Q: At 300 K consider a colloidal solution, M = 150 kg/mol, in a gravity field. If the concentration of ...
A: The concentration of colloidal solution can be expressed as: C=C0e-MgzRT (1)
Q: Use the Maxwell Relationships whether dz is an exact differential. dz= (x² + y²)(xdx + ydy)
A:
Q: In the laboratory a "coffee cup" calorimeter, or constant pressure calorimeter, is frequently used t...
A: Mass of Zn = 66.79 g
Q: Acetylene (C,H,) gas and oxygen (0,) gas react to form carbon dioxide (CO,) gas and water (H,O) vapo...
A: Given, Acetylene gas and oxygen gas reacts to form carbon dioxide gas and water vapor. Moles of C2H2...
Q: 1 The oxygen tank in a hospital respiratory unit has a pressure of 4840 mmHg. What is the pressure o...
A: 1. 1 mm Hg = 0.001316 atm
Q: O THERMOCHEMISTRY Understanding the definition of enthalpy Measurements show that the enthalpy of a ...
A: Equation for change in enthalpy (ΔH) is given by, ΔH = ΔE + PΔV ------ ( 1 ) ΔE = Change in ...
Q: Why is there a warning that all glassware must be dry when preparing an equilibrium solution?
A: Why is there a warning that all glassware must be dry when preparing an equilibrium solution?
Q: 8. Compute for the formal charge (FC) on each atom in the following compounds. a. H3N - BH3 b. [H2...
A: Since you have asked multiple questions, we will solve the first question for you. If you want any s...
Q: II. What intermolecular force exist in the following substances? 1. Oxygen gas with noble gases 2. C...
A: Answer Intermolecular force of attraction exsit in the follow...
Q: Use numbers to order the substances by decreasing strength of intermolecular forces. Let #1 be the s...
A:
Q: Name the highlighted chemical group in each molecule. Lewis structure name of highlighted group н н ...
A: The name of the functional group is given below
Q: Part II. Problem Solving: Solve the following problems. Show your complete solution. Express your fi...
A:
Q: IV. Determine if the solid sample is IONIC, COVALENT, MOLECULAR or METALLIC _1. Dry ice 2. Graphite ...
A: Ionic solid is composed of positive and negative ions. Ions having positive charge is called positiv...
Q: In an experiment, how is the basic character of amines are proven
A: A question based on amines that is to be accomplished.
Q: Submit Question 19.f of 33 Describe the structure, bonding, and properties of this organic functiona...
A:
Q: For many years, the extraction of gold - that is, the separation of gold from other materials - invo...
A: Given,
Q: nonpolar covalent bonds
A: In this question we have to choose correct option of the name of the bond that occurs between two at...
Q: Write the balanced chemical equations representing the following reactions. Identify the (a) type of...
A:
Q: P & LS A hot 101.6 g lump of an unknown substance initially at Substance Specific heat (J/(g•°C)) 16...
A:
Q: What I Can Do etivity 1.6: Solve the following stoichiometry problems 1. Using the following equatio...
A:
Q: How to find percentage of chlorine in agcl
A: Percentage of an atom is the ratio of molecular weight of atom present in the given molecule and Mo...
Q: An aluminum atom has a mass of 4.48 x10 -23 g and a small airplane has a mass of 5000. kg. Use this ...
A:
Q: Lewis structure name of highlighted group H H Н— С C н н H H H н— с. C С — Н н нн H – C= O н—с —н Н ...
A:
Q: Could you please balance and identify what type of reaction it is?
A: For a balanced reaction number of atoms of each element must be same on both sides.
Q: Restate the paragraph. The main objective of the laboratory activity was to make aspirin also known...
A: Apirin is commonly used to reduce pain, fever, or inflammation. The synthesis of Aspirin from salicy...
Q: Draw diagram and give detailed explanation of Nucleophilic aliphatic substitution reactions
A: Lewis bases are commonly as nucleophiles in an Nucleophilic aliphatic substitution reaction. Because...
