Chapter22: Carbonyl Alpha-substitution Reactions
Section22.SE: Something Extra
Problem 57AP: All attempts to isolate primary and secondary nitroso compounds result solely in the formation of...
Related questions
Question
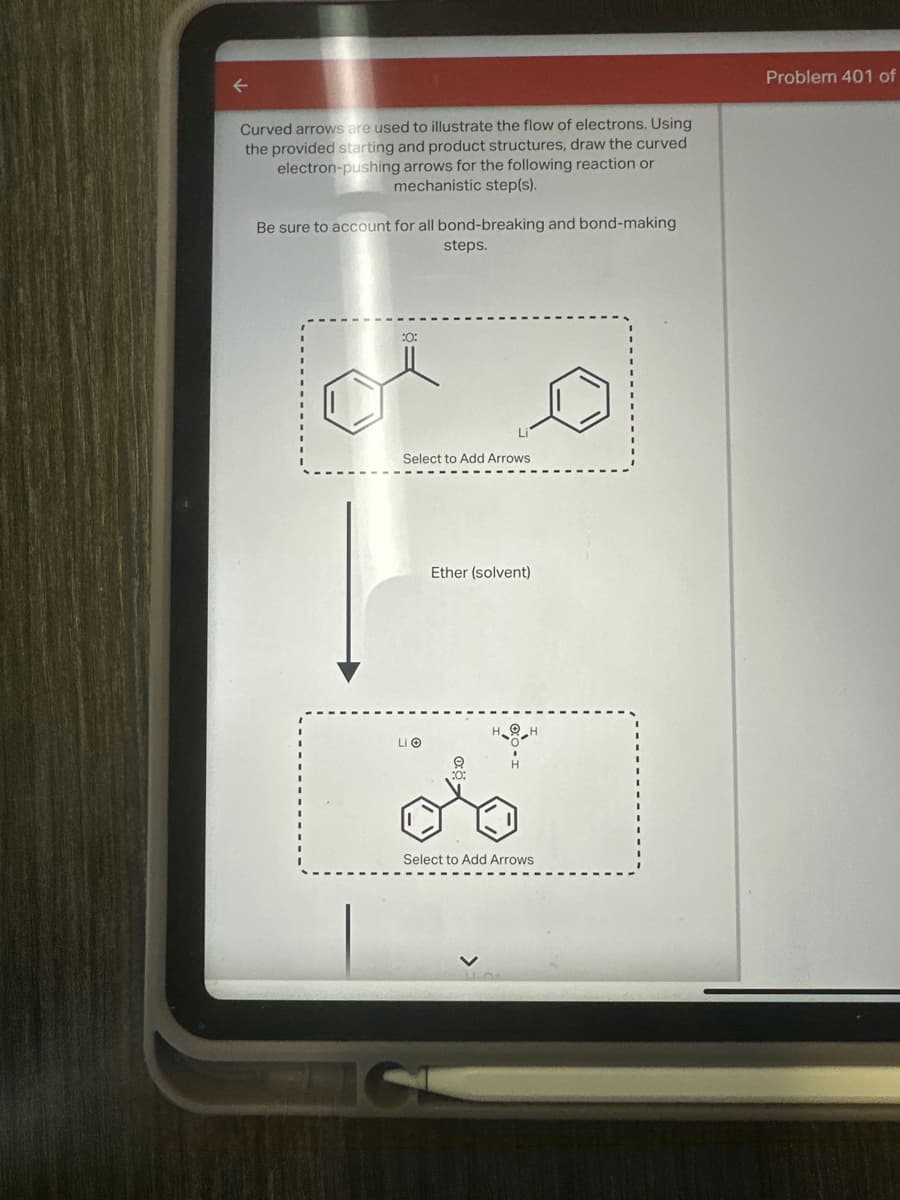
Transcribed Image Text:←
Curved arrows are used to illustrate the flow of electrons. Using
the provided starting and product structures, draw the curved
electron-pushing arrows for the following reaction or
mechanistic step(s).
