Q: The pH of a 0.64M solution of acrylic acid (HC3H₂CO₂) is measured to be 2.22. Calculate the acid…
A:
Q: Calculate the K₂ for a monoprotic acid, HX, if a 0.100000 M solution has a pH of 3.333. The data is…
A: The answer is as follows:
Q: 十 III
A: 1. Acetylene reacts with NaNH2 and methyl iodide produces methyl acetylene. Methyl acetylene on…
Q: How many KJ of energy are released when 6.39 g of Fe reacts with excess O₂? Given the following…
A: Given : Mass of Fe = 6.39 gm 4Fe + 3O2 -----> 2Fe2O3. ∆H = -1625 KJ
Q: 2. Electrolysis of CuBr₂ was done by passing a current of 4.50 A for 1 hr. Give: (a) the reaction at…
A:
Q: If a gas with a volume of 10mL and a pressure of 236 torrs inside a dewar flask is opened to an…
A:
Q: A 7.00 L tank at 10.8 °C is filled with 6.17 g of sulfur hexafluoride gas and 17.9 g of sulfur…
A:
Q: 4. The molar absorptivities of the indicator weak acid HIn (K₁ = 1.42 x 105) and its conjugate base…
A:
Q: Is it advantageous or disadvantageous for a pathogen to deadly. a. It is advantageous. b. It is…
A: We are to discuss if it is advantageous or disadvantageous for pathogen to be deadly.
Q: CI
A:
Q: At a certain temperature the rate of this reaction is first order in H₂PO4 with a rate constant of…
A:
Q: Structural form of Fischer projection and Haworth projection of D-Mannose
A:
Q: 9. H₂ Pa/c in Co
A:
Q: For the following reaction, 15.9 grams of chlorine gas are allowed to react with 7.20 grams of…
A: Mass of chlorine = 15.9 g Mass of water = 7.20 g Molar mass of Chlorine = 70.90 g/mol Molar mass of…
Q: Question for each cell, writhe a half-reaction occuring at each electrode. write the half-reaction…
A: Answer: In Galvanic cell, at the anode always oxidation takes place an at the cathode always…
Q: CH₂CH₂CCH₂CH₂CH, H₂O Br₂ tert-BuO CH₂CH₂CH₂CH₂Br CHỊCH,C—CH;CHỊCH; CH₂CH₂CH₂CH.Br CH₂CH3 tert-BuO…
A:
Q: Draw the balanced chemical equation for the synthesis of cyclohexene from bromocyclohexane. Classify…
A: To find: Draw the balanced chemical equation for the synthesis of cyclohexene from bromocyclohexane.…
Q: What is the molality of a solution in which 13.7 g of sodium sulfate, Na2SO4, has been dissolved in…
A:
Q: Match each molecule with a shape. H2S…
A: The shape of molecule is determined by the location of nuclei and it's electrons .the electron and…
Q: reaction + Ag (aq) + 2NH3(aq) – Ag(NH3)+ (aq) HBr(aq) + NH3(aq) → Br_(aq) + NH(aq) 2+ 2+ Zn(aq) + 6…
A: 1) Ag+(aq) + 2 NH3(aq) → Ag(NH3)2+(aq)
Q: Provide Major products H13C- C-ce + Na Br 12 Aldol addition of XS H3C-CH₂-C-H + -OH
A: ->NaBr has Br- nucleophile hence , it give nucleophilic addition elimination reaction. ->In…
Q: What type of eliminaton reactions occur in this process? Br Br Two E2 reactions Two E1 reactions An…
A:
Q: A brick has a mass of 4.0 kg and the Earth has a mass of 6.0 × 10²7 g. Use this information to…
A:
Q: Using Hess's Law determine the delta H for the formation of phosphorous pentachloride Note if the…
A: reaction for which the overall ΔH⚬ is negative is exothermic. When it is positive the it is…
Q: The enthalpy of combustion of methane (CH4) is 890 kJ/mol, which is released as heat. Calculate the…
A: Given One mole of methane is released 890 kj of heat
Q: Show that the reaction is not spontaneous under standard conditions by calculating A.G. Express your…
A:
Q: Review Reactions 1. MCPBA 2. H30+
A:
Q: Five liters of nitrogen gas initially at T = 27 degrees C and P = 1 atm are heated to T = 127…
A: Here 5 L of N2 gas initially at 27°C and P=1atm is heated to 127°C .We have to determine w, ∆E ,∆H…
Q: 600.0 mL of a gas is at a pressure of 8.00 atm. What is the volume of the gas at 2.00 atm?
A: Given : Initial volume(Vi) = 600 ml Initial pressure (Pi) = 8.00 atm Final pressure (Pf) = 2.00…
Q: 1. A constant current of 90 mA is applied to an electrochemical cell for 22 minutes. What is the…
A: The charge transport in a unit time is known as electric current. The current is calculated by using…
Q: Using the table of the weak base below, you have chosen Pyridine as your weak base in the buffer…
A: We have find out the values of PKb by using formula PKb= -log kb.
Q: What is the volume of gas in liters at 1.00 atm if it originally occupies 2.00 L at 158 mmHg?
A: We will use the formula as :- P1V1 = P2V2
Q: Determine if the synthesis of sulfuric acid is a spontaneous reaction at 25°C. Chemical Equation: S…
A:
Q: Calculate the potential developed by a voltaic cell using the following reaction dissolved species…
A:
Q: Give the mechanism of given 03 Joleow reactions > CH3CH2CEC - CH CH
A:
Q: Balance the reaction between Fe and NO3 to form Fe2+ and NO in acidic solution. When you have…
A: We are to balance the reaction Fe + NO–3 → Fe+2 + NO in an acidic medium.