Q: polar or nonpolar? at O polar
A:
Q: M* + e → M A A+ e- Answer Bank e+A A- reduction M M* +e- not balanced M+ M² + + e- oxidation A A +e
A:
Q: 3 Including activity coefficients, find [ Hg2*] in saturated Hg:Brz in 0.001 00 M KBr. Ans: 7.0 x 10...
A:
Q: topic: Proper Use and Calibration of the pH meter Calibration is done using buffer solutions every...
A: Calibration is done using buffer solutions every time the pH meter is used:
Q: CO2ET OH Me Ме LIAIH4 H.. Me. Me H.. Me Et20 RT Me Ме Ме
A:
Q: TMS n-Bu4N+ F, KF H МеCN Ме., Me.,, 80 °C, 2 h Me Me Me Me
A: In this reaction F- has given . And we knew that floride and silicon bond are very strong bond . So ...
Q: percentage by mass
A:
Q: 12. The mass and % abundance for the four isotopes of chromium is given in the table below. Isotope ...
A: Given,
Q: Given the standard enthalpy changes for the following two reactions: (1) 2Pb(s) + 02(g)→2P60(s) AH° ...
A: We have to calculate the standard enthalpy change for the reaction PbO(s) + Zn(s) → Pb(s) + ZnO(s).
Q: Energy A 30. Use the molecular orbital diagram to figure out the electronic configuration for N2. Th...
A: We have to use molecular orbital diagram to solve this problem
Q: 3/5 Calculating pressure-volume work A mixture of helium and hydrogen gas is expanded from a volume ...
A:
Q: 1. N, + 3H, 2NH3 (0) 2 (g) 2(g) + 2NH, » CO(NH,)2 + H,O) 2. CO. 3(g) (), 2(g)
A:
Q: How many hydrogen atoms are there in the formula C12H_ having 3 rings and 7 double bonds? and ClN2O3...
A: For a given organic compound., Degree of Unsaturation = C - H/2 - X/2 + N/2 + 1 C = Number of Carbo...
Q: а. NH2 OMe N. EtzN, THF b. Me2N Me2N 1. LIAIH4, THF 2. H3O*, H20
A:
Q: The partial pressure of oxygen is 0.20 atm at sea level at 25°C. Calculate its partial pressure at a...
A: Solution is shown below.
Q: what are the uses and application of acid batteries, cadmium batteries,metal hydride batteries,ion b...
A: Given:- what are the uses and application of acid batteries, cadmium batteries,metal hydride batteri...
Q: Use the concept of osmosis to answer this situation: What happens to the size of a sliced mango if i...
A: Osmosis is defined as the process in which there is movement of water from high water concentration ...
Q: Reaction Coordinate + BD B+ Potential Energy
A: Exothermic reaction is a reaction that releases energy from the system to its surroundings, usually ...
Q: 1. In a study of the reaction of pyridine (C,H;N) with methyl iodide (CH3I) in a ben- zene solution,...
A: Let us suppose a reaction-- C5H5N + CH3I ----> Product Then, Rate law can be written as, Rate = ...
Q: Calculate the effective nuclear charge for the outermost electron in an atom of calcium. Show your w...
A: Calculation of effective nuclear charge of Calcium ?
Q: Please explain the mode of action on the hair of the following temporary and permanent hair styling ...
A: There are lot of hair styling tools that are used now a days
Q: Acetylene (C,H,) gas is often used in welding torches because of the very high heat produced when it...
A:
Q: Provide the product expected from each of the reactions below. If you believe that two products will...
A:
Q: 5. Consider equal amount of wood and glass. Assuming that equal amount of heat is absorbed by these ...
A: Heat capacity is the amount of heat required by one gram of a substance to increase its temperature ...
For this step of the synthesis, draw the arrow pushing mechanism for the following reaction. Include stereochemistry.
