magnesium ribbon liquid A 25°C bright light water 60°C liquid B 25°C 7a What event is occuring? 8a. What event is occuring? 9a. What event is occuring? 7b. Circle 8b. Circle 9b. Circle Chemical Change or Physical Change Chemical Change or Physical Change Chemical Change or Physical Change
magnesium ribbon liquid A 25°C bright light water 60°C liquid B 25°C 7a What event is occuring? 8a. What event is occuring? 9a. What event is occuring? 7b. Circle 8b. Circle 9b. Circle Chemical Change or Physical Change Chemical Change or Physical Change Chemical Change or Physical Change
Introductory Chemistry: A Foundation
9th Edition
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Donald J. DeCoste
Chapter10: Energy
Section: Chapter Questions
Problem 10ALQ
Related questions
Question
Hi can you please help me with these following questions this is my last assignment thank you so much I really appreciate it
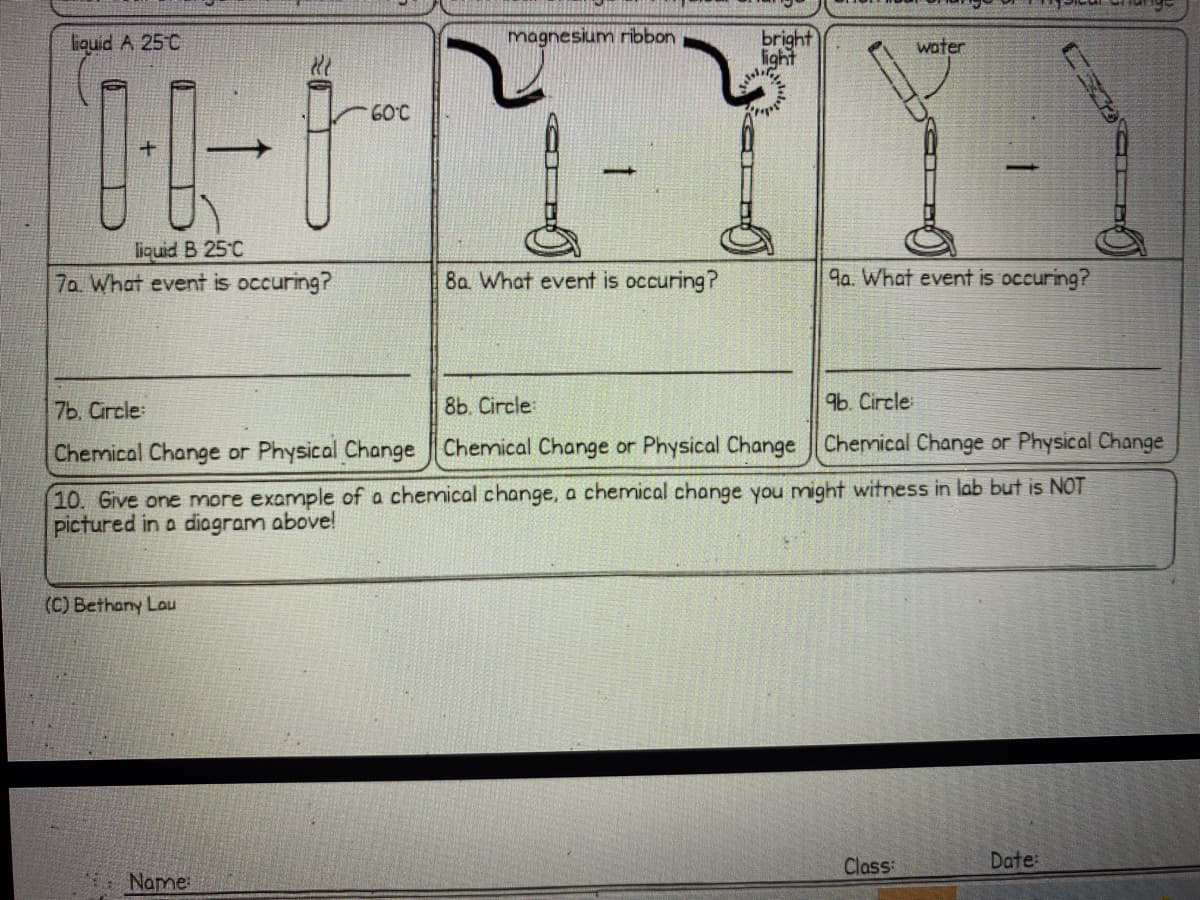
Transcribed Image Text:bright
light
liquid A 25°C
magnesium ribbon
water
60°C
liquid B 25°C
7a What event is occuring?
8a. What event is occuring?
9a. What event is occuring?
7Ь. Circle:
8b. Circle:
9b. Circle
Chemical Change or Physical Change Chemical Change or Physical Change Chemical Change or Physical Change
10. Give one more example of a chemical change, a chemical change you might witness in lab but is NOT
pictured in a diagram above!
(C) Bethany Lau
Class
Date:
Name
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

Chemistry for Today: General, Organic, and Bioche…
Chemistry
ISBN:
9781305960060
Author:
Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

Chemistry for Today: General, Organic, and Bioche…
Chemistry
ISBN:
9781305960060
Author:
Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:
Cengage Learning
