Q: Q3: The Figure shown a HPLC chromatogram of two compound.Calculate the following values using the…
A:
Q: Interconverting Ka and pka Complete the following table, which lists information about the measured…
A: There are several theories to define acid and Base. Here we are required to complete the table to…
Q: Short answer please Maximum 50 words Thank you
A: given is the diagrams of various atomic models in a different era of time.
Q: Calculate the amount of heat needed to boil 196. g of acetic acid (HCH3CO₂), beginning from a…
A: Mass of acetic acid = 196 g Number of moles = Mass /Molar mass
Q: What is the pH at 25°C of a saturated solution of silver hydroxide?
A: The pH at 25°C of a saturated solution of silver hydroxide is 10.5
Q: A voltaic cell is constructed in which the following cell reaction occurs. The half-cell…
A: Please repost the second question. As per the guidelines, I am answering the first question.…
Q: What is the pH of a solution made by mixing 25.00mL of .1500M NaOH with 25.00mL of .2500M HCIO2?
A:
Q: The decomposition of N2O5 to NO2 and O2 is first order with respect to the reactant N205 and first…
A:
Q: . Give the health and environmental effects of carbon monoxide, nitrogen dioxide, sulfur dioxide,…
A: carbon monoxide, nitrogen dioxide, sulfur dioxide, ozone, PM10 and lead, these all are pollutants.
Q: Experiment - Ideal Gas Law.pdf O a. Because having the water levels even ensures that the pressure…
A: A question based on ideal gas. A set up that measures the volume of CO2 formed is prescribed and the…
Q: A solution is made by mixing 0.400M in formic acid, HCOOH, and 1.00M in sodium formate, HCOONa?…
A:
Q: At what approximate positions might the compound below show IR absorptions? Associate functional…
A: The criteria for a molecule being IR active is that the molecule must have changing dipole moment…
Q: 8 H+(aq) + Cr2O72-(aq) + 3 H2S (aq)----> 2 Cr+3 (aq) + 7 H2O (l) + 3 S (s) a. Assign oxidation…
A:
Q: [Co²+] = = 1.17 M and [Fe²+] = 0.00935 M, is −42.9 kJ: AG = -42.9 kJ The free energy change for the…
A: As per the guidelines, I am solving the first question. Kindly repost the second question. The given…
Q: A certain reaction is first order in N₂ and second order in H₂. Use this information to complete the…
A:
Q: H HO- H CHO OA H CH H CHOH
A: -> Carbonyl compound reacts one equivalent alcohol and form Hemiacetal as product . If…
Q: H-C=C-H 1) Nalt 2)ph^Br I) NaNH 2) Li Ph
A:
Q: c. Car batteries are based on a lead acid system, draw a diagram representation of this kind of a…
A: In car, lead acid batteries are used.
Q: e) Based on Valence Bond Theory, show the steps formation of hybrid orbital for a sulfur atom in…
A: Valence Bond Theory (VBT): It is defined as a theory which explains chemical bonding. It states that…
Q: Consider a galvanic cell based on the reaction 2 Fe2+ (aq) + Cl2 (g) ------------>2 Fe3+…
A:
Q: N MeOH
A:
Q: how do you determine [OH-]? whats the value of [OH-]?
A:
Q: THIS IS AN EXAMPLE OF WHAT THE RESPONSE TO THIS QUESTION SHOULD LOOK LIKE. APPLY THIS TO THE…
A:
Q: Which statement is correct about the pH of a 0.100M solution of KC2H302? a. The cation is neutral,…
A: The salt given is KC2H3O2 . The true statement about the pH of 0.100 M solution of KC2H3O2.
