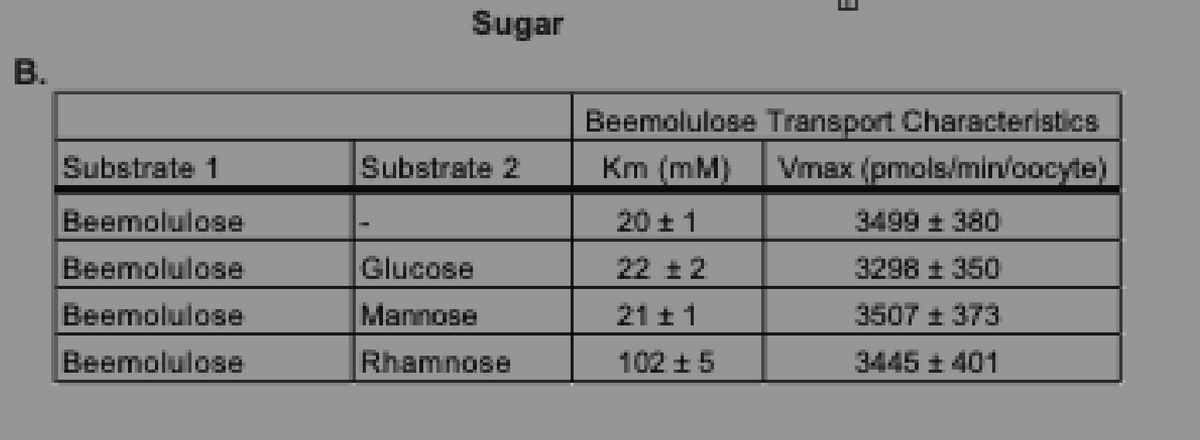
Measurement of beemolulose transport kinetics into oocytes in the presence of different sugars. 1) Analysis the table 2) The effect of each substrates into Km and Vmax
Q: How does this help the crocodile obtain its dinner?
A: Haemoglobin Haemoglobin is a red colour pigment that gives red colour to the blood. The main…
Q: Explain why 02 can pass through the plasma membrande but ions must use transmembrane proteins
A: Oxygen & carbon dioxide get across cell layers by means of simple diffusion, a cycle that needs…
Q: Hypothetical scenerio: When the membrane potential is 80mV across the digestive vacuoles (DV),…
A: All cells have an electrical potential difference or membrane potential across their plasma…
Q: why should triacylgycerol be insignificant component of plasma membeane
A: The fate of fatty acids present in the body of an organism is either to get incorporated into the…
Q: ending with the reduction of O2. 3. Indicate the sites and stoichiometry (per 2e) at which protons…
A: There are four complexes in electron transport chain which are arranged in the Inner mitochondrial…
Q: dysregulation/impaired function for each of these. Hypothetically, how can you rescue the…
A: G protein coupled receptors is a trans membrane domain of 7 helix meaning that they cross the cell…
Q: Suggest reason why iorganic ions such as K ,Na ,Mg ,Can do not cross biological membranes by simple…
A: Simple diffusion is the process by which solutes are moved along a concentration gradient in a…
Q: Describe the hallmarks of the GLUT transporter – a passive glucose transporter, and the basis behind…
A: A transmembrane glycoprotein called glucose transporter (GLUT) allows carbohydrate to cross the…
Q: calculate the actual free energy of hydrolysis of ATP, delta Gp in the erythrocytes of a new…
A: according to question we have to find the actual free energy of hydrolysis of ATP, delta Gp in the…
Q: Here is a chloride cell in the gill epithelium of a fish. For reference, NKA = Na+/K+ ATPaseNKCC =…
A: The chloride cells are present in the gills of freshwater and seawater fishes. In the seawater fish,…
Q: xplain why this is a serious concern and what this would mean for an organism
A: There are many toxins which can block the sodium channels, these toxins act by binding to the…
Q: Hemoglobin glycation (so named to distinguish it from glycosylation, which is the enzymatic transfer…
A: The test of glycated hemoglobin is used to determine the level of glucose present in the blood. The…
Q: In the blood coagulation pathway, several proenzymes are activated by unique proteolytic cleavage…
A: Blood Coagulation is a series through which the blood clotting enzymes are activated in order to…
Q: Define hypocapnia and hypercapnia. Name the pHimbalances that result from these conditions and…
A: Carbon dioxide is transported in the blood via three mechanisms: bound to hemoglobin, dissolved in…
Q: Tumor cells often lack an extensive capillary network and must function under conditions of limited…
A: Tumor cells: These are damaged or abnormal cells that multiply in numbers, resisting death.
