Atorvastatin (Lipitor) is used to decrease patient serum cholesterol levels. It works by inhibiting an enzyme called HMG-COA reductase. See"Connections to Biological Chemistry" one synthesis of atorvastatin that produces the desired single enantiomer of the final product, the following reagents are used. Draw the structures of synthetic intermedi- ates A and B. In -Cl NaCN НО. A B DMSO Pyridine
Atorvastatin (Lipitor) is used to decrease patient serum cholesterol levels. It works by inhibiting an enzyme called HMG-COA reductase. See"Connections to Biological Chemistry" one synthesis of atorvastatin that produces the desired single enantiomer of the final product, the following reagents are used. Draw the structures of synthetic intermedi- ates A and B. In -Cl NaCN НО. A B DMSO Pyridine
Organic Chemistry
8th Edition
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Chapter10: Alcohols
Section: Chapter Questions
Problem 10.57P
Related questions
Question
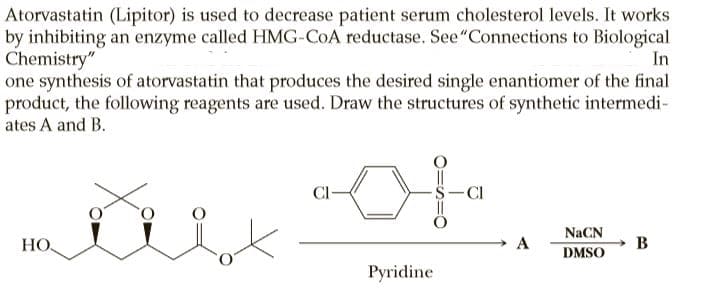
Transcribed Image Text:Atorvastatin (Lipitor) is used to decrease patient serum cholesterol levels. It works
by inhibiting an enzyme called HMG-COA reductase. See"Connections to Biological
Chemistry"
one synthesis of atorvastatin that produces the desired single enantiomer of the final
product, the following reagents are used. Draw the structures of synthetic intermedi-
ates A and B.
In
-Cl
NaCN
НО.
A
B
DMSO
Pyridine
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 3 images

Recommended textbooks for you

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning