Q: In which of the following compounds would hydrogen have an oxidation state of zero? A) HI B) NaH C) ...
A: Since you have posted multiple questions, we are entitled to answer the first only. Please repost ot...
Q: 1) A 50.0 mL sample of H2SO4 is neutralized with a KOH solution having aconcentration of 0.400 M. If...
A: In such type of questions equivalents of acid & base are equal.
Q: How can tungsten be conserved if it accidentally becomes contaminated?
A: Allowing the tungsten to touch the weld pool is one of the most common causes of contamination. This...
Q: Please answer my question fast
A: Reaction will be- CH4+Cl2→CH3Cl+HCl But in presence of excess of chlorine then reaction will not ...
Q: Select the correct mole-to-mole for the following chemical equation: 2As2O3 + 3C → 3CO2 + 4As Group...
A: The balanced reaction taking place is given as, => 2 As2O3 + 3 C ------> 3 CO2 + 4 As
Q: NaIO3 + 3 NaHSO3 → I- + 3 SO42- + 3 H+ + 4 Na+ 5 I- + NaIO3 + 6 H+ → 3 I2(s) + 3 H...
A: The two reactions given are, 1) NaIO3 + 3 NaHSO3 → I- + 3 SO42- + 3 H+ + 4 Na+ 2) 5 I- + NaIO3 ...
Q: In a flame spectrometric determination of sodium, lithium is used as an internal standard. Assume th...
A: The concentration and intensity are directly related to each other. According to Beer's-Lambert law...
Q: The atomic weight of chromium is less than that of copper. Which of the following statements is true...
A: No. of atoms in 345g of chromium = 345 X6.023X1023/51.99 = 39.96X1023
Q: 8. A 330 KJ/mol is the sublimation energy of aluminum, and its atomic radius is 118 pm. Discuss math...
A:
Q: Please answer the question show step by step and box answer once finished for a) and b) photos are a...
A: Mean = (sum of all measurements)/ number of measurements. Percent error =( |actual value - expected...
Q: Chemistry Question
A: IUPAC NAME CH3COOH IUPAc name
Q: Sketch the shape of each of the σ molecular orbitals for H2+ shown in Figure 6.5 in a plane perpendi...
A:
Q: Limiting reagent/percent yield
A: Limiting Reagent: It can be defined by saying that the compound which s completely consumed during t...
Q: an object with high kinetic energy would feel ??? to the touch -cool -rough -warm -smooth
A: Kinetic energy of the object is directly related to the temperature as follows: K.E. = (3/2)KBT ...
Q: Chemistry Question
A: A Balanced chemical reaction happens when The number of different atoms of elements on reactant side...
Q: How does absorption of water onto CO2(s) affect the Henry's Law constant? How does a gas leak in a s...
A: Henry's law is an important gas law which represents the relationship between the solubility of a ga...
Q: Letter D,E,F
A: Calculation of standard cell potential (Eocell) Eocell=Eocathode-EoanodeEocathode=standard reduction...
Q: Consider the equation:HA(aq) 1 H2O(l) N H3O1(aq) 1 A–(aq).a. If water is a better base than A–, whic...
A:
Q: 1
A: The balanced chemical reaction is- C3H8 (g) + 5O2 (g) → 3CO2(g) + 4O2 There are supposed to ...
Q: Write a chemical equation for HF(aq) showing how it is an acid or a base according to the Arrhenius ...
A: Arrhenius acid is a substance that release H+ ion in an aqueous solution. Arrhenius base is a substa...
Q: . In experiment of hydrogen peroxide, the decomposition was equal to (30%) after time is (35 min.) i...
A: NOTE : Since the order of reaction is not given, we can assume it to be 1 as the decomposition of H2...
Q: 3. How many grams are in 0.150 moles of the following: a. CaCl, b. Na,CO, 4. How many moles are in 2...
A: In these type of questions we need the formula: Moles= given weight/ molar mass
Q: Major product?
A:
Q: For the equilibrium system: H2O(g) + CO(g) <--> H2(g) + CO2(g). Delta heat of rxn: -42 kJ/mol ...
A: The reaction taking place is given as, => H2O (g) + CO (g) -------> H2 (g) + CO2 (g) ...
Q: A drop of phenolphthalein is added to a solution of 1.0 M H2SO4 with a volume of 10.0 mL. The soluti...
A: phenolphthalein is an acid base indicator which is colorless in acidic medium and pink in basic medi...
Q: 1. Balance the following chemical equations: a) CaHe O2 -> CO2 H20 b) Mg H3PO4 -> Mg3(PO4)2 H2
A:
Q: How many moles of O2 are required to produce 15.0 g of N2O4 in the reaction below? N2 + 2O2 N2O4
A: Ans. For the given reaction N2 + 2O2 ----->> N2O4 92 g of N2O4 required = 64 g of O2 Hence ...
Q: How many moles of Fe(NO3)2 are present in 32.176 g Fe(NO3)2?
A: Molar mass of Fe(NO3)2 = 56 + (14+ 16x3) x 2 = 56 + 124 = 180 g/mol Given mass of Fe(NO3)2 = 32.176 ...
Q: Major product and mechanisfor this ?
A: The given reaction is an example of wittig reaction in which alkene is formed as the final product.
Q: The balanced chemical equation is P₄(s) + 6 Cl₂(g) → 4 PCl₃(g). What is the mass in grams of phospho...
A:
Q: #124
A: Given :- molar concentration of NaOH = 0.001 M To calculate :- [OH-] and pH of the solution
Q: Define the term initial rate of reaction
A: Term = Initial rate Definition = ?
