Ме, Мe Si., НО Ме HO `Me HO H КCN, HCN Ме `CI Me `CN PPTS EtzN MezSio Me Me,SiO Me NH2 OH Me LIAIH4 H2SO4 aq. `CN H3O* benzen Dean-Stark pH = 6
Q: Use the information in the pK, table below to determine which side of the equilibrium is favored for…
A: Given : Table showing the pKa values To find : Complete the given table Solution : Acidic strength…
Q: Which of the following is most weakly basic? O CH3CO2 О но- O CH3CH2O¯ O CH3O"
A: A substance which accepts a proton is known as base. The substance which is formed after the…
Q: a) KBr CH3CH,CH,I SN1 SN2 no reaction likely b) CH;CH,OH + H,O SN2 no reaction likely c) CH-CH,ОН…
A: The SN1 reaction is a substitution nucleophilic unimolecular reaction. It is a two-step reaction. In…
Q: What steps would you need to take to calculate the pKa1 and pKa2 of something? For example:…
A: We can use acid-base titration experiment to find the pKa value of any acid: monoprotic, diprotic,…
Q: The major product formed in the reaction below is:
A:
Q: In the Micheal addition below, which carbon is most likely to lose a proton to the base? H₂C A OA D…
A:
Q: Rank the following organic compounds below in terms of increasing acidity, with 1 being the least…
A: Here the acidity is dependent on the ease of donation of hydrogen ion.
Q: Propose mechanisms for the conversion of A to B and of B to C. What is the function of sodium amide…
A: Bidisomide, is an antiarrhythmic drug and was selected for development from a series of chemically…
Q: Rank the following protons in order of increasing acidity. OH CH2CH Ha Hc A. H, <H<H, B. H, <H, < H…
A: Applying mesomeric effect and inductive effect.
Q: Rank the following active methylene compounds in order of decreasing acid strength. most acidic…
A: Acidity depends stability of conjuagate base formed after loss of proton Stability of conjgate base…
Q: Which of the following compounds is most basic? II IV
A: The basicity of atoms decreases along a period in the periodic table. Hence, the decreasing order of…
Q: For the following reaction, which is the acid, which is the base, which is the conjugate acid and…
A: Acid and base: In an acid-base reaction, a species that donates proton is an acid while the species…
Q: 1.) LIAIH, NH 2.) H,O* a.) b.) c.) но. NH NH Racemic d.) e.) но HO,
A:
Q: 2. Calculate the pOH for the following: a. [H*]= 1x 10³ M b. [OH]=1x 10$ M c. [H']=1x 10-7 M %3D
A:
Q: Please rank the following molecules in terms of their basicity. Assign 1 to the most basic molecule,…
A: The compounds given are,
Q: Propose a reagent for the conversion of B to C.
A: The given compound B contains a carbonyl carbon. The desired compound C contains an alcohol group in…
Q: Rank the indicated atoms from the most (1) to the least (3) basic. C ONH NH2 В
A: Basicity is the ability of any atom to donate electron easily Greater the ease of donation more…
Q: Supply the structures of compounds J–U in the following synthesis scheme.
A: The free radical halogenation reaction produces a haloalkane via the generation of the intermediate…
Q: Arrange the compounds in each set in order of increasing acid strength. consult Table 4.1 for pKa…
A: Introduction: The acid or base form after the losing or gaining a proton is called conjugate acid…
Q: In the following reaction in aqueous solution, the acid reactant is and its conjugate base product…
A: An acid is a substance that gives ions in its solution whereas a basic substance gives ions in its…
Q: СООН СООН СООН СООН Br NO2 CH3 CI I II III IV
A:
Q: pH of unboiled H2O: 5.90 [H+] for unboiled H2O [OH−] for unboiled H2O
A: In our daily life, importance of pH is there. The Importance of pH is everywhere in food and…
Q: a. tor each pair, State and explain which is mote basic : (t) (r) b. tor each pair Enantiomer,…
A:
Q: 4. Predict the product of the nitration reactions CH3 HNO3 H2SO4 CI of HO, CHO N. LOCH2CH2CH3 Br2…
A: Acidity of the compound depends on the number of electron withdrawing groups attached to it. If the…
Q: a. m-chloroethylbenzene I.CH.CCI AICI, ZaHg) H,NNH, or HCL, A HO, A CH CH,CH, CH, 2 HO or H Pd/C CI…
A: We have find out mechanism for both reactions of a and b.