Q: ince in this patient, pyruvate kinase is abnormal not only is less pyruvate made but intermediates…
A: Introduction: Pyruvate kinase is the last enzymatic reaction in the glycolysis pathway. It is…
Q: In one of the early tests of the chemiosmotic model, formation of ATP was assayed in a suspension of…
A: The "electron transport chain" is a sequence of redox reactions in which electrons are transferred…
Q: the acclimatization of B.subtilus to degrade phenol
A: B.subtilus is a rod-shaped, gram-positive bacteria with a tough, protective endospore that helps it…
Q: A cell’s ATP-driven sodium-potassium pump fails. Will thecell undergo crenation of hemolysis?
A: A Na+-K+ (sodium-potassium) pump is present in the cells and is driven by the energy produced by ATP…
Q: Explain why the large, steroid hormone, estradiol (MW 272) readily crosses membranes by simple…
A: Hormones are signaling molecules that act as chemical messengers. They are secreted by endocrine…
Q: which are the specific proteins that might be targeted for up or down regulate adsorption when…
A: Implantable materials are those that are used to design an implantable device. For, example…
Q: Cyclin B and and degraded by active APC/C. are ubiquitylated
A: The cell cycle includes the interphase and M phase.
Q: Discuss the operation of Na+ - K+ pump in further detail (2 pages including a few figures, if…
A: * The sodium potassium pump can be found in many cell plasma membranes which can be Powered by ATP…
Q: Describe the changes in clotting and bleeding balance during disseminated intravascular coagulation…
A: Disclaimer: As per Bartleby guidelines, we will answer only the first three subparts of the…
Q: Need help with number 2
A: General strains of E. coli are not able to cause them harm, due to the acidic medium in the stomach.…
Q: Carrier proteins are highly selective for theri transport molecules and tend to be more rapid in…
A: There are different types of transport proteins present on plasma membrane. The transport proteins…
Q: Atherosclerosis can obstruct or block flow in arteries, including those that provide oxygenated…
A: Artherosclerosis is a cardiovascular disorder caused due to build up of fat deposits in the walls of…
Q: oa obtain tne following data for the uptake of leucine and valine inside the cell with and without…
A: V0=Vmax[S]initial locationKt+ [S]initial locationHere V0 and Vmax are the initial and maximum rate…
Q: SUDH3W
A: The cell membrane consists primarily of a thin layer of amphipathic phospholipids that spontaneously…
Q: Lysozyme is a catalytic enzyme that cleaves NAG-NAM units in the bacte- rial cell wall, and is found…
A: Lysozyme is a naturally occurring enzyme found in secretions such as tears, saliva and milk. It…
Q: Number 3. I would say no burn can’t explain why’s exactly. IS it right? And how would I explain…
A: Escherichia coli abbreviated as E. Coli is a bacterium commonly found in the digestive system of…
Q: Calculate the actual free energy of hydrolysis of ATP, delta Gp in the erythrocytes of a new…
A: Actual free energy (∆G) is the maximum amount of energy which is available to perform work. Standard…
Q: Mammals excrete most nitrogen atoms as urea. The ureacycle itself is costly, requiring considerable…
A: The urea cycle or ornithine cycle is a cycle of biochemical reactions that produces urea from…
Q: Describe the hallmarks of the GLUT transporter – a passive glucose transporter, and the basis behind…
A: Glucose transporter 1 (or GLUT1) is involved in the facilitated transport of glucose across the…
Q: Correlate how the binding of 2,3-bisphosphoglycerate (abbreviated as 2,3-BPG or simply BPG) to the…
A: BPG is present in the Red blood cells. It is a highly charged anion. One molecule of BPG binds with…
Q: Glucose transporter present in erythrocytes is- A. GLUT 1 B. GLUT 2 C. GLUT 3 D. GLUT 4
A: Glucose transporter, transports glucose down the concentration gradient. Glucose transporter is also…
Q: O, saturation curve is shown below for hemoglobin at various pHs. Detail this Bohr effect from the…
A: Hemoglobin is an allosteric protein. It facilitates the transport of oxygen and carbon dioxide in…
Q: Suppose that a macro molecule(M) inside a dialysis bag with a net charge of +24 at a concentration…
A: Dialysis represents the process by which molecules in a solution can be separated based on the rate…
Q: During a temperature-programmed desorption (TPD) experiment it is observed that the temperature of a…
A: Gases expand on supplying heat. The same mole of a gas occupies a larger volume at high temperature…
Q: ignaling pathways are 10 hM (hanomoles per iter). Assuming that the average concentration of CAMP in…
A: Biological Signaling Pathways -- Organism can aware any event occurring around .There are some basic…
Q: What qualitative effect would you expect each of the following to have onthe P50 of hemoglobin?(a)…
A: Hemoglobin is a protein that binds and transports oxygen in the blood. The hemoglobin's binding to…
Q: Given: Factor VIIa is a man-made protein analog to Factor VII, which is involved in coagulation.…
A: Chloromethylketone (rFVIIa), and aprotinin are the competitive inhibitors of factor VIIa while…
Q: How a membrane transport protein powers the movement of Mimosa pudica. Please explain
A: Introduction: Mimosa pudica is a sensitive plant, belonging to the family Fabaceae. The plant…
Q: In an erythrocyte undergoing glycolysis, what would happen if there is a sudden increase in the…
A: AMP Adenosine Monophosphate a molecule that contains single phosphate group. AMP combine with two…
Q: Describing pharacokrelic properties mechanisms of action, chirical application pharmacologic and…
A: Pharmacology is a branch of science where drugs and their impact on the body are studied. Drugs are…
Q: the sodium channel exchanger NCX transports sodium into and calcium out of cardiac muscle cells.…
A: Active Transport across a lipid bilayer takes place when we have to move an ion against its…
Q: Briefly write down the working principle of methanobactin. And what are the function of MbnB and…
A: A chromophore is a compound which has the capacity to absorb and transmit the light energy. It not…
Measurement of beemolulose transport kinetics into oocytes in the presence of different sugars.
