q: Hydroxy aldehydes A and B readily cyclize to form hemiacetals. Draw the stereoisomers formed in this…
A: The formation of a cyclic hemiacetal is an acid catalyzed reaction. The cyclic hemiacetals formed by…
Q: Draw a stepwise mechanism for the following reaction, a key step in the synthesis of the…
A: Answer : Step in the synthesis of the anti-inflammatory drug Celebrex is :
Q: What is Hofmann elimination?
A: Hofmann Elimination:The quaternary ammonium salts gets converted into tertiary amine and alkene on…
Q: Draw the products (including stereochemistry) for attached reaction.
A: Please find below the reaction along with the stereochemistry. Since the attack is possible from…
Q: Which compound in each pair forms the higher percentage of gem-diol at equilibrium?
A: Carbonyl compound reacts with water in presence of acid or base forms gem-diol or hydrate. The yield…
Q: Draw a stepwise mechanism for the following substitution. Explain why 2-chloropyridine reacts faster…
A: Given reaction,
Q: Draw a stepwise mechanism for the following Robinson annulation. This reaction was a key step in a…
A:
Q: Name attached compound and decide which stereoisomer will react faster inan E2 elimination reaction.…
A: E2 elimination – This is a one step mechanism. Here two bonds C-H and C-X bonds break simultaneously…
Q: Label the alkene in each attached drug as E or Z. Enclomiphene is one component of the fertility…
A: If the relatively higher priority groups are on the same side then it is a (Z)-alkene. If they are…
Q: Click the "draw structure" button to launch the drawing utility. What product(s) (excluding…
A:
Q: Which ring in each compound is more reactive toward electrophiles?
A: The species that are electron deficient can be defined as electrophiles. Since, they have less…
Q: Alkene A can be isomerized to isocomene, a natural product isolated from goldenrod, by treatment…
A: The mechanism is given below.
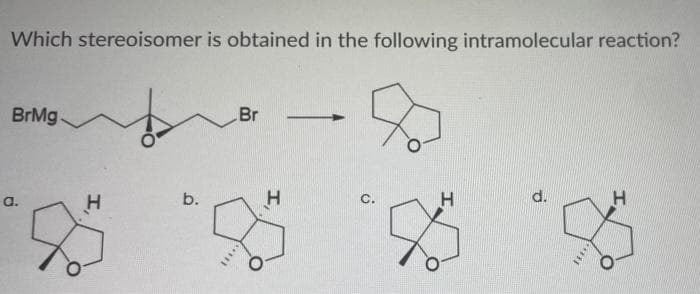
Q: How many stereoisomers are obtained from each of the syntheses described
A: The carbon center to which the four groups attached are different from each other, then that carbon…
Q: "green" oxidation The above reaction is one of example of environmental benign synthesis which does…
A: Green oxidation requires use of hydrogen peroxide and oxidation.
Q: B A OH Compound A can be converted to cyclobutanol using the reagent Compound B can be converted to…
A: Organic chemistry in which we deal with organic reactions which covert organic reactant in to…
Q: Treatment of α,β-unsaturated carbonyl compound X with base forms the diastereomer Y. Write a…
A: In the reaction, racemization of a methyl group linked to gamma -carbon atom takes place. For…
Q: Which alkene in each pair has the larger heat of hydrogenation?
A: The given pair of alkenes are,
Q: Draw all constitutional isomers formed in attached elimination reaction.Label the mechanism as E2 or…
A: The base and the type of alkyl halide in a given reaction determine the mechanism. In the given…
Q: Label the alkene in each drug as E or Z. Enclomiphene is one component of the fertility drug Clomid.…
A: a. The alkene in given drug Enclomiphene labelled as E or Z is shown below,
Q: What acetylide anion and alkyl halide can be used to prepare each alkyne? Indicate all possibilities…
A: Hydrocarbons carrying C-C triple bond are designated as "alkynes". The common formula used for…
Q: Select the member of each pair that undergoes SN1 solvolysis in aqueous ethanol more rapidly
A: In SN1, the rate determining step is formation of carbocation and order of the substrate that…
Q: When a carbonyl compound is attacked by a nucleophile resulting in the formation of a new…
A: In the carbonyl compound nucleophilic addition reaction occurs.
