
Organic Chemistry
9th Edition
ISBN: 9781305080485
Author: John E. McMurry
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 22.SE, Problem 57AP
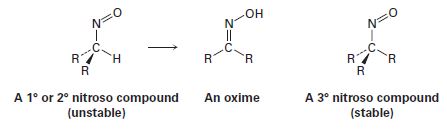
All attempts to isolate primary and secondary nitroso compounds result solely in the formation of oximes. Tertiary nitroso compounds, however, are stable. Explain.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
EXPLAIN BY WORDS IN ORDER FOR ME TO UNDERSTAND THE GENERAL REACTION SCHEME. IN THIS CROSS-COUPLING REACTION WHICH ORGANOHALIDE (R-X) REACTS WITH ORGANOBORANE [R1B(OR)2].
1)2PbS + 3O2 on heating gives 2PbO + 2SO2
2)PbS + 2O2 on heating gives PbSO4
Why is it that 2 reaction shows different products? Please explain in detail
In the early age of the development of Organometallilc Chemistry, the preparation of Organometallic Compound containing M-R(R: alkyl group) bond always led to the decomposition of the M-R bond. It was later known to be caused by the so called “beta-hydrogen elimination” process. Which of the following statements is not correct?
This process can be prevented by using all halogenated alkyl group.
This process can be prevented by carrying out reaction at high temperature.
The Grignard’s reagent, RMgX or R2Mg is stable and does not undergo “b-hydrogen elimination” process.
The anti-knocking agent, Pb(C2H5)4 is stable and does not undergo “b-hydrogen elimination” process.
The ferrocene, Cp2Fe, is free from the “b-hydrogen elimination” process.
Chapter 22 Solutions
Organic Chemistry
Ch. 22.1 - Prob. 1PCh. 22.1 - How many acidic hydrogens does each of the...Ch. 22.1 - Prob. 3PCh. 22.3 - Write the complete mechanism for the deuteration...Ch. 22.3 - Prob. 5PCh. 22.4 - If methanol rather than water is added at the end...Ch. 22.5 - Prob. 7PCh. 22.5 - Draw a resonance structure of the acetonitrile...Ch. 22.6 - If methanol rather than water is added at the end...Ch. 22.7 - Prob. 10P
Ch. 22.7 - Draw a resonance structure of the acetonitrile...Ch. 22.7 - Why do you suppose ketone halogenations in acidic...Ch. 22.7 - Prob. 13PCh. 22.7 - Prob. 14PCh. 22.7 - Prob. 15PCh. 22.7 - Prob. 16PCh. 22.SE - Prob. 17VCCh. 22.SE - Prob. 18VCCh. 22.SE - Prob. 19VCCh. 22.SE - Prob. 20MPCh. 22.SE - Predict the product(s) and provide the mechanism...Ch. 22.SE - Predict the product(s) and provide the mechanism...Ch. 22.SE - Prob. 23MPCh. 22.SE - In the Hell–Volhard–Zelinskii reaction, only a...Ch. 22.SE - Prob. 25MPCh. 22.SE - Nonconjugated , -unsaturated ketones, such as...Ch. 22.SE - Prob. 27MPCh. 22.SE - Using curved arrows, propose a mechanism for the...Ch. 22.SE - Prob. 29MPCh. 22.SE - One of the later steps in glucose biosynthesis is...Ch. 22.SE - The Favorskii reaction involves treatment of an...Ch. 22.SE - Treatment of a cyclic ketone with diazomethane is...Ch. 22.SE - Prob. 33MPCh. 22.SE - Amino acids can be prepared by reaction of alkyl...Ch. 22.SE - Amino acids can also be prepared by a two-step...Ch. 22.SE - Heating carvone with aqueous sulfuric acid...Ch. 22.SE - Identify all the acidic hydrogens (pKa 25) in the...Ch. 22.SE - Rank the following compounds in order of...Ch. 22.SE - Prob. 39APCh. 22.SE - Base treatment of the following , -unsaturated...Ch. 22.SE - Prob. 41APCh. 22.SE - Prob. 42APCh. 22.SE - Prob. 43APCh. 22.SE - Which, if any, of the following compounds can be...Ch. 22.SE - Prob. 45APCh. 22.SE - Prob. 46APCh. 22.SE - Prob. 47APCh. 22.SE - How might you convert geraniol into either ethyl...Ch. 22.SE - Prob. 49APCh. 22.SE - One way to determine the number of