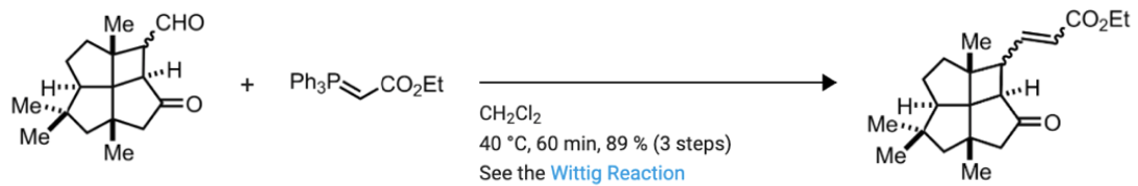
Step by step
Solved in 3 steps with 2 images

- Given the following information, calculate the reaction velocity at saturating substrate concentrations.Your numerical answer is assumed to be in units of M sec-1. [S] = 100 mM k1 = 10 sec-1 k2 = 3000 sec-1 k-1 = 20 sec-1 [E]T = 1 \muμMGiven the following information, calculate the reaction velocity at saturating substrate concentrations. Your numerical answer is assumed to be in units of M sec-1. [S] = 100 mM k1 = 10 sec-1 k2 = 3000 sec-1 k-1 = 20 sec-1 [E]T = 1 \muμMC. Purification of Water 1.For this part of the experiment, watch the video on the link(s) below: a. Flocculation: https://www.youtube.com/watch?v=5uuQ77vAV_U&t=82s Question: Observation before flocculation. Observation after Flocculation
- Table 2: Molarity of H2O2 and KI and Reaction Rate Trial H2O2 Concentration, M KI Concentration, M Reaction Rate(Reciprocal Slope) 1 0.29 M 0.40 M 14.08 2 0.29 M 0.20M 25 3 0.023 M 0.40 M 20 Please help me with this part, and please show me all steps Order with respect to H2O2: Order with respect to KI:Describe type I mechanism in PDT? please asap within 15 minIn Experiment 4- Chemical Kinetics “The Rate of Chemical Reaction”, the following data were obtained: Rxn Mixture time(s) temperature (˚C) 1 190 23.0 2 378 23.0 3 97 23.0 4 205 23.0 5 44 23.0 6 90 35.0 Concentrations and portions for reaction mixture number one: H2O 0.050 M Buffer 0.050 M KI (mL) 0.1% Starch (mL) 0.0450 MNa2S2O3(mL) 0.8525 M H202 (mL) 75.0mL 30.0mL 25.0 5.00 5.00 10.00 Use the data above to answer the following questions where applicable. (a) Write the formulas of the reactants, there are three, in the reaction being studied.Signs + or - or subscripts may be entered all on the same line; S2O32- can be entered as S2O3^2-, the symbol, ^, is not required if the…
- provide full reaction mechanismsTable 2: Molarity of H2O2 and KI and Reaction Rate Trial H2O2 Concentration, M KI Concentration, M Reaction Rate(Reciprocal Slope) 1 0.29 M 0.40 M 14.08 2 0.29 M 0.20M 25 3 0.023 M 0.40 M 20 help me with this part please, and show me all steps Order with respect to H2O2: Order with respect to KI:An enzyme-catalyzed reaction has a Km of 1.4 mM and a Vmax of 7 nM/s. What is the initial velocity when the substrate concentration is 0.6 mM?
- Create an illustration showing the preparation of two-fold serial dilution follows the ratio of the final dilutions as shown in the table above.If a mixture was prepared by diluting 7.0 mL of the Blue#1 solution with water to a final volume of 10.0 mL and mixing this with 2.0 mL of diluted bleach how many seconds would it require for the mixture to have an absorbance of 0.120? Use your measured values of b and of the average rate constant, k’ to answer this question. Show all work. Pls help me out asap!!Is the product pure? how do you know? here is the results: Starting mass of benzyl 1.0883g Collected mass of product 0.8957g Product melting point 135˚C Mixture melting point (product mixed with benzoin) 114˚C Mixture melting point (product mixed with meso-hydrobenzoin) 135˚C