Q: Write the Lewis structure for CrO4^2- and determine the formal charges to explain the correct Lewis ...
A:
Q: Chemistry Question
A:
Q: 4
A: 4) Given that, Mass of CH4 = 12 gms Molar mass = 16 gm/mol Number of moles of CH4=Given massMolar ma...
Q: s aniline point is
A: Relation between both temperature scale : [ T(°C) • 9/5 ] + 32 = T( °F ) T( °F ) = [ 40°C • ( 9 / ...
Q: Q.4: Find the fraction of the body centered cubic unit cell volume filled with hard spheres as shown...
A: Given total volume = 343 cm3 we know volume =43×π × r3 343 cm3 =43×3.14 × r3 ...
Q: What do you notice about the number of each atom in the reactants and of each atom in the products? ...
A: Let us consider a balanced chemical reaction:HCl + NaOH → NaCl + H2OIn this above example we can not...
Q: How Many of water do You drink per day
A: . For adults, the general recommendation from The U.S. National Academies of Sciences, Engineering, ...
Q: Help plz
A: The ideal gas equation gives the quantitative relationship between the pressure, volume, temperature...
Q: What change in charge does an ion have when it is oxidized?
A: There is a loss of electrons in the oxidation process.
Q: convert 0.366 mole of Zn to grams of Zn
A: Given Number of mole of Zn = 0.366 mole Mass of zn = ?
Q: CHCASE Dashboard Item 3 A student modeks four types of land use in a city. Each model has the same a...
A: Ground water is recharged by some ways like rainwater,harvesting and raining. Water from rain seeps ...
Q: Chemistry Question
A: Common radioactive decay products are alpha , beta , gamma etc. They are ordered by their ability to...
Q: Which of the following has the proper partial positive and partial negative labels for the molecule ...
A: Lithium is element of s-block and it is highly electropositive in nature While, chlorine is element ...
Q: Write the word (true) in front of the correct sentence and the word (false) in front of the incorrec...
A: We can find out acid strength by using general rule such as, a more electronegative central atom p...
Q: Chemistry Question
A: IUPAC NAME OF THE COMPOUND:- 1-methyl-5-cyclopentyl-Cyclodecane
Q: What led Rutherford to believe that atoms contain a positive nucleus? none of the above some alpha p...
A: Rutherford deduced that the atomic nucleus was positively charged because the alpha particles that h...
Q: PE RCENT C 19) Determin the the fo a) C,„H2 12 22
A: mass of C = 12 g/mol
Q: If H2S reacted with Oz to produce SO2 and H20, and the theoretical yield was to be 15.94 g of H20, b...
A: Given:The balanced reaction is:H2S + O3 → SO2 + H2OTheoretical yield = 15.94 g Experimental yie...
Major product for these?
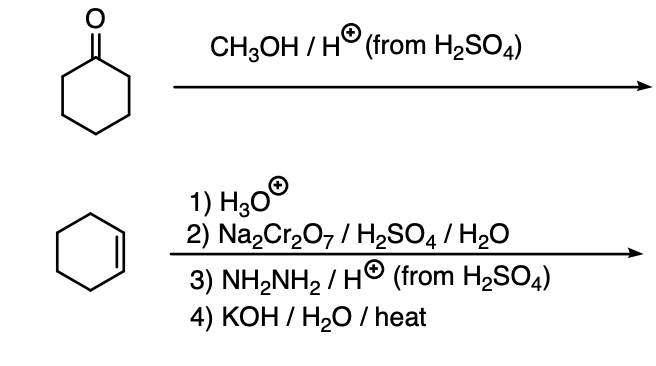
Step by step
Solved in 2 steps with 2 images

- What is the value of K' if ∆G°' = -185J? Round to 3 decimal places. R = 8.314 J•mol-1 K-1 ∆G°' = -RTlnK' ∆G' = ∆G°' + RTlnQ'C2H4 (g) + 3 O2 (g) ⟶ 2 CO2 (g) + 2 H2O(l) What is the numerical value of Z in the equation below? delta(c02)/delta(t)= z x delta(c2h4)/delta(t)2 LiOH(s) → Li2O(s) + H2O(l) ΔH° = 379.1 kJLiH(s) + H2O(l) → LiOH(s) + H2(g) ΔH° = -111.0 kJ2 H2(g) + O2(g) → 2 H2O(l) ΔH° = -285.9 kJ Compute ΔH° in kJ for 2 LiH(s) + O2(g) → Li2O(s) + H2O(l)
- Cl(g) ΔH°f, = +121.3 kJ mol–1 Cl–(aq) ΔH°f, = –167.2 kJ mol–1 HCl(g) ΔH°f, = –92.3 kJ mol–1 HCl(aq) ΔH°f, = –167.2 kJ mol–1 Cl2(g) ΔH°f, = 0 kJ mol–1 H2O(l) ΔH°f, = –286 kJ mol–1 ClO2(g) ΔH°f, = +102.5 kJ mol–1 Cl2O(g) ΔH°f, = +80.3 kJ mol–1 O(g) ΔH°f, =+249.0 kJ mol–1 H(g) ΔH°f, =+218.2 kJ mol–1 Which material is most 'stable' at 298 K? Question 1 options: Cl2(g) Cl–(aq) Cl(g)TABLE A-V (H2) Gf= 0 kJ/mol (CO) Gf= -137.15 kJ/mol (HCHO) Gf= -113 kJ/molI got 2.71 and it says its wrong can someone explain please