1) Analysis the table
2) The effect of each substrates into Km and Vmax
Step by step
Solved in 3 steps

- Assessing the Effect of Active-Site Phosphorylation on Enzyme Activity (Integrates with Chapter 15.) The serine residue of isocitrate dehydroenase that is phosphorylated by protein kinase lies within the active site of the enzyme. This situation contrasts with most other examples of coa1ent modification by protein phosphorylation. where the phosphorylation occurs at a sate remote from the active site. What direct effect do you think such active-site phosphorylation might have on the catalytic activity of isocitrate dehydrogcn.ise? (Sec Barford, D., 1991. Molecular mechanisms for the control of enzymic activity by protein phosphorytation. Biochimica et Biophysica Acta 1133:55—62.)Computation: Ratio Strength, PPM, mg%Show your complete solution.1. Diabetes is diagnosed by any of the following: two consecutive fasting blood glucose tests that are equal to or greater than 125 mg/dL, any random blood glucose that is greater than 200 mg/dL, and a two hour oral glucose tolerance test with any value over 200 mg/dL.A.) what is yje equivalent value express in terms of milligrams percent?Equivalent value of 126 mg/dL in mg% = ?Equivalent value of 200 mg/dL in mg% = ?B.) how many milligrams of glucose would be present in 10-mL sample serum (use the FASTING BLOOD SUGAR LEVEL)?Calculcate Kcat for PNP substrate for both enzyme concentrations. enzyme volume: 20 ul Bovine Intensince Alkaline phosphatase molecular weight: 140,000 Bovine intenstine Alkaline phosphatase activity: 300 units/ml and 14 units/mg extinction coefficient PNP: 18.5 abs (mM-1 cm-1) Vmax: 0.332 moles/sec a) enzyme 1 concentration: undiluted b) enzyme 2 concentration: 1:1 dilution
- a) Determine kcat (in units of sec-1) for a particular enzyme, given the following information: Vo = 144 mmol/min; [S] = 2 mM; Km = 0.5 mM; Enzyme Molecular weight = 40,000 mg/mmole; 8 mg of enzyme used in assay generating this data. b) In general, explain how the total enzyme concentration affects turnover number and Vmax?ΔG°′ for the isomerization reaction glucose-1-phosphate (G1P) ⇌ glucose-6-phosphate (G6P) is −7.1 kJ · mol−1. Calculate the equilibrium ratio of [G1P] to [G6P] at 25°C.6. Reciprocal regulation of opposing pathways is necessary to avoid the wasteful synthesis anddegradation of metabolic intermediates. Provide two distinct examples of reciprocal regulation. Bespecific, and be sure to explain the conditions that signal enzyme activation and/or inhibition.
- Suggest a reason why the cell membrane of bacteria grown at 20oC tend to have a higher proportion of unsaturated fatty acids than the membranes of bacteria of the same species grown at 37oMULTIPLE CHOICE In patients with Lesch Nyhan Syndrome, purine nucleotides are overproduced and over excreted. The hypoxanthine analogue Allopurinol, which effectively treats gout , has no effect on the severe neurological symptoms of Lesch- Nyhan patients because it does not A. decrease de novo pyrimidine synthesis B. decrease de novo purine synthesis C. decrease urate synthesis D. increase PRPP levels (Phosphoribosyl pyrophosphate)Describe the endogenous mechanisms that regulate cholesterol synthesis. Want solution ASAP
- Ketosis in Dairy Cattle: 1. Explain why propionate can contribute to the next synthesis of glucose but acetic acid cant.Molecular detail of spike Y453FMany enzymes obey simple Michaelis–Mentenkinetics, which are summarized by the equationrate = Vmax [S]/([S] + Km)where Vmax = maximum velocity, [S] = concentration ofsubstrate, and Km = the Michaelis constant.It is instructive to plug a few values of [S] into theequation to see how rate is affected. What are the rates for[S] equal to zero, equal to Km, and equal to infinite concen-tration?





