M4.24 (Sl units) Aluminum has a density of 2.70 g/cm2 at room temperature (20°C). Determine its density at 650°C, using data in Table 4.1 of the book for reference (Hint: Assume a 1 cm2 cube, 1 cm on each side). Round to the nearest thousandth of a g/cm2. Table 4.1 Volumetric properties in U.S. customary units for selected engineering materials Coclicient of Thermal Expansion, a Cx10F10 Melting Point, T Density, p g/em Material Metals Aluminum 2.70 (0.098) (13.3) 660 (1220) 24 Copper 8.97 (0.324) 17 1083 (1981) (94) Iron 7.87 (0.284) 12.1 (6.7) 1539 (2802) Lead 11.35 (0.410) 29 (621) (16.1) 327 Magnesium 1.74 (0.063) 26 (144) 650 (1202) Nickel 8.92 (0.322) 13.3 (7.4) (2651) 1455 Steel (0.284) 7.87 12 (6.7) a Tin 7.3 (0.264) 23 (12.7) 232 (449) Titanium 4.51 (0.163) 8.6 (4.7) 1668 (3034) Tungsten 19.30 (0.697) 4.0 (2.2) 3410 (6170) Zinc 7.15 (0.258) 40 (22.2) 420 (787) Ceramics and Silicon Glass 2.5 (0.090) 1.8-9.0 (1.0-5.0) Alumina 3.8 (0.137) 9.0 (5.0) 2072 (3762) Silica 2.66 (0.096) 0.55 (0.31) 1600 (2912) Silicon 2.33 (0.085) 2.6 (1.4) 1414 (2577) Polymers Phenol resins 1.3 (0.047) 60 (33) Nylon Polyethylene I.16 (0042) 100 (55) 260 (500) 0.92 (0.033) 180 (100) 115 (240) Polystyrene 1.05 (0.038) 70 (39) 240 (464) Polyvinylchloride 140 (0.051) 50 (28) 212 (414) Compiled from. 121.131. 141. 15). and other sources Melting tesmperature depends on composition Low-density polyethylene Chemically degrades at high temperanures because it is a thermosetting polymer, ocher polymers listed are thermoplastic
M4.24 (Sl units) Aluminum has a density of 2.70 g/cm2 at room temperature (20°C). Determine its density at 650°C, using data in Table 4.1 of the book for reference (Hint: Assume a 1 cm2 cube, 1 cm on each side). Round to the nearest thousandth of a g/cm2. Table 4.1 Volumetric properties in U.S. customary units for selected engineering materials Coclicient of Thermal Expansion, a Cx10F10 Melting Point, T Density, p g/em Material Metals Aluminum 2.70 (0.098) (13.3) 660 (1220) 24 Copper 8.97 (0.324) 17 1083 (1981) (94) Iron 7.87 (0.284) 12.1 (6.7) 1539 (2802) Lead 11.35 (0.410) 29 (621) (16.1) 327 Magnesium 1.74 (0.063) 26 (144) 650 (1202) Nickel 8.92 (0.322) 13.3 (7.4) (2651) 1455 Steel (0.284) 7.87 12 (6.7) a Tin 7.3 (0.264) 23 (12.7) 232 (449) Titanium 4.51 (0.163) 8.6 (4.7) 1668 (3034) Tungsten 19.30 (0.697) 4.0 (2.2) 3410 (6170) Zinc 7.15 (0.258) 40 (22.2) 420 (787) Ceramics and Silicon Glass 2.5 (0.090) 1.8-9.0 (1.0-5.0) Alumina 3.8 (0.137) 9.0 (5.0) 2072 (3762) Silica 2.66 (0.096) 0.55 (0.31) 1600 (2912) Silicon 2.33 (0.085) 2.6 (1.4) 1414 (2577) Polymers Phenol resins 1.3 (0.047) 60 (33) Nylon Polyethylene I.16 (0042) 100 (55) 260 (500) 0.92 (0.033) 180 (100) 115 (240) Polystyrene 1.05 (0.038) 70 (39) 240 (464) Polyvinylchloride 140 (0.051) 50 (28) 212 (414) Compiled from. 121.131. 141. 15). and other sources Melting tesmperature depends on composition Low-density polyethylene Chemically degrades at high temperanures because it is a thermosetting polymer, ocher polymers listed are thermoplastic
Welding: Principles and Applications (MindTap Course List)
8th Edition
ISBN:9781305494695
Author:Larry Jeffus
Publisher:Larry Jeffus
Chapter26: Welding Metallurgy
Section: Chapter Questions
Problem 12R: What is a eutectic composition?
Related questions
Question