Q: The isomerization reaction, CH3NC → CH3CN, is first order and the rate constant is equal to 0.33 s-1…
A:
Q: if 25.0g of NaOH is added to 0.750L of 1.00M Cd(NO3)2 how many grams of Cd(OH)2 will be fromed in…
A:
Q: Identify the ligands in the following complex ions. [AlCl4] - [Cu (NH3)4]2+ [Fe EDTA ]3+ 1 How…
A:
Q: a Which of the following monomers could form an addition polymer? CH4 HO-CH(CH3)-COOH HO CH₂-CH₂-CH3…
A:
Q: Enter your answer in the provided box. A constant current is passed through an electrolytic cell…
A:
Q: For the aqueous [Co(NH₂) complex K = 1.58 × 10³ at 25 °C. 3+ Suppose equal volumes of 0.0012 M Co…
A:
Q: Fill in the molecular orbitals for the H2 molecule. How do you describe the highest occupied…
A: Fill in the molecular orbitals for the H2 molecule. How do you describe the highest occupied…
Q: Complete the balanced equation for HC₂H3O2 dissolved into water. Label the Acid, Base, Conjugate…
A: Given : We have to balance the given incomplete reaction.
Q: Methylamine Ethylamine H3NH₂ C₂H5NH₂ H₂NH3 C₂H5NH3+ 4.38 X 10 5.6 x 10-4 Aniline CH;NH, C6H5NH3 +…
A:
Q: An electrochemical cell is designed using copper as one electrode and another metal higher than…
A: An electrochemical cell is designed using copper as one electrode and another metal.
Q: Question 7 An unusual correlation exists between the potency of an inhaled anesthetic and its…
A: We are to discuss the unusal correlation vetween potency of inhaled anesthetic and its ability.
Q: A. ENDOTHERMIC AND EXOTHERMIC REACTION: Decide whether each of this reaction is exothermic or…
A: Given,
Q: Use standard reduction potentials to calculate the standard free energy change in kJ for the…
A:
Q: A solution is 0.0420 m KI. What is the molarity of the solution if the density is 1.10 g/mL? MW KI =…
A:
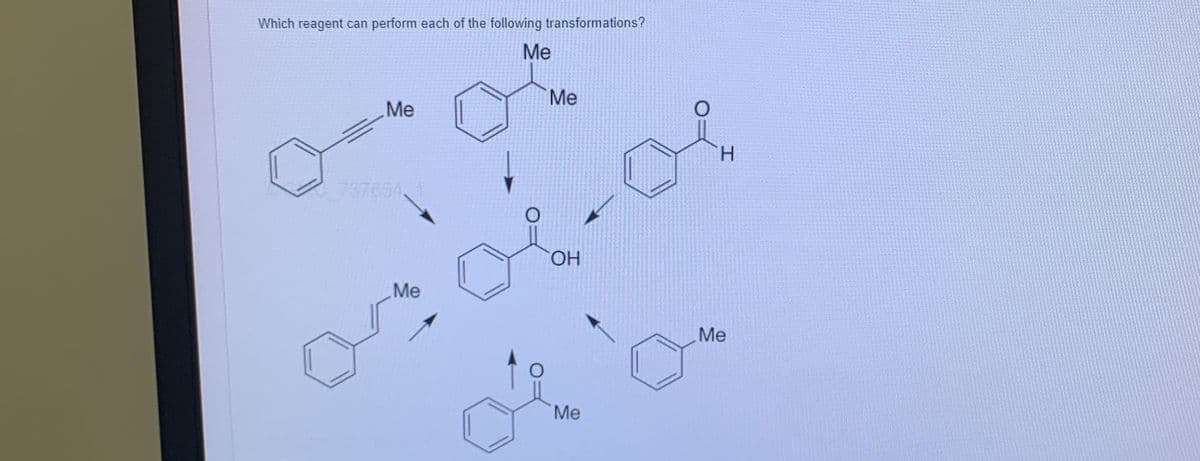
Step by step
Solved in 2 steps with 1 images

- Show how m-toluidine can be converted tom-toluidineCH3 NH2the following compounds, using any necessary reagents.m-toluonitrileWhich set of reagents are most likely to affect the E2 elimination? a. CH3CH2O-, CH3CH2OH b. (CH3)3CO-, (CH3)3COHShow how m-toluidine can be converted tom-toluidineCH3 NH2the following compounds, using any necessary reagents.m-toluonitrile(a) CH3 C N CH3 CH2NH2m-methylbenzylamine(b)m-iodotolueneCH I 3 (c)m-cresol(d) CH3 OH3-methyl-4-nitroanilineCH3 NH2O2N(e)NHCH3N-cyclopentyl-m-toluidine
- Describe the given synthetic pathway. Discuss type of reactions involved, the purpose of each reagent, and the relevance of each step. Provide brief explanation. Create a retrosynthetic map for the synthetic pathway in Question 1.Which of the following is the strongest activating group in electrophilic aromatic substitution reactions? a) -CH2CH2OH b) –OCOCH3 c) –N(CH3)2 d) -N(CH3)3 e) -COOHPropose an efficient synthetic route for each of the following transformations (sketch each intermediate)