acidic...Ch. 22.SE - Prob. 51APCh. 22.SE - Prob. 52APCh. 22.SE - Prob. 53APCh. 22.SE - Prob. 54APCh. 22.SE - Prob. 55APCh. 22.SE - Prob. 56APCh. 22.SE - All attempts to isolate primary and secondary...Ch. 22.SE - How would you synthesize the following compounds...Ch. 22.SE - Prob. 59APCh. 22.SE - Prob. 60APCh. 22.SE - Prob. 61APCh. 22.SE - Prob. 62APCh. 22.SE - As far back as the 16th century, South American...Ch. 22.SE - The key step in a reported laboratory synthesis of...Ch. 22.SE - Prob. 65AP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Using the trans effect sequence given in the text, devise rational procedures for selectively synthesizing each of the three isomers of [Pt(py)NH3NO2Cl].arrow_forwardSuggest a reason why the nitroso group, -N=O, is ortho-para directing whereas the nitro group, -NO2, is meta directing.arrow_forwardSuggest an explanation for the fact that the order of reactivity of the halides toward n-butylbrosylate in acetone is Cl” > Br” > I~ when (C4H9)4N* is the cation of the halide salt butI” > Br” > Cl” when Li’ is the cationarrow_forward
- Why cis-Ru(II)Cl2(DMSO)4 reacts with pyridine, et cetera, to give substitution of the DMSO but not the chloride ligands, but trans-Ru(II)Cl2(py)4 react with suitable Na+ and K+ salts in aqueous pyridine to afford chloride-substituted derivatives. write the reactions equations.arrow_forwardThe reaction of 1-bromoheptane with the propionate anion to give heptyl propionate ester is greatly accelerated by adding catalytic amounts of KI. Explain.arrow_forwardHighlight the allylic hydrogen(s) in this structure.arrow_forward
- Give a detailed reaction mechanism for the reaction expected to occur when 2-bromo-2-methylpentane is heated with sodium methoxide. Draw clear structural formulas of all relevant species and use curved arrows to represent electron flow. Discuss the stability of the final compoundarrow_forwardQuestion: How do quantum mechanical effects influence the stability and reactivity of molecules with non-classical carbocations, such as the 2-norbornyl cation, and how does this impact the reaction mechanisms and outcomes?arrow_forwardAnswer the following question pertaining to the reaction of maleic anhydride and 1,3-butadiene: Why does 1,3-butadiene react more rapidly with maleic anhydride than with another molecule of itself?arrow_forward
- Write TRUE if the underlined word/phrase makes the statement correct. Otherwise, write the correct WORD/PHRASE that will make the statement true Addition of KMnO4 to 2,3-dihydroxycyclohexanone results in a purple solution. 2-phenylhexanol will result in the formation of brown precipitate in neutral KMnO4 and clear solution in Lucas reagent. The-CHO group stabilizes the sigma complex formed in electrophilic aromatic substitution. In the aromatic bromination using elemental bromine, using a polarizing solvent will result to a faster decolorization of solution.arrow_forwardAfter E2 elimination, will the following molecule contain deuterium? Why or why not?arrow_forwardi need help writing all of the detailed methods: in Preparation of Cyclohexanone by Hypochlorite Oxidationarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole


Macroscale and Microscale Organic Experiments
Chemistry
ISBN:9781305577190
Author:Kenneth L. Williamson, Katherine M. Masters
Publisher:Brooks Cole
SAR of Anticancer(Antineoplastic) Drug/ Alkylating agents/ Nitrogen Mustard; Author: Pharmacy Lectures;https://www.youtube.com/watch?v=zrzyK3LhUXs;License: Standard YouTube License, CC-BY