Q: When the Pb2+ concentration is 9.35x10-4 M, the observed cell potential at 298K for an…
A:
Q: A 20.0 mL sample of whiskey was diluted to 500.0 mL. A 2.00 mL aliquot of the diluted sample was…
A: According to the question, The volume of the K2Cr2O7= 50 ml = 0.050 L The molarity of the K2Cr2O7=…
Q: 4. Balancing an equation for a redox reaction in basic conditions. Aqueous sodium hypochlorite…
A:
Q: 14. Consider the following energy diagram for a reaction. Calculate the activation energy Ea and the…
A: An energy diagram for a reaction is given. We need to find the activation energy, Ea and Enthalpy of…
Q: What type of holes do the blue spheres occupy in the ionic solid depicted below? Give evidence to…
A: When Coordination number surrounding a particular void is 4 that means 4 atoms surround that void,…
Q: Electromagnetic radiation with a wavelength of 575 nm appears as yellow light to the human eye. If a…
A: Answer:- This question is answered by using the simple concept of calculation energy of Photon using…
Q: The bicarbonate/ carbonic acid buffer is of extreme importance to our bodies. The pH of importance…
A:
Q: A) B) J D) AICI 3 (CH₂)P- Zn(Hg) HCI NaOH heat DMSO I
A: A) here benzene ring react with ethyl chloride in presence of Aluminium chloride to give ethyl…
Q: 10. 0.94 The base protonation constant K of ammonia (NH3) is 1.8 × Calculate the pH of a 0.94 M…
A: Given- Concentration of Ammonia= 0.94 M Kbof Ammonia= 1.8×10-5 pH of solution = ? To find
Q: A voltaic cell is constructed from a standard Cl₂|CI¯ half cell (Eºred = 1.360V) and a standard Mg2+…
A: Less the reduction potential, less reduced the cell is . More the reduction potential, more…
Q: ): A compound had a molar absorptivity of 2.93 x 10' L.cm mol¹. What concentration of the compound…
A: It is an application of Lambert Beer law Here we are required to find the concentration of the…
Q: C) D) E) NH₂ 1. Excess CH3l 2. Ag₂O, H₂O 3. heat Br₂ CH3CO₂H I Excess HBr
A: C. This is called Hoffman Exhaustive Elimination reaction. D. This is called bromination of…
Q: b. How many electrons are involved for the half-reaction: MnO₂ ‒‒‒‒‒‒ Mn²+
A:
