MasteringChemistry: CHE154 S X + i session.masteringchemistry.co This Mac can't connect to iClou a problem with "ayanaboo10@g Open iCloud preferences to fix this pro Later iCloud Pr
MasteringChemistry: CHE154 S X + i session.masteringchemistry.co This Mac can't connect to iClou a problem with "ayanaboo10@g Open iCloud preferences to fix this pro Later iCloud Pr
Physical Chemistry
2nd Edition
ISBN:9781133958437
Author:Ball, David W. (david Warren), BAER, Tomas
Publisher:Ball, David W. (david Warren), BAER, Tomas
Chapter8: Electrochemistry And Ionic Solutions
Section: Chapter Questions
Problem 8.9E
Related questions
Question
Transcribed Image Text:MasteringChemistry: CHE154 S X +
i session.masteringchemistry.co
This Mac can't connect to iClou
a problem with "ayanaboo10@g
Open iCloud preferences to fix this pro
Later
iCloud Pr
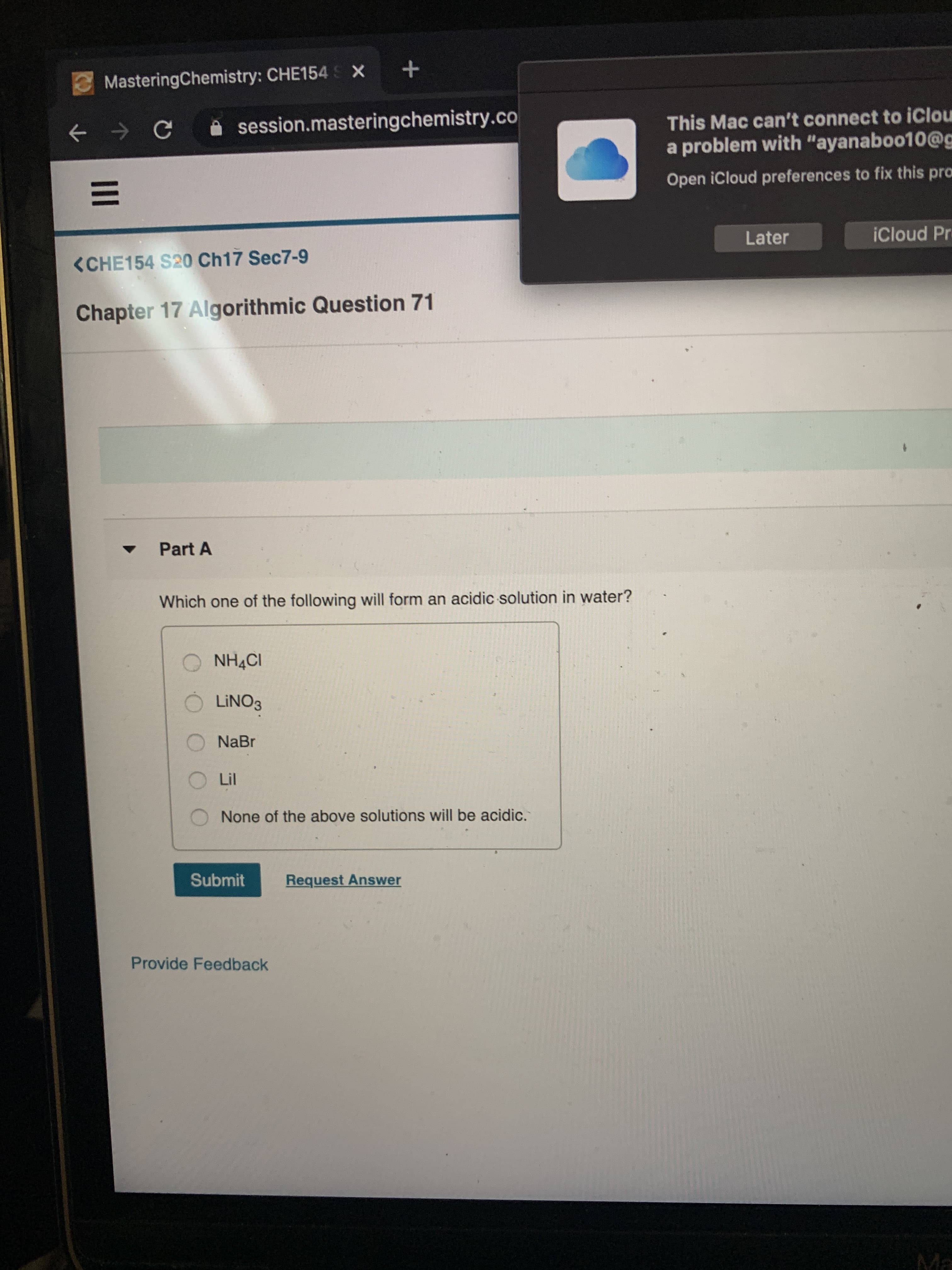
<CHE154 S20 Ch17 Sec7-9
Chapter 17 Algorithmic Question 71
Part A
Which one of the following will form an acidic solution in water?
NH4CI
LINO3
NaBr
Lil
None of the above solutions will be acidic.
Submit
Request Answer
Provide Feedback
Ma
II
Expert Solution
Step 1
The reaction between a strong acid and a strong base result in the formation of neutral salt.
A salt of a strong acid and weak base can produce an acidic solution.
A salt of a strong base and weak acid can produce a basic solution.
Step by step
Solved in 2 steps

Recommended textbooks for you

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning