Q: Propose a plausible mechanism for the following transformation. OH [H,SO] MESH Mes
A: Sequence in the mechanism :- Protonation of etheral oxygen Attack of nucleophile to the most…
Q: Every transformation can be completed in one 'synthetic ste added sequentially, ex. "1. BH3, THF; 2.…
A: Given : We have to write the reagent for the following.
Q: 1) Propose an efficient synthesis for the following transformation: + en Br Koty .2 HO Br Hz S04…
A: Organic synthesis of a specific molecule requires a specific amount and type of reagent . It's not…
Q: Propose a mechanism for the following reaction.
A: A mechanism for the following reaction has to be proposed
Q: a. Propose a stepwise mechanism that accounts for the transformations below. 1. NaOEt/ELOH 2. H,O+,…
A: Organic reaction mechanisms
Q: 6. A synthesis of a molecule being explored as an antidepressant drug (J. Org. Chem 2012) utilized…
A: AlCl3---> It is a lewis acid. it accepts the electron. CH2=CH2 (Ethene): It acts as a nucleophile…
Q: 4. Draw the final major product(s) for each multistep synthesis reaction. H30* -OMe H3PO4 Jones…
A: 4a. Ketal deprotection using acid. b. hydration of cyclohexene. oxidation of cyclohexanol.…
Q: 4. Propose a plausible mechanism for the Wittig reaction and then the enol ether hydrolysis. ( H,C-O…
A: Since, you have asking multiple questions, so I will solved first question for you. You can see…
Q: Show the reagents and intermediate products in the following multi-step synthesis. CH3 а) CН — -H…
A:
Q: I need help with the mechanisms on these two reactions please :) NaOH A b. 1. Draw a complete arrow…
A: b.)
Q: Rxn involve cleavage of -H Na NaOH CH3CH,C-OH Na,CO3 + iii) Reduction i. LİA||1, CH;CH,C– OH ii.…
A: Introduction: Carboxylic acid: Carboxylic acid is an organic acid with general formula RCOOH. R can…
Q: te a full mechanism for each reaction shown below. (I) Clearl (iii) show stereochemistry where…
A: According to our guideline, I can answer only the first three sub-parts.
Q: Propose a plausible mechanism for the following transformation. NaH CI HO 13.38e This substitution…
A: Organic reaction mechanisms
Q: 9. Provide a mechanism for the following transformation: H,Cro, он HO H,O
A: In presence of H2CrO4 , alcohol is oxidised to acid through the formation of aldehyde.
Q: What reagent(s) would accomplish the following synthesis? CH CH3 "O., O NAOH, H20 O H20/ROOR H20/H*…
A:
Q: C. Show the product formed in each step of this multi-step synthesis. benzene He CI 1. CH3CI, AICI 3…
A:
Q: 13) Propose a synthesis: moold, H3CO rOCH3
A: Dimethyl malonate on reaction with base, NaOMe generates carbanion which attacks on isobutylbromide…
Q: Propose a mechanism for the reaction shown below. а) CI OH ONH2 + CI Heat NO2 NO2 b) NO2 HNO3 H2SO4
A: Given reactions are : And Mechanism of these reactions = ?
Q: 3. Draw a mechanism for the following reaction. A) HOʻ 1. СH;CH-MgBr 2. H3O* HO B) 1. но |H,8o| H.…
A: Since you have posted a question with multiple sub parts, we will solve first three sub parts for…
Q: Bonus: Propose a mechanism Must be perfect for any points NaOEt OEt EtO EtO HOE!
A: Given reaction,
Q: Propose a mechanism for attached reaction:
A:
Q: 4. The following reaction occurs through an Elcb mechanism followed by a tautomerization. Provide a…
A:
Q: a) H20 .CI Br2/H20 b)
A: The first part will follow the SN1 reaction. In this reaction, the good leaving group will leave by…
Q: 0. 1) СО2, Р, д 2) Н2О, H*
A: This is a klobe synthesis in which major product is salicylic acid
Q: ) ezs Memyes 2) Hb O excess
A:
Q: Propose an efficient synthesis CI MeO O OMe OEt COOH Br NO₂ O EtO
A: We have to propose an efficient synthesis.
Q: 6. A synthesis of a molecule being explored as an antidepressant drug (J. Org. Chem 2012) utilized…
A:
Q: Propose a mechanism for each reaction. (а) + H3C НО ОН CH3 HO H,SO, (b) Ph С—ОН Ph Ph Ph
A: There are many mechanisms in organic chemistry such as nucleophilic substitution, nucleophilic…
Q: 3. Draw the mechanism for each reaction below; HO H, H CH,CH CH,CH,NH, HyC H. H (pH = 5) H. 1. Ph,P…
A: Note: As per company policy we are supposed to attempt first three subparts only. Please repost…
Q: Please complete the following synthesis. Br 3 A E B D Brz, hv Br2, DCM HBr, DCM LDA HBr, H2O2 H I F…
A:
Q: show products and mechanism for reaction below: ** O Unel Tommation| (a) Pd(PPH3)4 OMe EtzN (b) 1)…
A: In presence of Pd catalyst, aryl halide undergo oxidative addition and followed by coupling…
Q: H. HEAT ENDO PRODULT Exo FRODUCT XO
A: Both are Dieles Alder reactions and give Dieles Alder Product via [4+2]π cycloaddion reaction. You…
Q: 4. Propose a mechanism for the reaction below. ( HS SH cat. H
A: Reaction- In analogy for the formation of acetals from alcohols and acid, treatment of…
Q: C. Show the product formed in each step of this multi-step synthesis. Br2, H3O+ KOtBu immmmmm NaOH,…
A: The answer is as follows:
Q: 4. Draw the final major product(s) for each multistep synthesis reaction. H3O* OMe H3PO4 Jones Et,NH…
A: 1. Acetal compound can be deprotected in presence of H3O+ and forms ketone or aldehyde compound. 2.…
Q: What is the best choice of reagent(s) to perform the following 0. ? transformation? a. Hg(OAc)2 and…
A: The best choice of reagents for the given transformation can be identified as,
Q: H,SO4 Нeat
A:
Q: Me „CHO CO2E Me + Ph3Ps .CO2E† Me CH2CI2 Me 0: Me Me 40 °C, 60 min, 89 % (3 steps) Me See the Wittig…
A: Wittig reaction is an organic chemical reaction wherein an aldehyde or a ketone is reacted with a…
Q: N H .N. NaNH2 CI CI
A:
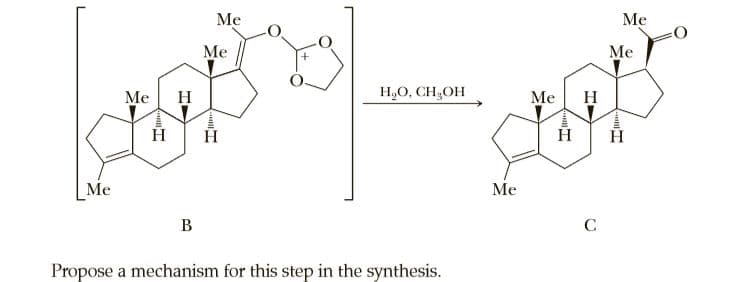
Q: Propose a mechanism for the following reaction:
A:
Q: Propose a detailed reaction mechanism for the following conversion. он НО Acetone HCI но. он OH HO
A: In this question, we have to find out the correct answer of given problem by the help of the…
Q: 6. Propose a synthesis H3CO H.CO- H3CO OH - CH3 H₂CO — HyCO H₂CO NH₂ CH,CH,NH, mescaline
A:
Q: Propose a synthesis of the target molecule from the starting material(s) provided Please be…
A: Step 1 : Hydrogenation. Step 2 : PCC oxidation of secondary alcohol. Step 3 : Acetlide addition to…
Q: Please explain the mechanism of the following reaction and why these products are formed in equal…
A:
Q: Me Me n-BuLi Me Me SnBu3 Hexane(s) O °C, 57% (2 steps) See the Wittig Rearrangement-[2,3] Me HOʻ Me…
A:
Q: 4- Propose a synthetic pathway for each of the following compound Show all steps. Starting from…
A: The given compound has an alkyl group substituted at the para position. The first step in the…
Q: Match each reaction sequence to a product below. Assume any necessary workup. Answers may be…
A:
Q: a) HO OH from b) H3C, OH
A:
Q: HBY Cexcess) -> 4.
A:
Q: Propose a mechanism for the reaction shown below. ÇI OH ONH2 + CI Нeat NO2 NO2
A: NOTE: The product given is incorrect and the correct product will have NH2 group in the place of OH.…
Step by step
Solved in 5 steps with 6 images

- Explain the E2 mechanism (bimolecular elimination) of of Elimination ?Draw a stepwise mechanism for the following Robinson annulation. This reaction was a key step in a synthesis of the steroid cortisone by R. B. Woodward and co-workers at Harvard University in 1951.Please help with the following ochem mechanisms.... 1. Provide the stepwise mechanisms for the following reactions (see attached picture)