Be sure to account for all bond-breaking and bond-making
steps.
Problem 401 of
:0:
Select to Add Arrows
Ether (solvent)
HOH
Li
Select to Add Arrows
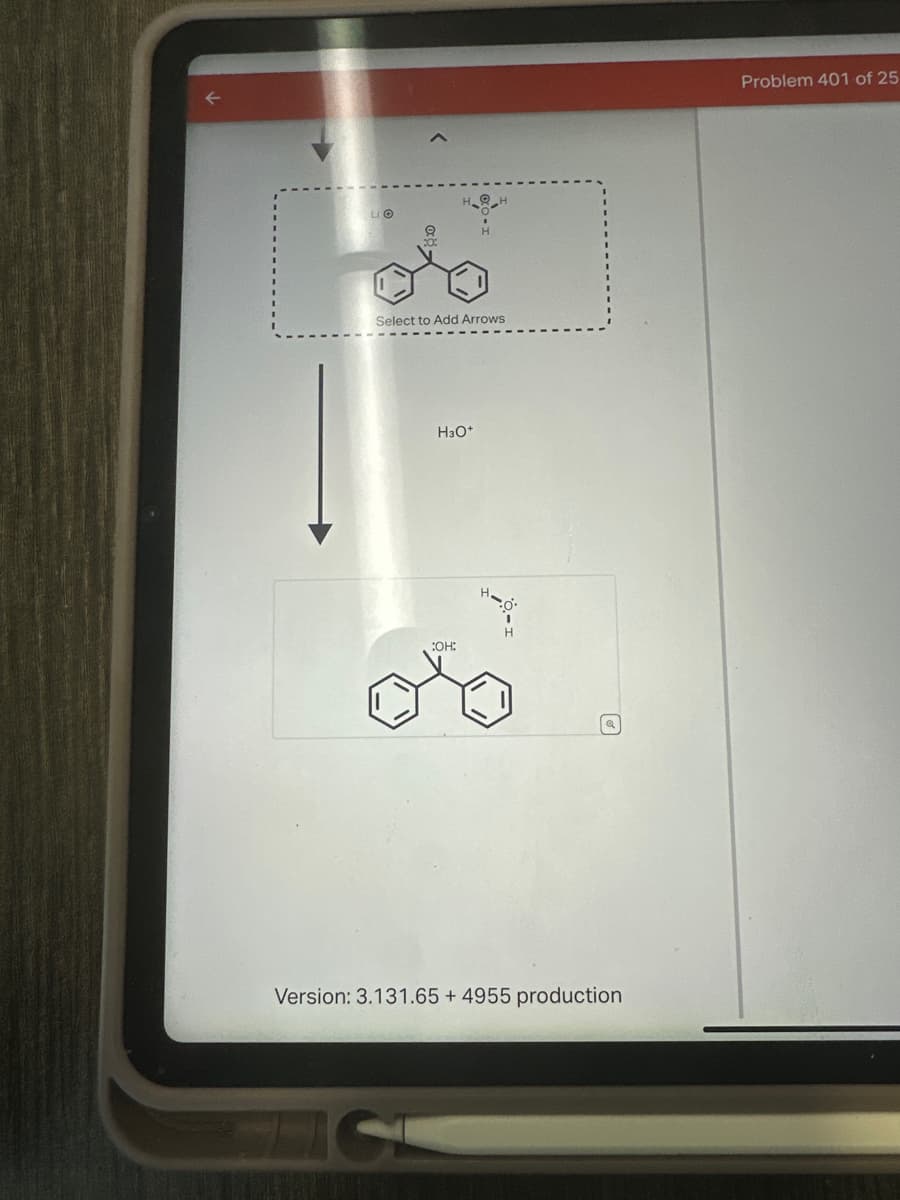
Transcribed Image Text:LIO
10:
H
Select to Add Arrows
H3O+
:OH:
Version: 3.131.65 + 4955 production
Problem 401 of 25
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