Q: (b) Calculate the pH of 0.0005 mol dm³ ethanoic acid when its pK₁ = 4.75 CH3COOH → CH3COO(+ H* K₁ =…
A: Given, [CH3COOH]=0.0005 mol dm-3 pKa=4.75 pH=?
Q: Which of the acids below would have the lowest ka value O CH3CICHCO2H O CH3CH2CH2OH O CH3CH2CO2H O…
A: Acidic character is directly propotional to ka.
Q: Which molecule has the most acidic proton? لا iioM OH ند C شد ه مید
A: The acidic proton means the proton that can be easily transfer or can be easily donated. The…
Q: acetyl chloride 2-methylproanoyl chloride 1. Hg(OAc)2, H20 2. NaBH4 SoCl2 NAOH Jones HCI/ Sn AICI3…
A: The reaction taking place is given as,
Q: Rank the following compounds in order of increasing acidity, putting the least acidic first. CH:COOH…
A:
Q: The following syntheses have flaws in them. What is wrong with each? (a) .CH3 1. Cl2, FeCl3 .CO2H 2.…
A: 1) Methyl group is a moderate electron donating group and it directs the incoming electrophile…
Q: TXT 9.2 Rank the labeled H's in the following molecules in order of increasing acidity (1 = least…
A:
Q: HB On the compound on the right, note that four hydrogens are designated with subscripts as HA, HB,…
A: If the conjugate anion is more stable, the compound will be more acidic.
Q: CH3NH3+ + H20 CH3NH2 + H3O+ For the equilibrium below show which ones are acid and which ones are…
A: Bronsted Lowry acid base theory- Acids are proton donor and bases are proton acceptors. Conjugate…
Q: Tepkime: H(g) + BR,(g)--> HBr(g) + Br(g) O 1.13 kJ mol-1 O 7.19 J mol-1 O 7.19 kj mol-" -1.13 kJ…
A: For the reaction given below, the arrhenius actication energy is 15.5 kj.mol-1. Then Find Δ≠ H0
Q: CHO CH,CCH, NaOH CH CHCCH=CH• MW 40.01 Dibenzalacetone Acetone Benzaldehyde MW 106.13, bp 178°C den.…
A: This reaction is an example of aldol condensation. Here base abstracts alpha hydrogen from carbonyl…
Q: IR order of decreasing acidity. OH> HO, HO, H-CO H3C .జ ఒప ఎంగడిలక క- OH > HO. HO. он O2N H3CO H3C…
A:
Q: D. H,O, H* 9. Н, Pt 10 cold dilute neutral KMNO4 H3C CH3 11 1. mCPBA 2. Н,О, Н" 12 13 mCPBA
A: Alkene double bond shows addition Reaction due to presence of pi electron density
Q: 1) CO2 (s) g) Ph(CH2)MgBr 2) 11 (aq) H'(aq/A h) 6-Bromohexanoic Acid + NACN - 1) NaOEt/E:OH i)…
A: Solution: g) Grignard reagent react with co2 in presence of acidic condition corresponding…
Q: Below is the structure of chloroquine. Circle the basic sites on the molecule.
A:
Q: Label the most acidic hydrogen in each molecule and justify your choice by using appropriate pKa…
A: Given molecule:
Q: Complete the following acid-base reaction . Show all valence electrons on the interacting atoms and…
A: Concept introduction: Curved arrows: A curved arrow points from the electron donor to the electron…
Q: Arrange these compounds in terms of increasing acidity: HF<CH3CH2CO0H< CH3CH2 CH2OH< CH3CH2COOH2+ )…
A:
Q: te lacionl NH2 H HO ОН O ad Chk Se Aosors to e ol aers I'
A: -> Atom are group of atoms which is attached to an organic compound and it is responsible for…
Q: Rank the following organic compounds below in terms of increasing acidity, with 1 being the least…
A: Acidity of any compound depends on the stability of conjugate base formed . More stable is the…
Q: LDA CH;-I B 0°C NH,CH;CH;NH; + SOCI; D + E HO H Ag;0, NaOH H;O* H;O. A LIAIH, H;0 CH;CH,CN H Eter H*…
A: When a carbonyl group with alpha hydrogen is treated with a base, enolate will be formed. The…
Q: Rank the following organic compounds below in terms of increasing acidity, with 1 being the least…
A: In this question, we will arrange all four Compounds in Increasing order of Acidity from least…
Q: LDA CH;-I B 0°C NH,CH;CH;NH; + SOCI; OH D + E Ag-0. NaOH H;O" G HO. A LIAIH, H;O CH;CH,CN H Eter H"…
A: Introduction: The first reaction is substitution of methyl group on the cyclohexane ring. The LDA…
Q: Rank the labeled H's in the following molecules in order of increasing acidity (1 = least acidic, 5…
A:
Indicate the complete mechanisms for each step of the synthesis below.