Q: How can pentan-2-one be converted to each attached compound?
A: (a) (b)
Q: a) b)
A: Applying reagent and suitable chemical reaction As LDA RMgX etc.
Q: Which compound in each pair absorbs UV light at longer wavelength?
A: In both these cases, the interesting aspect is to note the conjugation of the alkene moiety.…
Q: Label the alkene in each attached drug as E or Z. Enclomiphene is one component of the fertility…
A: Given molecule:
Q: What cyclic product is formed when each decatetraene undergoes thermal electrocyclic ring closure?
A:
Q: Select the member of each pair that undergoes SN1 solvolysis in aqueous ethanol more rapidly
A: Concept introduction: SN1 reaction: The displacement of atom or group by nucleophilic is known as…
Q: Draw a stepwise mechanism for the following reaction that forms ether D. D can be converted to the…
A: Since KOC(CH3)3 is a strong base. Hence it will take proton from the OH group and convert it into O-…
Q: Draw a stepwise mechanism for the following reaction that forms ether D. D can be converted to the…
A:
Q: What cyclic product is formed when each decatetraene undergoes photochemical electrocyclic ring…
A: a.
Q: Draw all constitutional isomers formed when X is treated with NBS + hv.
A: The products formed are as given below:
Q: Draw all products, including stereoisomers, in attached reaction.
A: The nucleophilic substitution reaction involves the removal of a group bonded to a carbon center…
Q: Draw the major organic product of the reaction. Ignore stereochemistry
A: First step is the formation of stable carbocation.
Q: Draw enol tautomer(s) for each attached compound. Ignore stereoisomers.
A: Introduction: Those compounds which the same molecular formula but differ in the position of protons…
Q: What is the stepwise mechanism
A: The reactions involving organic compounds are called organic reactions. Organic reactions are of…
Q: Draw the product formed (including stereochemistry) in attached pericyclicreaction.
A: Electrocyclic reactions are a part of pericyclic reactions that occur in the presence of heat or…
Q: Click the "draw structure" button to launch the drawing utility. What product(s) (excluding…
A: Here we have to determine the suitable reagent required to convert the given reactant to target…
Q: Which alkene reacts faster with HBr? Explain your choice.
A: Hydrogen bromide reacts with an alkene to form alkyl bromide. The reaction completes in two steps…
Q: III. Give name from structure: a) b)
A:
Q: Draw a stepwise mechanism for the attached substitution. Explain why 2-chloropyridine reacts faster…
A: In this substitution reaction, ethoxide ion will attack carbon which is bonded to chorine because…
Q: The triphenylmethyl cation is so stable that a salt such as triphenylmethyl chloride can be isolated…
A: The stability order of carbocation is 30 > 20 > 10.
Q: Label each heterocycle as aromatic, antiaromatic, or not aromatic.
A: The aromaticity of a molecule is governed by the following factors: Cyclic Planar Delocalization of…
3
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- Draw a stepwise mechanism for the following Robinson annulation. This reaction was a key step in a synthesis of the steroid cortisone by R. B. Woodward and co-workers at Harvard University in 1951.What products are obtained from the reaction of the attached compound with one equivalent of Br2, using FeBr3 as a catalyst?Draw all stereoisomers formed in each reaction.
- Draw the major organic product of the reaction. Ignore stereochemistryA key step in the synthesis of the narcotic analgesic meperidine (tradename Demerol) is the conversion of phenylacetonitrile to X. (a) What isthe structure of X? (b) What reactions convert X to meperidine ?(a) Draw a stepwise mechanism for the conversion of A to B. (b) What product would be formed if C was exposed to similar reaction conditions?