Transcribed Image Text:M4.24 (Sl units) Aluminum has a density of 2.70 g/cm2 at room temperature (20°C). Determine its
density at 650°C, using data in Table 4.1 of the book for reference (Hint: Assume a 1 cm2 cube, 1 cm on
each side). Round to the nearest thousandth of a g/cm2.
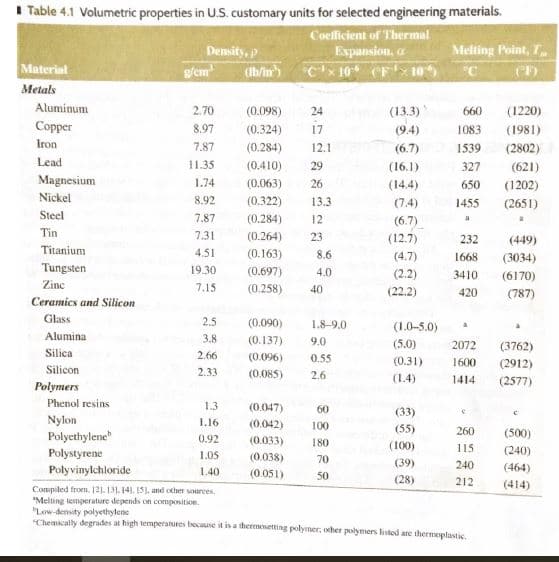
Transcribed Image Text:Table 4.1 Volumetric properties in U.S. customary units for selected engineering materials
Coclicient of Thermal
Expansion, a
Cx10F10
Melting Point, T
Density, p
g/em
Material
Metals
Aluminum
2.70
(0.098)
(13.3)
660
(1220)
24
Copper
8.97
(0.324)
17
1083
(1981)
(94)
Iron
7.87
(0.284)
12.1
(6.7)
1539
(2802)
Lead
11.35
(0.410)
29
(621)
(16.1)
327
Magnesium
1.74
(0.063)
26
(144)
650
(1202)
Nickel
8.92
(0.322)
13.3
(7.4)
(2651)
1455
Steel
(0.284)
7.87
12
(6.7)
a
Tin
7.3
(0.264)
23
(12.7)
232
(449)
Titanium
4.51
(0.163)
8.6
(4.7)
1668
(3034)
Tungsten
19.30
(0.697)
4.0
(2.2)
3410
(6170)
Zinc
7.15
(0.258)
40
(22.2)
420
(787)
Ceramics and Silicon
Glass
2.5
(0.090)
1.8-9.0
(1.0-5.0)
Alumina
3.8
(0.137)
9.0
(5.0)
2072
(3762)
Silica
2.66
(0.096)
0.55
(0.31)
1600
(2912)
Silicon
2.33
(0.085)
2.6
(1.4)
1414
(2577)
Polymers
Phenol resins
1.3
(0.047)
60
(33)
Nylon
Polyethylene
I.16
(0042)
100
(55)
260
(500)
0.92
(0.033)
180
(100)
115
(240)
Polystyrene
1.05
(0.038)
70
(39)
240
(464)
Polyvinylchloride
140
(0.051)
50
(28)
212
(414)
Compiled from. 121.131. 141. 15). and other sources
Melting tesmperature depends on composition
Low-density polyethylene
Chemically degrades at high temperanures because it is a thermosetting polymer, ocher polymers listed are thermoplastic
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

Recommended textbooks for you

Welding: Principles and Applications (MindTap Cou…
Mechanical Engineering
ISBN:
9781305494695
Author:
Larry Jeffus
Publisher:
Cengage Learning

Welding: Principles and Applications (MindTap Cou…
Mechanical Engineering
ISBN:
9781305494695
Author:
Larry Jeffus
Publisher:
Cengage Learning