Q: .Draw the Lewis dot structure(s) for iodine triflouride: What is its electron domain shape? What is…
A: Answer:- This question is answered by using the simple concept of determination of shape by writing…
Q: 15. Consider the following elementary reactions. Write the overall reaction and determine the…
A: A chemical reaction can be added or subtracted like an algebraic equation. The given reaction…
Q: Calculate the Ecell for the system Cu²+ + Zn Cu + Zn²+
A:
Q: ОН ОН Draw the molecule on the canvas by choosing buttons from the Tools (for bonds and charges),…
A: Pinacol-pinacolone rearrangement: 1,2-diols in the presence of acid gives a carbocation, then…
Q: In a DEPT-135 spectrum, which labeled carbons in the molecules below would show positive and which…
A: A)Two postive peaks and two negative peaks. B) three postive peaks and zero negative peak
Q: At a certain temperature this reaction follows first-order kinetics with a rate constant of 0.00108…
A:
Q: signment/takeCovalentActivity.do...@ %D0 [Review Topica] [References] Use the References to access…
A:
Q: Nitric acid is added to a sample of zinc. Based on the following information, what is the…
A: Initial mass of Zn = 9.50 g Final mass of Zn = 3.23 g Mass of Zn reacted = 9.50 - 3.23 = 6.27 g
Q: Identify each of the following substances as an electrolyte or a nonelectrolyte and predict which…
A: A substance is an electrolyte when it dissociate into ions when dissolved in water and these ions…
Q: 2. Indicate the type of crystal (molecular, metallic, ionic, or covalent-network) each of the…
A: We need to classify the given compounds upon solidification as molecular solids, metallic solids,…
Q: For the following chemical reaction, CO (g) + H₂O (g) → CO2 (g) + H2 (g) What is the Kc expression…
A:
Q: d e (D CI CH₂Br CH3OH conc.H₂SO4 NaOEt EtOH A
A: In presence of polar solvent substitution reaction is favoured where in presence of less polar or…
Q: The decomposition of N2O5 to NO2 and O2 is first order with respect to the reactant N2O5 and first…
A:
Give a synthesis for the following reactions
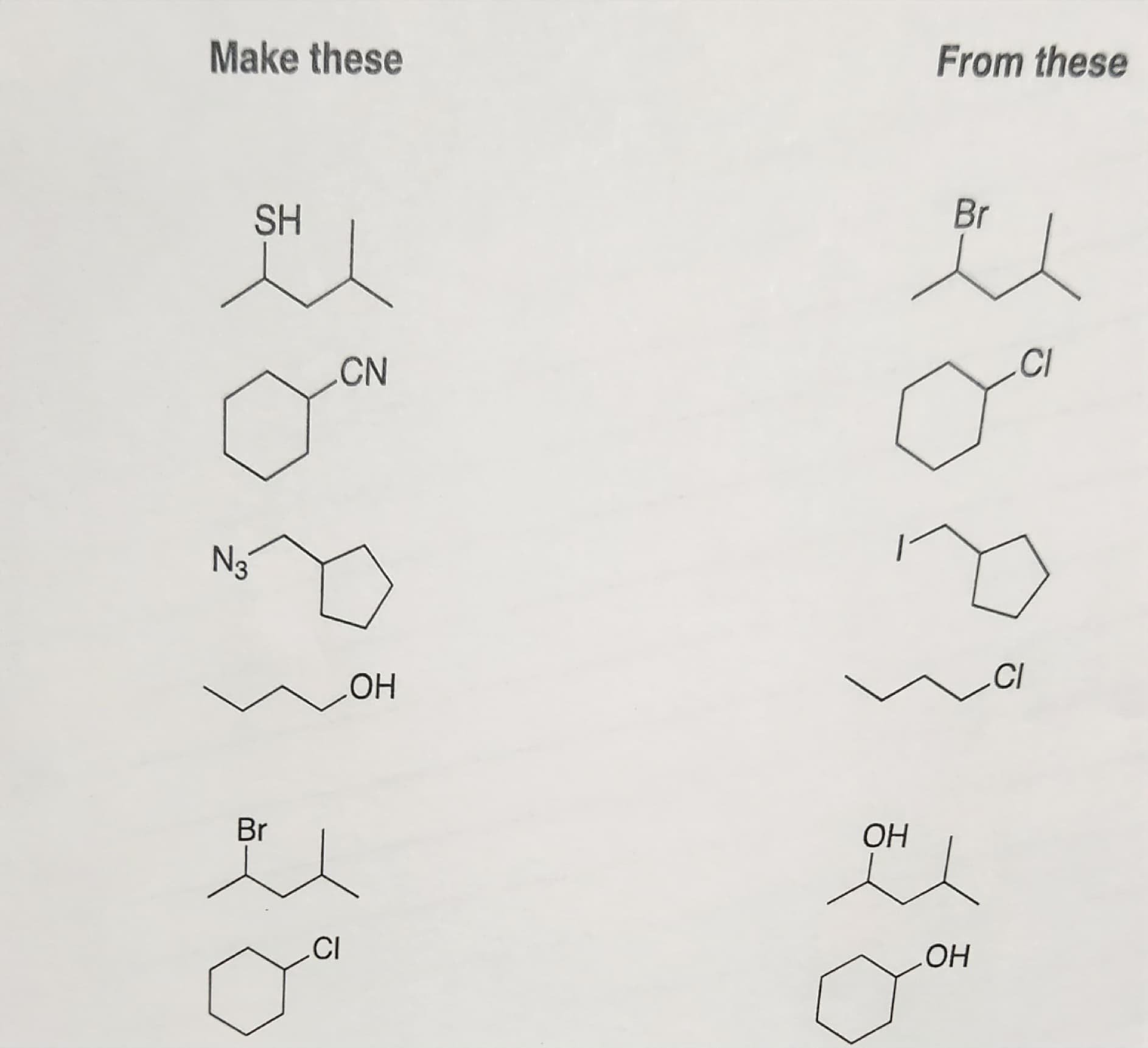
Step by step
Solved in 2 steps with 2 images