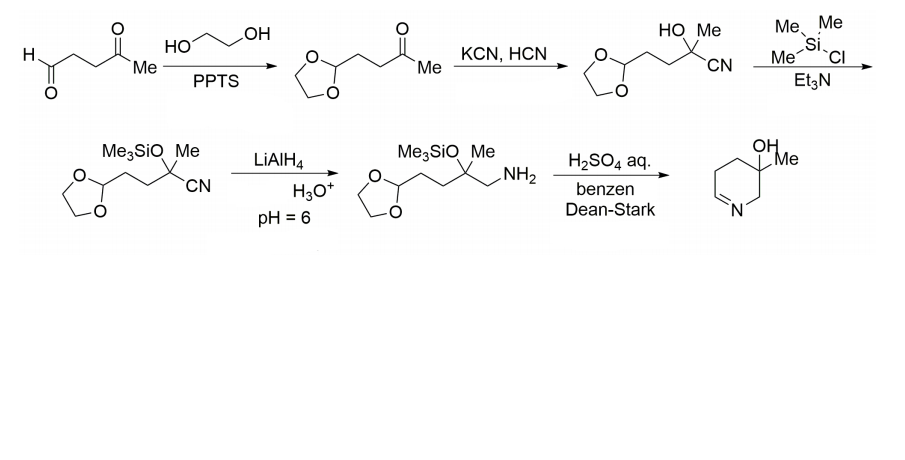
Step by step
Solved in 2 steps with 2 images

- please provide the machanisms of 1a, 1e, 1fWe have a hydrocarbon, C16H26. The compound has two triple bonds and the acids produced by ozonolysis are CH3(CH2)4CO2H and H2OCCH2CO2H. What is a likely structure for this hydrocarbon?. Discuss the truth of the following statement. Explain why it is true or false Every SN1 reaction produces racemic mixtures in the products
- Polymer PXX has a glass transition temperature of 49°C. Describe the molecular conformation and arrangement of this polymerI’m currently trying to write a lab report for the synthesis of dimolybdenum tetraacetate [Mo2(O2CCH3)4] from the reaction of molybdenum hexacarbonyl, Mo(CO)6 in glacial acetic acid and acetic anhydride under a nitrogen atmosphere, involving the difficult formation of a quadruple bond and requires high heat and long reaction time (approximately 20 hours). But these are the question I’m stumped on: 1. Why need the reaction be done under nitrogen? We also added dichlorobenzene and hexanes during the reaction. 2. Explain the purpose of dichlorobenzene and hexanes. 3. Why does the reaction take 20 hrs?Calculate ΔSsys° (J/K) for the catalytic hydrogenation of acetylene to ethane: C2H2(g) + H2(g) → C2H4(g) Substance S° (J/K·mol) C2H2(g) 200.8 H2(g) 130.58 C2H4(g) 219.4
- A compound of formula C6H10 takes up 1 mole of H2 on reduction. Give one possible structure that would fit the information.An unknown hydrocarbon Q has a formula C6H12. Q reacts with osmium tetroxide to give a diol R when oxidized with KMnQ4 in an acidic medium Q gives two products. One product is propanoic acid and the other is ketone S. Provide reaction equations to identify the possible structures of Q, R and S.Cumene -> i. Butyl chloride ii. Dimethyl amine iii. H2, Pt Provide the bond-line structures for the major organic products obtained in each step
- In the reactions given below, write to which organic molecule Y, Z, T, A ‘belongs, and give information about the reaction types that take place.What would be the expected major product when cyclooctene reacts with Cl2 in aqueous solution with H2O as the solvent?Kindly identify the product. Show its stable structure. ty this is rush and the topic is alkenes, alkynes and benzenes

